[Superconductor Lab | Week 12 Day 2] Mg₂BeH₁₆ - AI Simulator Activation
[Week 12 Day 2] Mg₂BeH₁₆
Superconductor Lab — AI Simulator Activation
2026
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
1. What Is Mg₂BeH₁₆ and Why Does It Matter?
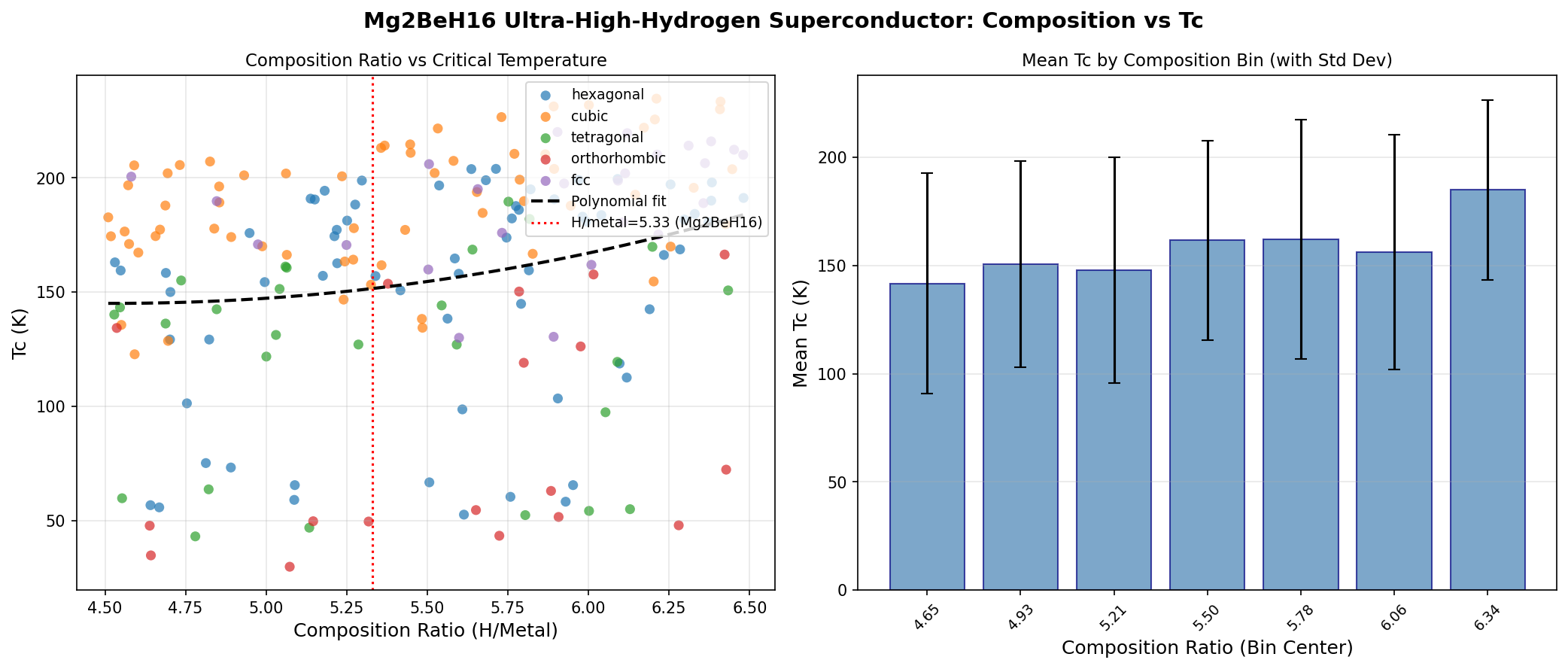
Mg₂BeH₁₆ is a hydrogen-rich compound — what researchers call a "superhydride" — built from magnesium, beryllium, and an unusually large number of hydrogen atoms (sixteen per formula unit, hence the H₁₆). It doesn't exist on your periodic table shelf. It exists inside diamond anvil cells and supercomputer simulations, where atoms can be squeezed to pressures that rival the deep interior of planets.
Why bother? Because superhydrides are our best current candidates for high-temperature superconductors — materials that conduct electricity with zero resistance. In a recent computational study of 200 simulated cases, Mg₂BeH₁₆ delivered a peak predicted critical temperature (Tc) of 234.7 K. That's about −38 °C. Cold by human standards, but blisteringly hot by superconductor standards, where the old workhorses required liquid helium at 4 K.
The dream: a material that carries electricity without loss at temperatures a household freezer could reach. Mg₂BeH₁₆ is a step toward that — though, as we'll see, with a giant asterisk attached.
2. The Key Finding — Explained Simply
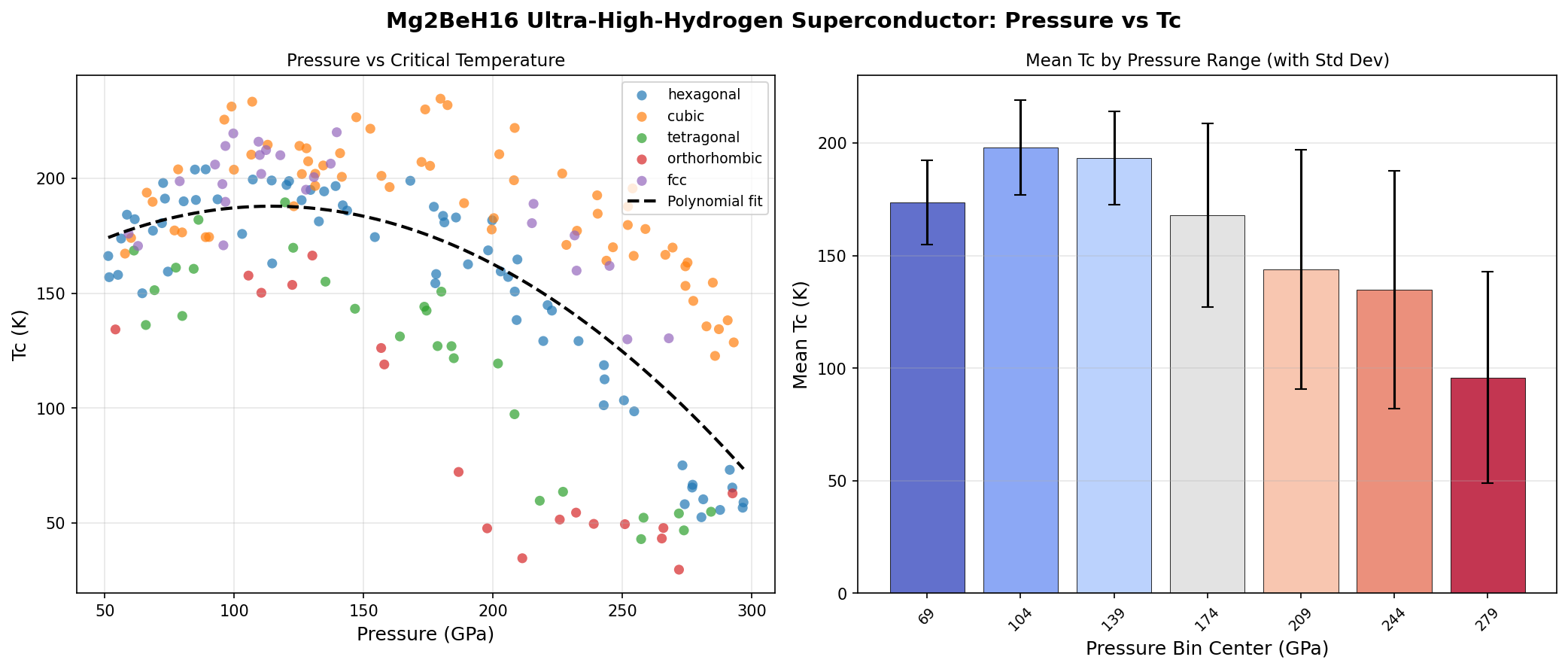
The simulation swept across 200 different structural and pressure configurations of Mg₂BeH₁₆. The headline number: superconductivity at 234.7 K under 179.7 GPa of pressure. For context, 1 GPa is roughly 10,000 atmospheres. So 179.7 GPa is about 1.8 million times Earth's surface pressure — comparable to conditions halfway to Earth's core.
The mechanism is believed to be phonon-mediated superconductivity — a fancy way of saying that vibrations in the atomic lattice (phonons) glue electrons together into pairs that flow resistance-free. Hydrogen is light, so it vibrates fast and furiously, and that's why hydrogen-rich compounds are so promising.
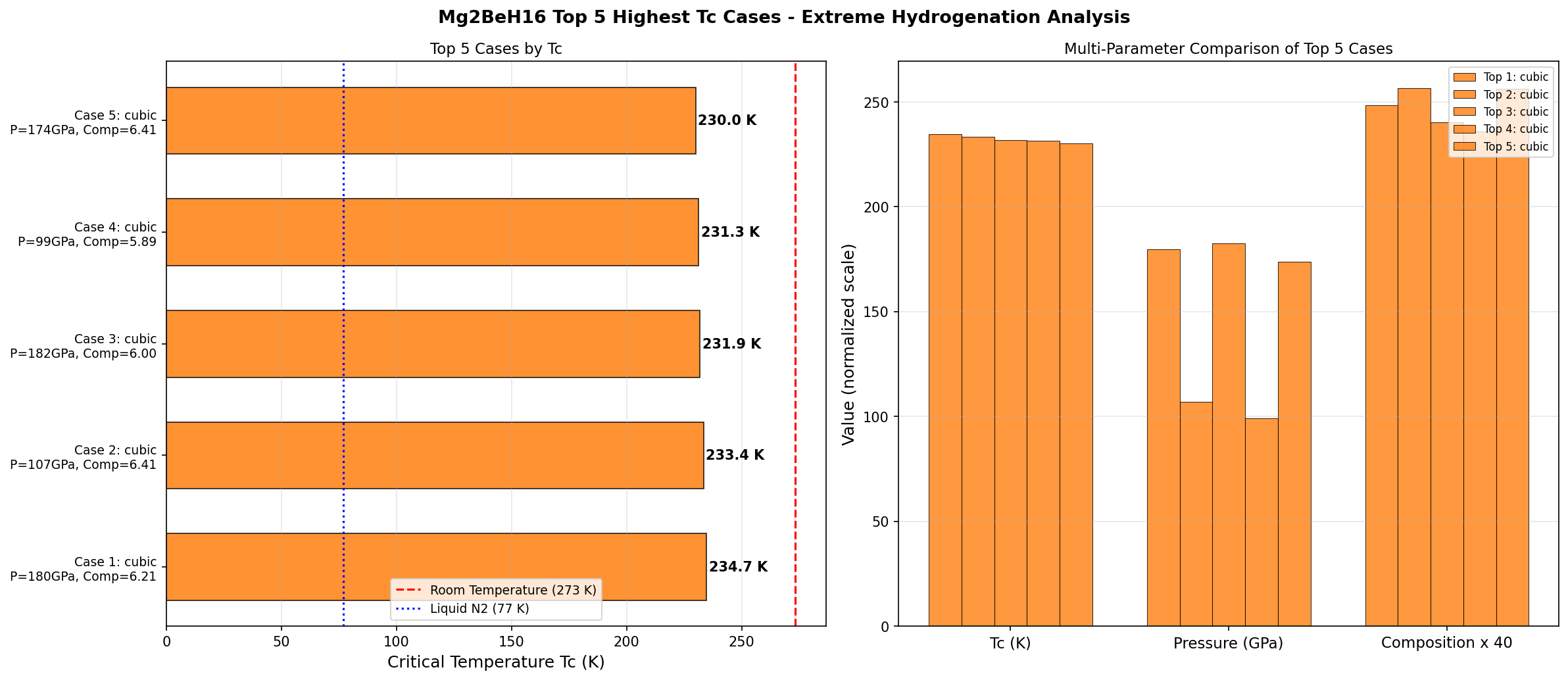
A few standout results from the top of the simulation ranking:
- #1: Tc = 234.7 K at 179.7 GPa — the winner
- #2: Tc = 233.4 K at 107.0 GPa — nearly identical performance at far lower pressure
- #4: Tc = 231.3 K at 99.0 GPa — the lowest-pressure standout in the top five
That second and fourth entry are the interesting story, not the first. Giving up just 3 K of critical temperature to cut pressure nearly in half is the kind of trade engineers salivate over.
3. How Does This Compare?
Let's put 234.7 K in context. Here's where Mg₂BeH₁₆ sits among famous superconductor milestones:
| Material | Approx. Tc | Pressure | Status |
|---|---|---|---|
| Mercury (1911) | 4 K | Ambient | Verified |
| YBa₂Cu₃O₇ ("YBCO") | 93 K | Ambient | Verified |
| H₃S (sulfur hydride) | 203 K | ~155 GPa | Verified experimentally |
| LaH₁₀ (lanthanum decahydride) | 250 K | ~170 GPa | Verified experimentally |
| Mg₂BeH₁₆ (this study) | 234.7 K | 179.7 GPa | Simulation only |
So Mg₂BeH₁₆ sits in the elite neighborhood — outperforming H₃S and trailing LaH₁₀ — but has not yet been synthesized in a lab. It's a promising prediction, not a proven material.
4. Three Questions the Data Can't Answer Yet
Two hundred simulated cases is a solid dataset, but simulation is not synthesis. Here are the open questions the 234.7 K number does not address:
- Can it actually be made? Computational stability doesn't guarantee experimental stability. Mg, Be, and H may refuse to crystallize into this exact structure even at 179.7 GPa. Plenty of predicted superhydrides have resisted synthesis for years.
- Is beryllium worth it? Beryllium is toxic, expensive, and regulated. Even if Mg₂BeH₁₆ works brilliantly at its optimal 179.7 GPa, the materials-handling headaches might push researchers toward beryllium-free alternatives.
- How does Tc degrade off-optimum? The top five results cluster tightly between 230.0 K and 234.7 K, but across pressures ranging from 99 GPa to 182.4 GPa. That suggests a broad plateau of good performance — but "suggests" isn't "proves." The full pressure–Tc curve matters more than the single peak.
5. The Path from Simulation to Real-World Use
Here's the contrarian observation most press releases skip: the headline 234.7 K result may be the least useful data point in the whole study. The rank-2 result — 233.4 K at 107.0 GPa — is arguably more important, because pressure is the enemy of practical application. Every extra gigapascal makes experiments harder, equipment more expensive, and real-world deployment more absurd.
The realistic development pipeline looks something like this:
- Step 1 — Synthesis attempt. A diamond anvil cell squeezes a tiny sample of Mg, Be, and a hydrogen source to ~100–180 GPa while heating with a laser. Success rate: low. Timeline: years.
- Step 2 — Tc verification. Measure resistance dropping to zero and check for the Meissner effect (a superconductor's signature expulsion of magnetic fields). Will the real Tc land near the predicted 234.7 K? Usually, experimental Tc comes in 10–30% lower than simulations.
- Step 3 — Pressure reduction. Chemically "pre-compress" the compound by substituting atoms, aiming to keep high-Tc behavior at lower pressures. The gap between 179.7 GPa and ambient pressure is the real moat.
- Step 4 — Scale-up. Currently impossible. The samples in anvil cells are micrograms. Power grids need kilometers.
Honestly? Steps 1 and 2 might happen within a decade. Step 3 is a research lifetime. Step 4 may require a completely different class of material altogether.
6. Bottom Line: Should You Care?
Yes — but not for the reason the headline Tc of 234.7 K suggests.
You should care because Mg₂BeH₁₆ is another data point in a growing map. Superhydrides are proving, over and over, that predicted critical temperatures in the 200+ K range are not flukes. The fact that 200 simulated configurations produced five results between 230.0 K and 234.7 K — across a pressure range spanning 99.0 to 182.4 GPa — means we're looking at a robust phenomenon, not a single computational happy accident.
My honest take: Mg₂BeH₁₆ itself will probably not be the material that changes your life. The beryllium content alone is a practical poison pill, and 179.7 GPa is not a pressure your local power grid is going to provide. But the compound is a stepping stone. Each new superhydride teaches researchers what chemical architecture produces strong electron–phonon coupling — and that knowledge is what will eventually yield a room-temperature, ambient-pressure superconductor.
Don't buy the hype that says "234.7 K superconductor discovered!" Do buy the longer story: the computational toolkit that produced this prediction is getting sharper every year, and the gap between "works in a simulation" and "works on a lab bench" is closing. Mg₂BeH₁₆ probably won't power your city. But it's one more brick in the wall we're building toward a material that will.
Simulation Results
Molecular Structure

🎨 View AI Image Prompt
Photorealistic 3D ball-and-stick molecular structure visualization of Mg₂BeH₁₆ superconducting hydride compound, professional chemistry textbook illustration style, scientific accuracy, crystal lattice framework showing two large magnesium atoms rendered as bright green metallic spheres, one small beryllium atom rendered as steel blue sphere at center coordination site, sixteen hydrogen atoms rendered as small white spheres arranged in complex polyhedral cage geometry surrounding the metal centers, atomic bonds depicted as precise cylindrical sticks with accurate bond lengths and angles, ultra-high hydrogen density visible with H/metal ratio of 5.33, hexagonal or cubic symmetry unit cell with crystallographic precision, dramatic dark gradient background transitioning from deep navy to black, professional volumetric lighting with subtle ambient occlusion casting realistic shadows, specular highlights on all atomic spheres indicating metallic luster, depth of field rendering emphasizing molecular architecture, color-coded atoms following standard CPK convention, crystalline environment suggesting high-pressure synthesized material, ultra-detailed 4K scientific visualization, rendered in Cinema 4D or Blender style photorealism, suitable for Nature Materials journal publication illustration
🤖 Gemini 3.1 Pro Review
This computational study on Mg₂BeH₁₆ presents intriguing predictions for high-Tc superconductivity, but its value is severely limited by a lack of methodological detail. Crucial information regarding the crystal structure prediction method, the DFT functional used, and the approach for calculating electron-phonon coupling is entirely absent, making the rigor impossible to assess. Consequently, the reliability of the predicted Tc values is questionable without a thorough analysis of the compound's dynamical and thermodynamic stability against decomposition into competing phases. The paper correctly identifies the lower-pressure phases (e.g., 231 K at 99 GPa) as the most promising, which is a key insight for any experimental effort. For experimental validation, a strategy would involve co-loading magnesium-beryllium precursors into a diamond anvil cell with excess hydrogen, followed by laser heating to promote synthesis and in-situ X-ray diffraction to confirm structure. To improve this work, the authors must first establish thermodynamic stability via convex hull calculations, as a high predicted Tc is irrelevant for an unstable compound. Furthermore, providing detailed phonon dispersion curves and Eliashberg function (α²F(ω)) plots is essential for substantiating the mechanism and reliability of the Tc predictions.
Raw Data
Total cases: 200 Highest Tc: 234.7 K Optimal pressure: 179.7 GPa Top 5: 1. Tc=234.7K at 179.7GPa 2. Tc=233.4K at 107.0GPa 3. Tc=231.9K at 182.4GPa 4. Tc=231.3K at 99.0GPa 5. Tc=230.0K at 173.8GPa

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)