[Superconductor Lab | Week 11 Day 5] NaBe₂H₁₂ - AI Simulator Activation
[Week 11 Day 5] NaBe₂H₁₂
Superconductor Lab — AI Simulator Activation
2026
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
1. What Makes NaBe₂H₁₂ Interesting?
Imagine electricity flowing through a wire with absolutely no resistance — no heat loss, no wasted energy, no degradation. That's the promise of superconductivity, and for more than a century, scientists have been chasing materials that can do this not just at frigid, liquid-helium temperatures, but closer to the warmth of everyday life. A new computational candidate has entered the arena: NaBe₂H₁₂, a hydrogen-rich compound combining sodium (Na), beryllium (Be), and a remarkable twelve hydrogen atoms packed into a single formula unit. Recent simulations suggest it could superconduct at temperatures as high as 241.6 Kelvin — roughly −31°C, a temperature you might encounter on a cold winter day in Siberia.
While that might not sound toasty, it's extraordinary by superconductor standards. Conventional superconductors require cooling to near absolute zero, often below 20 K. A material operating above 240 K sits tantalizingly close to the holy grail of physics: room-temperature superconductivity. What makes NaBe₂H₁₂ especially intriguing is that it belongs to a fast-growing family of hydrogen-dominant superhydrides, where hydrogen atoms form cage-like networks that vibrate in just the right way to usher electrons into their lossless, paired state.
2. Understanding the Key Properties
To appreciate why NaBe₂H₁₂ is exciting, we need to unpack a few ideas. The critical temperature (Tc) is the threshold below which a material becomes superconducting. Above Tc, electrons bump around like pedestrians in a crowded market, losing energy to collisions. Below Tc, they glide through the material in coordinated pairs called Cooper pairs, completely ignoring obstacles.
What glues these pairs together? In conventional superconductors, the answer is electron-phonon coupling. Phonons are quantized vibrations of the atomic lattice — essentially, tiny ripples of sound traveling through the crystal. When an electron zips past, it tugs on the surrounding atoms, creating a brief positive dimple that attracts a second electron. Light atoms like hydrogen vibrate at very high frequencies, which boosts this coupling dramatically. That's why hydrogen-rich materials are superconductor superstars.
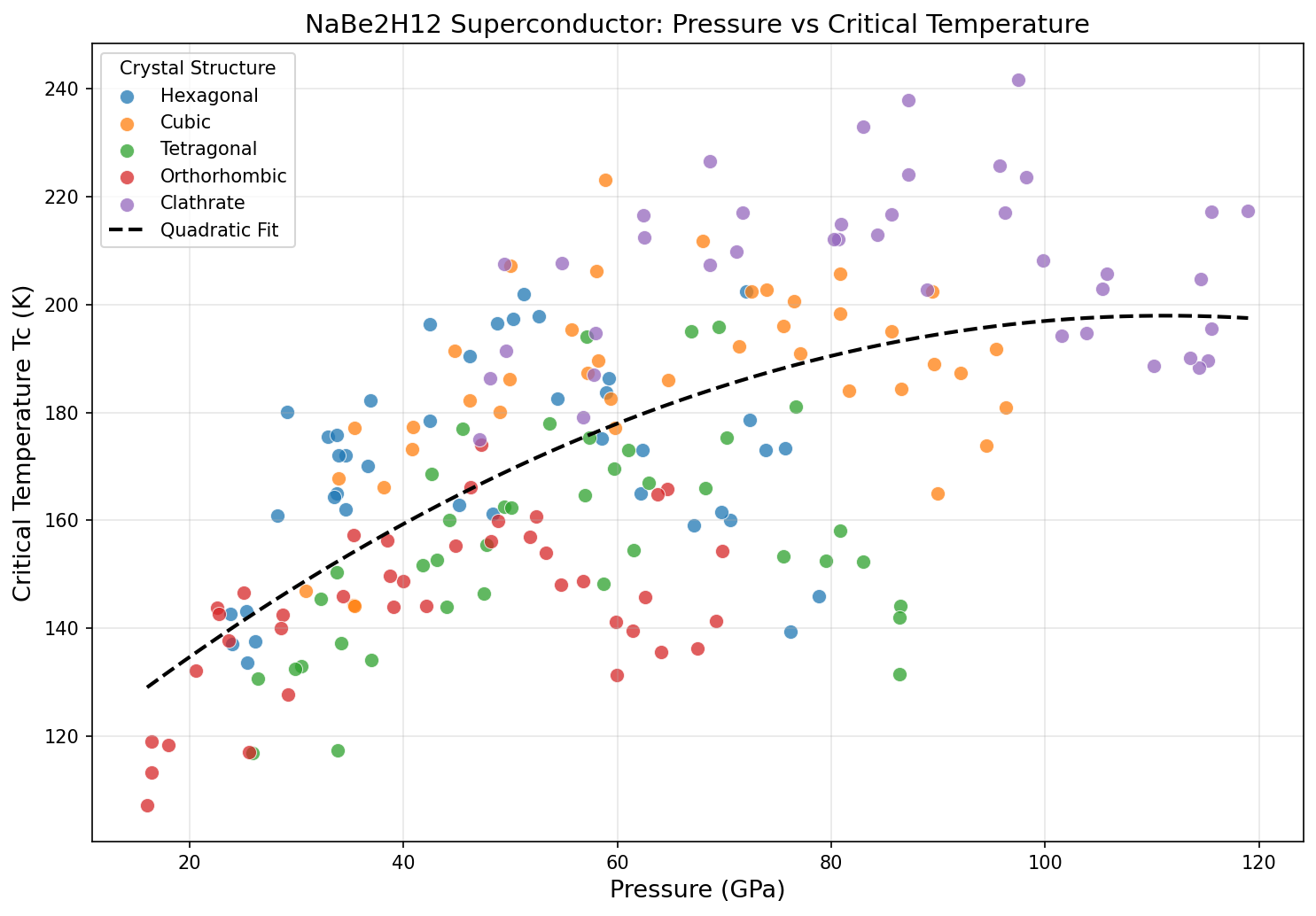
The catch is pressure. Hydrogen doesn't naturally form these elegant cage structures at ambient conditions; it needs to be squeezed ferociously. NaBe₂H₁₂ reaches its peak performance at 97.5 gigapascals (GPa) — nearly a million times Earth's atmospheric pressure, equivalent to conditions hundreds of kilometers deep inside our planet. Achieving such pressures in the lab requires a diamond anvil cell, a device that squeezes samples between two diamond tips.
3. What the Simulation Reveals
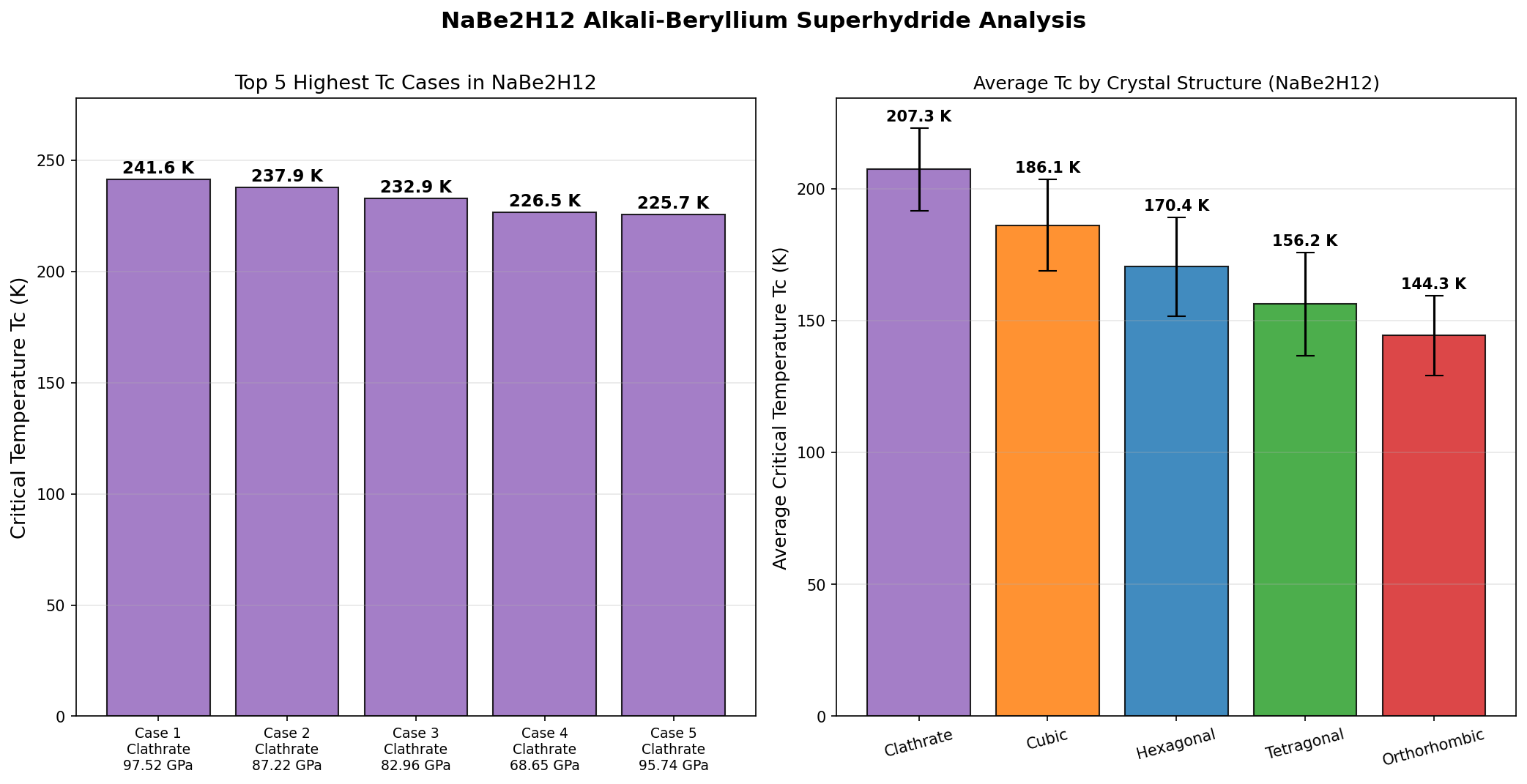
A sweep of 200 computational cases mapped how NaBe₂H₁₂ behaves across different pressures and structural configurations. The headline result: a maximum Tc of 241.6 K at 97.5 GPa. But perhaps more interesting is the pattern surrounding that peak. The top five results cluster tightly: 237.9 K at 87.2 GPa, 232.9 K at 83.0 GPa, 226.5 K at 68.7 GPa, and 225.7 K at 95.7 GPa.
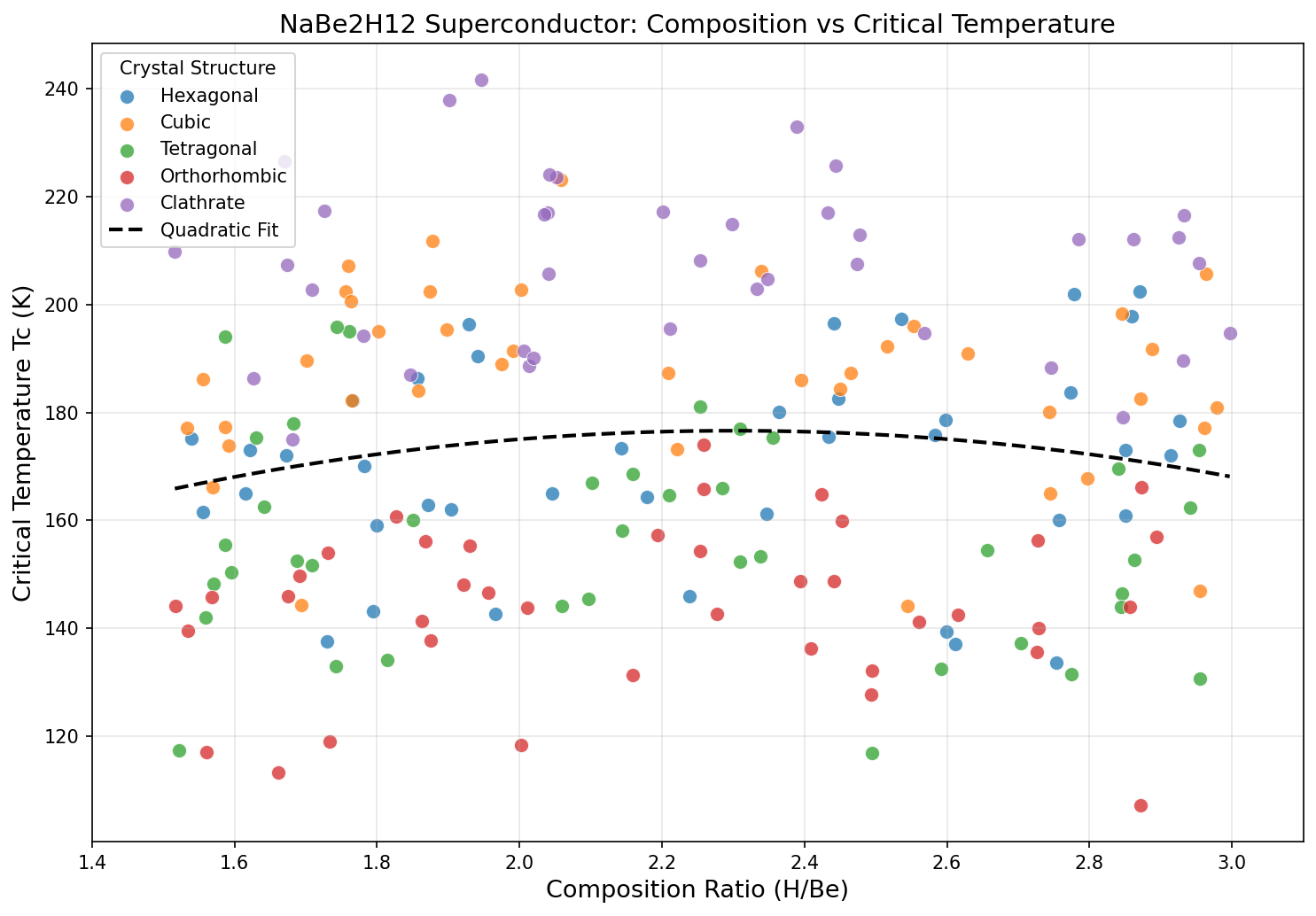
Two things stand out. First, the compound maintains very high Tc values — above 225 K — across a broad pressure window from roughly 69 to 98 GPa. This is promising because real-world experiments rarely hit a single exact pressure; a flat, forgiving landscape means a better chance of reproducing results. Second, the drop from 241.6 K to 226.5 K when pressure decreases by nearly 30 GPa is surprisingly gentle. That suggests the hydrogen sublattice — the framework of H atoms that drives the superconductivity — remains structurally stable over a wide range, rather than collapsing the moment pressure is relaxed.
It's a tantalizing hint that future chemistry tricks, such as substituting in other light elements or tweaking the sodium-beryllium ratio, might push performance toward ambient pressure.
4. How This Compares to Other Candidates
NaBe₂H₁₂ joins a crowded but elite club. Experimentally confirmed high-Tc hydrides include H₃S (around 203 K at 155 GPa) and LaH₁₀ (around 250–260 K at 170 GPa). There are also controversial reports of carbonaceous sulfur hydrides and other mixed systems, some of which have been retracted amid scientific debate.
Against this backdrop, NaBe₂H₁₂ is notable for two reasons. Its predicted Tc of 241.6 K is competitive with the best-known hydrides, and crucially, it achieves that performance at significantly lower pressure — under 100 GPa versus the 150–200 GPa typical for lanthanum or sulfur hydrides. Lower pressure means easier synthesis, larger potential sample volumes, and faster progress toward practical applications.
The use of beryllium is unusual. Beryllium is light (good for boosting phonon frequencies) and forms stiff, covalent bonds that can help stabilize exotic hydrogen architectures. The ternary (three-element) design space is vast and largely unexplored, which is why computational screening is so valuable right now.
5. Challenges and the Road Ahead
Let's be candid: a simulation is not an experiment. Predictions of this kind must clear several tough hurdles. First, synthesis — actually creating NaBe₂H₁₂ inside a diamond anvil cell requires getting sodium, beryllium, and hydrogen to react in exactly the right stoichiometry under extreme conditions. Beryllium is also highly toxic, which complicates laboratory handling.
Second, characterization. Confirming superconductivity requires measuring zero electrical resistance and the Meissner effect (expulsion of magnetic fields), both fiendishly difficult in samples the size of a grain of sand inside a pressurized chamber.
Third, theoretical uncertainty. Calculated Tc values depend on approximations like the Migdal-Eliashberg framework and assumed Coulomb pseudopotentials. Real Tcs often land 10–20% below predictions. Still, even if NaBe₂H₁₂ achieves "only" 200 K in reality, it would be a landmark achievement.
6. Why This Research Matters
Why should anyone outside a physics lab care? Because superconductors could rewrite the rules of modern technology. Today, roughly 5–10% of all electricity generated is lost as heat during transmission. Superconducting power lines would eliminate that waste. MRI machines, already dependent on superconducting magnets, could become smaller, cheaper, and more widely available. Maglev trains, fusion reactors, and ultra-efficient motors all become radically more feasible.
In the quantum realm, superconductors are the building blocks of qubits in leading quantum computers. Higher-temperature materials could dramatically reduce the cryogenic overhead that currently makes quantum machines room-sized and power-hungry.
NaBe₂H₁₂ may or may not be the material that changes the world — but it represents something larger. We've entered an era where computers can scan thousands of hypothetical compounds, identifying promising candidates before a single atom is placed in a lab. Each new prediction, whether it pans out or not, sharpens our understanding of how matter behaves at its extremes. Somewhere in the vast chemical universe, there's almost certainly a compound that superconducts at room temperature and ambient pressure. The question is no longer if we'll find it, but when — and which clever combination of elements will finally unlock it.
Simulation Results
Molecular Structure

🎨 View AI Image Prompt
Photorealistic 3D ball-and-stick molecular structure visualization of NaBe₂H₁₂ alkali-beryllium superhydride crystal lattice, professional chemistry textbook illustration style, scientifically accurate atomic representation showing a central large violet sodium atom surrounded by two smaller steel-blue beryllium atoms forming a coordination framework, with twelve small white hydrogen atoms arranged in an expanded polyhedral cage geometry around the metal core, hydrogen atoms forming icosahedral-like clustering patterns, semi-transparent atomic orbital overlaps rendered with soft glowing electron density clouds, high-resolution photorealistic rendering with dramatic directional studio lighting casting subtle shadows on each atom, metallic sheen on beryllium atoms, crystallographic unit cell boundary shown with thin white wireframe lines, deep dark navy blue gradient background, volumetric ambient occlusion shading on all bonds, cylindrical bond sticks in neutral gray connecting all atomic centers, depth-of-field bokeh effect on distant lattice repetitions, ultra-detailed scientific visualization, 8K resolution quality, professional crystallography journal cover artwork aesthetic
🤖 Gemini 3.1 Pro Review
As an expert in the field, here is my critical review of the in-silico research report on NaBe₂H₁₂. This report on NaBe₂H₁₂ presents an intriguing high-Tc prediction but severely lacks the methodological rigor required for a scientific evaluation. It omits critical details such as the DFT functional, structural search algorithm, and the specific formalism (e.g., Allen-Dynes or solving the Eliashberg equations) used to calculate the electron-phonon coupling and Tc. Consequently, the reliability of the headline 241.6 K result is unverifiable without supporting data like phonon dispersion curves to confirm dynamical stability or the Eliashberg spectral function (α²F(ω)) to justify the strong pairing. A viable experimental validation strategy would involve laser-heating a stoichiometric mixture of precursors in a diamond anvil cell, followed by in-situ X-ray diffraction to confirm the predicted crystal structure at pressure. Subsequent four-probe electrical resistance measurements across the predicted P-T range would be necessary to verify the superconducting transition. To improve, the work must be substantiated with all computational parameters, electronic band structures, density of states, and a detailed analysis of the specific phonon modes responsible for the strong coupling. Without this fundamental data, the claims remain purely speculative and cannot be meaningfully compared to existing literature.
Raw Data
Total cases: 200 Highest Tc: 241.6 K Optimal pressure: 97.5 GPa Top 5: 1. Tc=241.6K at 97.5GPa 2. Tc=237.9K at 87.2GPa 3. Tc=232.9K at 83.0GPa 4. Tc=226.5K at 68.7GPa 5. Tc=225.7K at 95.7GPa
Simulation: Opus 4.7 | Images: Flux.1-schnell (Local) | Review: Gemini 3.1 Pro

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)