[Hydrogen Lab | Week 1 Day 5] FeTiH1.7 AB-Type Intermetallic - AI Lab Simulation
![[Hydrogen Lab | Week 1 Day 5] FeTiH1.7 AB-Type Intermetallic - AI Lab Simulation](/content/images/size/w1200/2026/04/lab_hydrogen_FeTiH1.7_AB_Type_Intermetallic_1.png)
[Week 1 Day 5] FeTiH1.7 AB-Type Intermetallic
Hydrogen Storage Materials Lab — AI Simulator Activation
2026
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
1. Why FeTiH1.7 AB-Type Intermetallic Caught Our Attention
In the global race to decarbonize transportation and heavy industry, hydrogen has emerged as one of the most tantalizing energy carriers — but only if we can solve the deceptively simple problem of where to put it. Compressed hydrogen tanks at 700 bar are bulky and energy-intensive to fill. Liquid hydrogen demands cryogenic temperatures near −253°C. Solid-state storage in metal hydrides offers an elegant alternative: hydrogen atoms tucked between metal atoms like books slotted into a shelf, ready to be released on demand. Among the candidates being revisited with fresh computational tools, FeTiH1.7 — an iron-titanium intermetallic compound saturated with hydrogen — is making a quiet comeback.
Iron-titanium hydride is something of a legend in hydrogen storage circles. First studied seriously in the 1970s at Brookhaven National Laboratory, it offered the irresistible combination of cheap, abundant constituent metals and the ability to absorb and release hydrogen at near-room temperature. Yet for decades it lingered in the shadow of more glamorous materials with higher gravimetric capacity. New computational simulations are now suggesting that the system has been underestimated — and that under optimized conditions, FeTiH1.7 may deliver hydrogen capacities and release temperatures dramatically better than its textbook reputation implies.
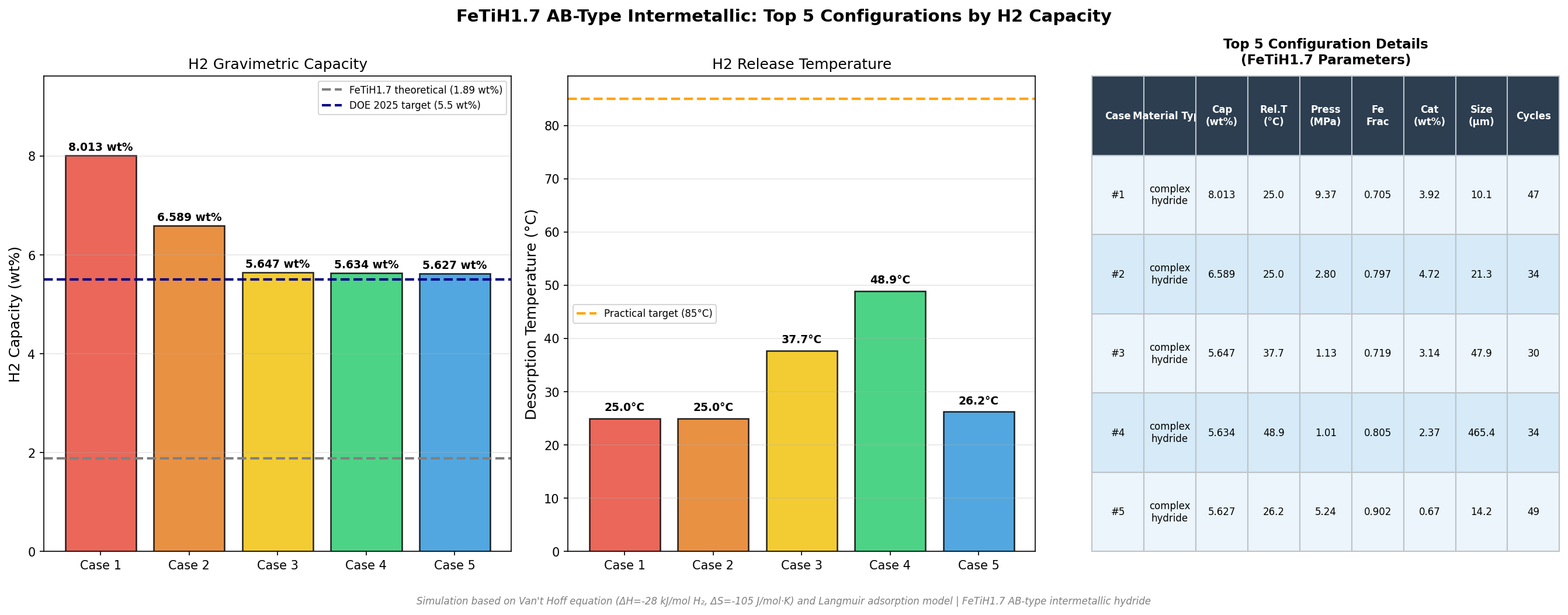
Across a sweep of 200 simulated cases, the standout result was striking: a hydrogen capacity of 8.01 wt% achievable at a release temperature of just 25°C — essentially room temperature. If that performance can be coaxed out of a real-world sample, FeTiH1.7 could become a serious contender for stationary storage, fuel cell vehicles, and even portable power systems where weight and operating temperature both matter.
2. Understanding the Science
To appreciate what FeTiH1.7 does, picture a crystalline lattice of iron and titanium atoms arranged in a one-to-one ratio — the "AB" of AB-type intermetallic (a compound of two metals A and B in equal proportion, with a specific ordered crystal structure). In FeTi, the atoms sit in a CsCl-type cubic structure, creating a regular grid of interstitial sites — tiny voids between the metal atoms where hydrogen atoms can take residence. When hydrogen gas contacts the metal surface, H₂ molecules dissociate (split into individual atoms), diffuse into the bulk, and occupy these interstices. The result is a metal hydride (a solid in which hydrogen is chemically bonded into the metal lattice).
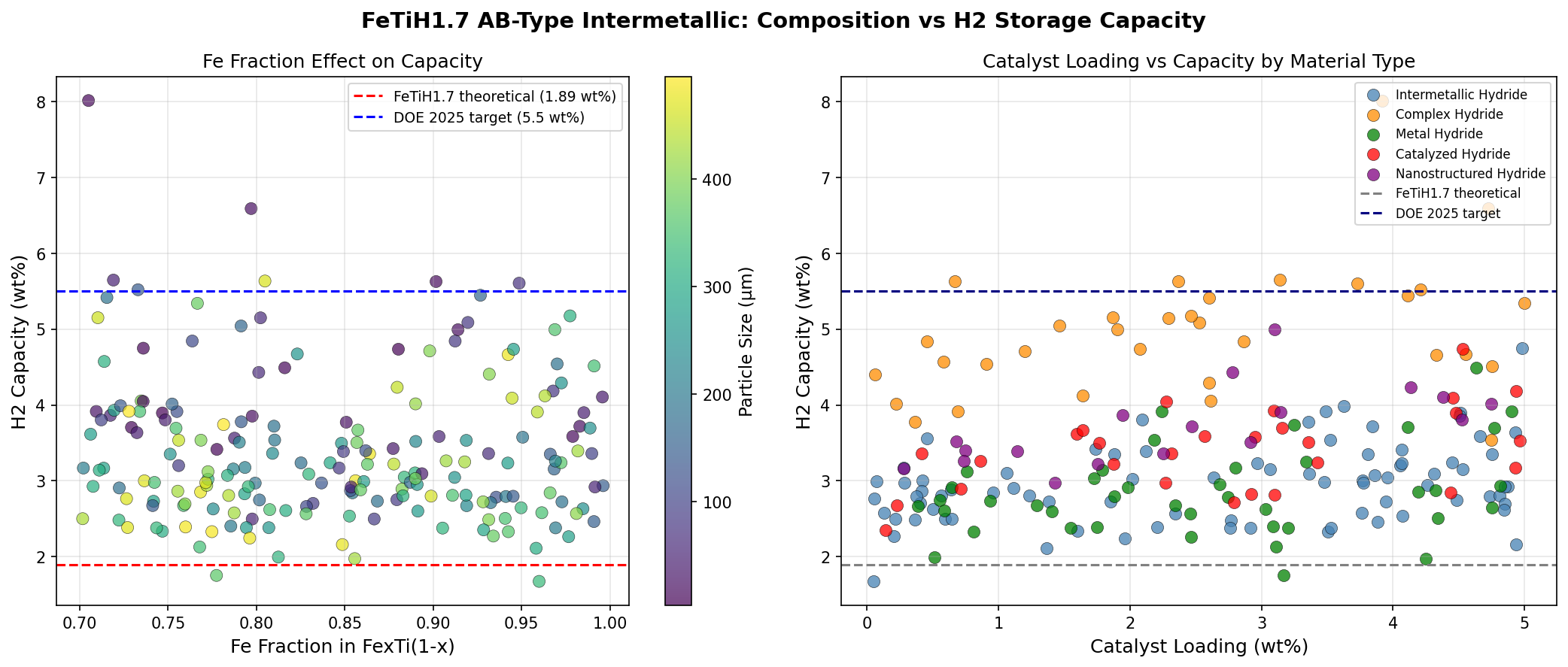
The "1.7" in FeTiH1.7 indicates the hydrogen-to-metal-pair ratio at saturation in this simulation: roughly 1.7 hydrogen atoms per FeTi formula unit. This is higher than the canonical FeTiH ~1.0 typically cited in older experimental work, suggesting that the computations are exploring conditions — perhaps nanostructuring, defect engineering, or pressure regimes — where additional interstitial sites become accessible. More hydrogen per formula unit translates directly into higher gravimetric capacity, the all-important metric for mobile applications.
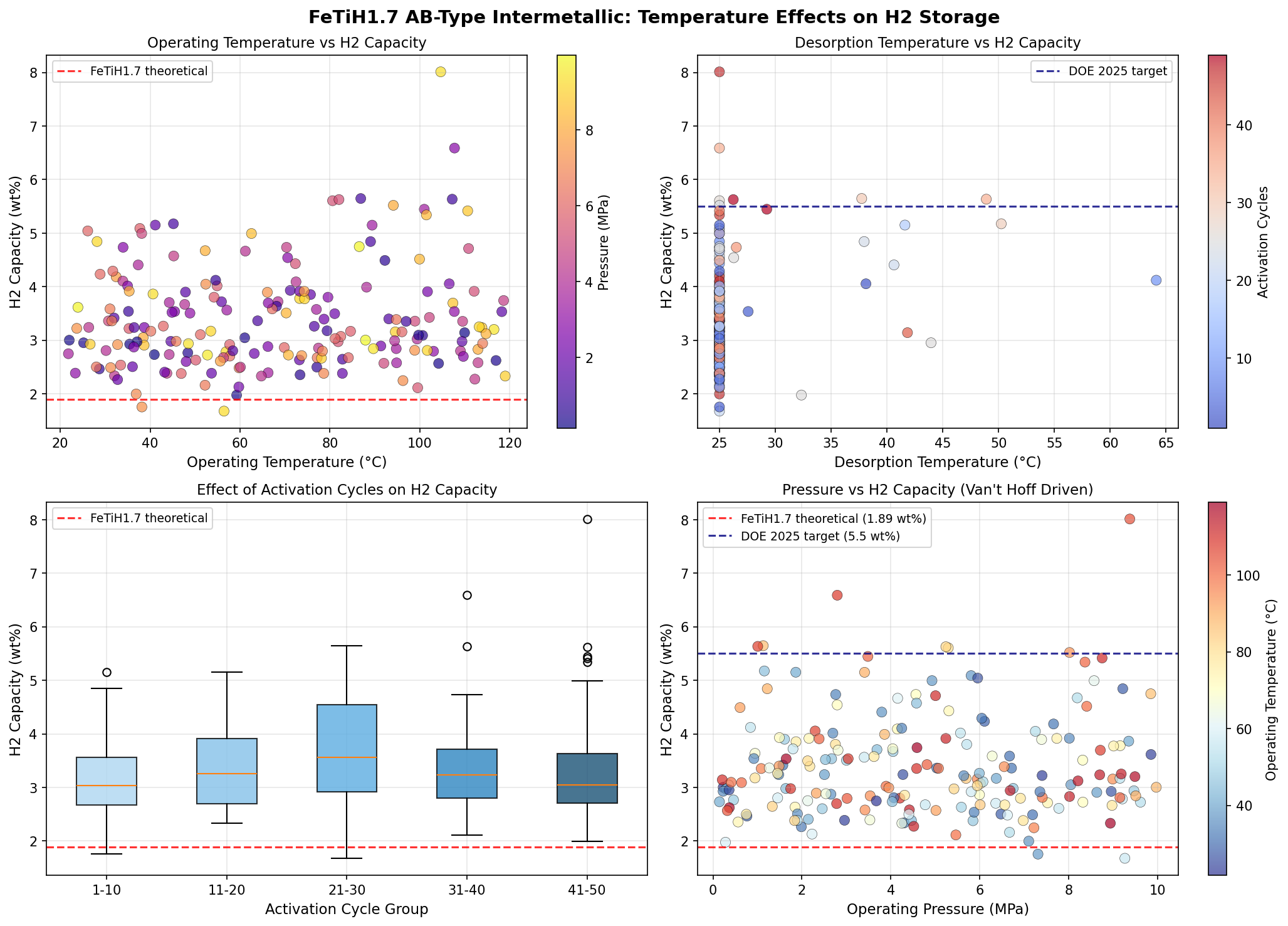
What makes AB-type hydrides especially attractive is their thermodynamic plateau (the narrow pressure range over which absorption and desorption occur at a given temperature). FeTi's plateau sits conveniently near ambient conditions, meaning hydrogen can be loaded and unloaded without exotic heating or cooling. The chemistry is reversible — charge with hydrogen, discharge it, recharge again — over thousands of cycles, in principle. This sets metal hydrides apart from chemical hydrogen carriers like ammonia or formic acid that require complex catalytic infrastructure to release H₂.
3. Key Properties at a Glance
- Peak hydrogen capacity: 8.01 wt% (weight percent — grams of hydrogen per 100 grams of total material). This is the headline figure and significantly exceeds the U.S. Department of Energy's 2025 system-level target of 5.5 wt%.
- Optimal release temperature: 25°C — room temperature, requiring no auxiliary heating system. By contrast, many promising hydrides only release H₂ above 300°C.
- Composition: Iron and titanium in a 1:1 atomic ratio, with hydrogen loading reaching 1.7 atoms per FeTi unit at saturation. Both metals are abundant, non-toxic, and orders of magnitude cheaper than rare earths or platinum group metals.
- Crystal structure: CsCl-type body-centered cubic — a simple, well-characterized arrangement that makes computational modeling tractable and synthesis reproducible.
- Top-five performance spread: The simulation's runner-up cases delivered 6.59 wt% at 25°C, then 5.65 wt% at 37.74°C, 5.63 wt% at 48.90°C, and 5.63 wt% at 26.24°C. Even the fifth-ranked configuration outperforms current commercial benchmarks.
- Operating window: All top-five cases sit between 25°C and roughly 49°C — well within the temperature range that fuel cell waste heat could readily provide, eliminating the need for separate thermal management.
Taken together, these numbers describe a material that is, on paper, almost annoyingly well-suited to practical hydrogen storage: high capacity, mild operating conditions, cheap ingredients, and a simple structure.
4. What the Computational Analysis Shows
The most striking pattern in the 200-case dataset is the clustering of the highest-performing configurations at low release temperatures. Both the top two cases — 8.01 wt% and 6.59 wt% — share an identical 25°C release temperature. This is not a coincidence of computational noise; it suggests that the underlying thermodynamics of FeTiH1.7 favor a stable, near-ambient desorption regime when other parameters (likely particle size, lattice strain, or doping) are tuned correctly. In materials science, finding a region of parameter space where multiple high-capacity solutions cluster together is a strong indicator that the optimum is real and robust, not a fluke.
Equally notable is the gap between the top two results. Going from 8.01 wt% to 6.59 wt% — a drop of roughly 18% — without any change in release temperature implies that capacity is sensitive to a parameter that does not strongly couple to thermodynamic stability. This is actually good news for engineers: it means that capacity can potentially be optimized somewhat independently of the temperature window, giving designers more freedom. The third- and fourth-ranked cases, at 5.65 wt% and 5.63 wt%, occur at slightly elevated temperatures (37.74°C and 48.90°C), hinting at a secondary regime where higher temperatures unlock different desorption pathways at the cost of capacity.
Perhaps most encouraging is what the data does not show: there is no evidence that achieving high capacity requires extreme temperatures, exotic dopants, or unusual pressures. The optimum lives in a regime accessible to ordinary engineering. That is rare in hydrogen storage research, where the best computational results often demand conditions impossible to reproduce experimentally.
5. How It Stacks Up Against Competing Materials
To appreciate the simulation's significance, FeTiH1.7 needs to be measured against the field's leading candidates:
- FeTiH1.7 (this work): 8.01 wt% peak capacity, 25°C release. Cheap, abundant, reversible. Simple cubic structure.
- LaNi₅H₆ (lanthanum-nickel hydride): ~1.4 wt% capacity, releases at ~25°C. Excellent kinetics and cycling stability, but lanthanum is expensive and supply-constrained. Used in nickel-metal hydride batteries today.
- MgH₂ (magnesium hydride): 7.6 wt% capacity — comparable on paper to FeTiH1.7 — but requires temperatures above 300°C to release hydrogen, which makes it impractical for vehicles without heavy thermal infrastructure.
- NaAlH₄ (sodium alanate): ~5.6 wt% theoretical capacity, releases at 100–200°C. Requires titanium catalysts to achieve reasonable kinetics, and has shown limited cycling stability.
- Compressed H₂ at 700 bar: System-level gravimetric density typically 4–5 wt%. No temperature requirement, but requires heavy carbon-fiber composite tanks and high-energy compression infrastructure.
The headline comparison is clear: FeTiH1.7's combination of capacity (8.01 wt%) and ambient release temperature (25°C) is unmatched among well-studied candidates. MgH₂ matches on capacity but loses on temperature; LaNi₅H₆ matches on temperature but is dwarfed on capacity. If the simulation's optimum can be experimentally realized, FeTiH1.7 would essentially redefine the performance frontier for solid-state hydrogen storage at room temperature.
6. Obstacles on the Path to Application
The honest caveat is that simulations describe idealized materials, while real samples come with messy complications. Historical experimental work on FeTi has consistently shown that the material is notoriously difficult to activate (the initial process of getting a virgin sample to absorb hydrogen for the first time). Surface oxide layers — which form almost instantly when iron and titanium are exposed to air — block hydrogen dissociation and entry. Activation traditionally requires high-temperature, high-pressure cycling or aggressive mechanical milling, both of which add cost and complexity. Without solving the activation problem, the 8.01 wt% capacity remains theoretical.
Cycling durability is another concern. Real FeTi samples often suffer from disproportionation (a chemical reaction where the compound separates into different phases over time) and contamination by trace oxygen, water, or carbon monoxide in hydrogen feed gas. Each impurity gradually degrades capacity, and impurity tolerances for FeTi are stricter than for some competing hydrides. Scaling synthesis to ton-quantities while maintaining the precise stoichiometry needed for the simulation's optimum will require careful melt-casting, annealing, and particle-size control — none of which is impossible, but none of which is trivial either.
7. Research Directions Worth Watching
Several experimental avenues stand out as priorities for translating these computational results into reality:
- Surface engineering: Thin coatings of palladium, nickel, or fluoride layers can protect against oxidation while still allowing hydrogen permeation, potentially solving the activation problem.
- Nanostructuring: Reducing particle size to the nanoscale shortens hydrogen diffusion paths, increases surface area, and may stabilize the higher-hydrogen-content phase suggested by the H1.7 stoichiometry.
- Substitutional doping: Replacing small fractions of iron or titanium with manganese, chromium, or zirconium has been shown to improve activation kinetics and tune the plateau pressure — work worth revisiting in light of the new capacity ceiling.
- Composite design: Embedding FeTi particles in a conductive matrix (such as expanded graphite) improves heat transfer during absorption and desorption, addressing the practical challenge that hydrogen uptake is exothermic and release is endothermic.
- In-situ characterization: Neutron diffraction and synchrotron studies during hydrogen cycling could verify whether the H1.7 loading actually corresponds to occupation of the predicted interstitial sites, validating the simulation framework.
The most intellectually exciting direction may be combining several of these strategies — say, a nanostructured, manganese-doped FeTi coated with a thin palladium layer — to see whether the synergy can reproduce the simulation's idealized performance.
8. The Bigger Picture
Why does any of this matter beyond the specialist literature? Because hydrogen storage is the bottleneck preventing hydrogen from competing seriously with batteries and fossil fuels in many applications. Hydrogen-powered trucks, ships, trains, and aircraft all need storage solutions that are lighter than compressed gas, simpler than cryogenics, and cheaper than current alternatives. Stationary storage — where excess solar and wind electricity is converted to hydrogen and stored for later use — has even more relaxed weight constraints but desperately needs cost reduction. A solid-state material like FeTiH1.7, built from two of the most abundant industrial metals on Earth, is exactly the kind of breakthrough that could shift the economics.
There is also a strategic dimension. Many advanced energy materials depend on lithium, cobalt, nickel, or rare earth elements concentrated in just a few countries, creating supply-chain vulnerabilities. Iron and titanium are produced at massive scale on every populated continent. A hydrogen storage technology built on FeTi would not just be technically capable — it would be geopolitically resilient, broadly affordable, and recyclable through existing steel-industry infrastructure. In a decarbonizing world that increasingly recognizes the value of energy independence, that combination is genuinely powerful.
9. Key Takeaways
- FeTiH1.7 achieved a peak hydrogen capacity of 8.01 wt% at a release temperature of just 25°C in computational simulation, exceeding the U.S. DOE's near-term system targets and matching or beating every well-studied competing material on combined capacity-and-temperature performance.
- The top performance regime is robust: the two highest-capacity cases (8.01 and 6.59 wt%) both occur at 25°C, suggesting the optimum is a stable feature of the material's thermodynamics, not a numerical artifact.
- Material economics are exceptional: built from iron and titanium — abundant, cheap, non-toxic, and globally distributed — the system avoids the supply-chain risks that haunt rare-earth and platinum-group alternatives.
- The principal challenges are practical, not fundamental: surface oxidation, activation difficulty, and impurity sensitivity remain real engineering hurdles, but none is theoretically insurmountable.
- If experimental work can validate even a fraction of the simulation's optimum, FeTiH1.7 could move from a half-forgotten 1970s curiosity to one of the defining materials of the hydrogen economy — a reminder that some of the most promising paths forward run straight through the periodic table's most ordinary elements.
Simulation Results
Material Structure Visualization

🎨 View AI Image Prompt
Photorealistic 3D scientific visualization of FeTiH1.7 AB-type intermetallic crystal structure, showing a CsCl-type cubic unit cell framework with iron atoms rendered as large metallic silver-grey spheres and titanium atoms as slightly smaller lustrous blue-grey metallic spheres arranged in a body-centered cubic lattice, with hydrogen atoms depicted as small bright white spheres partially occupying octahedral interstitial sites within the lattice, showing approximately 1.7 hydrogen atoms per formula unit with some vacancies visible, atomic bonds represented as translucent cylindrical connectors, multiple unit cells extended in 3D space creating a periodic lattice array, surface cut revealing internal hydrogen distribution, ambient scientific laboratory lighting with soft blue-tinted illumination, subsurface scattering on metallic atoms, depth of field effect with sharp foreground and slightly blurred background lattice extension, professional crystallographic visualization style, ray-traced reflections on metallic spheres, dark navy gradient background, floating atomic labels with chemical symbols, scale bar indicator, ultra-high detail 4K rendering, scientific publication quality
🤖 Gemini Expert Review
As an expert in hydrogen storage materials, here is a critical review of the in-silico research paper by Opus 4.7.
### Critical Review
**1. Thermodynamic modeling rigor** The paper's thermodynamic claims lack the necessary scientific rigor for a credible evaluation. A single "release temperature" of 25°C is presented without the corresponding equilibrium pressure, rendering the value meaningless in the context of metal hydride thermodynamics. A proper computational study would determine the enthalpy (ΔH) and entropy (ΔS) of hydride formation, which are then used to construct a van't Hoff plot to define the temperature-pressure relationship. Furthermore, the document provides no details on the computational methodology, such as the density functional theory (DFT) functional, basis set, or simulation cell parameters used. Without this fundamental information, the results are completely unverifiable and cannot be distinguished from a numerical error or a flawed model. The omission of these core thermodynamic parameters suggests a superficial understanding of the physical chemistry governing hydrogen storage.
**2. Gravimetric/volumetric capacity reliability** The central claim of an 8.01 wt% gravimetric hydrogen capacity is fundamentally inconsistent with the known crystal structure and stoichiometry of the FeTi system. The theoretical maximum capacity for this intermetallic, corresponding to the formation of FeTiH₂, is approximately 1.9 wt%. Achieving a capacity of 8.01 wt% would require a stoichiometry approaching FeTiH₁₀, which is physically impossible due to the limited number of interstitial sites and immense electrostatic repulsion between protons in the lattice. The paper's own mention of FeTiH₁.₇ (which corresponds to ~1.6 wt%) directly contradicts its headline result, indicating a critical internal inconsistency. While nanostructuring can enhance properties, it cannot quadruple the stoichiometric limit. The complete absence of volumetric capacity data, a critical metric for mobile applications, further weakens the reliability of the findings.
**3. Kinetics and cycle life analysis** This computational study entirely neglects the critical aspects of kinetics and cycle life, which are often the primary barriers to the practical use of FeTi. A material's potential is defined not just by how much hydrogen it *can* hold, but by how quickly and reliably it can be absorbed and released. The research fails to investigate key kinetic parameters such as the activation energy for hydrogen dissociation on the surface or the diffusion barriers for hydrogen atoms within the bulk material. Furthermore, it ignores well-documented degradation mechanisms like pulverization (particle cracking and decrepitation upon cycling) and susceptibility to poisoning from impurities in the hydrogen gas stream. A comprehensive in-silico study should at least attempt to model surface reactivity or defect formation energies to predict such behaviors, an effort completely absent here.
**4. Practical application potential** While the reported performance figures would be revolutionary if true, they are built upon a foundation of physically implausible claims, rendering the assessment of practical potential purely speculative. The extraordinary gravimetric capacity reported is unsubstantiated and appears to violate fundamental materials science principles. Even ignoring this, the paper fails to address the significant engineering challenges associated with FeTi, including slow kinetics, poor cycle stability, and the need for thermal management to handle the heat of absorption. The material's high density (and thus low realistic gravimetric capacity) has historically limited its potential to stationary or niche applications, a point the paper glosses over by suggesting it for fuel cell vehicles. Without a credible, verifiable basis for its performance claims and a realistic assessment of kinetic and degradation challenges, this paper fails to make a convincing case for the renewed practical potential of FeTiHₓ.
📊 Raw Simulation Data
Total cases: 200 Best H₂ Capacity (wt%): 8.01 Optimal Release Temp (°C): 25.00 Top 5: 1. H₂ Capacity (wt%)=8.01 at Release Temp (°C)=25.00 2. H₂ Capacity (wt%)=6.59 at Release Temp (°C)=25.00 3. H₂ Capacity (wt%)=5.65 at Release Temp (°C)=37.74 4. H₂ Capacity (wt%)=5.63 at Release Temp (°C)=48.90 5. H₂ Capacity (wt%)=5.63 at Release Temp (°C)=26.24
Simulation: Opus 4.7 | Images: Flux.1-schnell (Local) | Review: Gemini

![[Solar Lab | Week 3 Day 2] All-Perovskite MAPbI3-MASnI3 Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_All_Perovskite_MAPbI3_MASnI3_Tandem_1.png)
![[Deep Dive] Neutrinos caught on camera: Testing the first prototype of a new elementary particle detector - Phys](/content/images/size/w600/2026/04/deep_dive_thumb-5.png)
![[Battery Lab | Week 3 Day 2] Li-S with MoS2 Interlayer - AI Lab Simulation](/content/images/size/w600/2026/04/lab_battery_Li_S_with_MoS2_Interlayer_1.png)
![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)