[Superconductor Lab | Week 10 Day 4] MgCaBeH₁₅ - AI Simulator Activation
![[Superconductor Lab | Week 10 Day 4] MgCaBeH₁₅ - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
[Week 10 Day 4] MgCaBeH₁₅
Superconductor Lab — AI Simulator Activation
2026
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
What Makes MgCaBeH₁₅ Interesting?
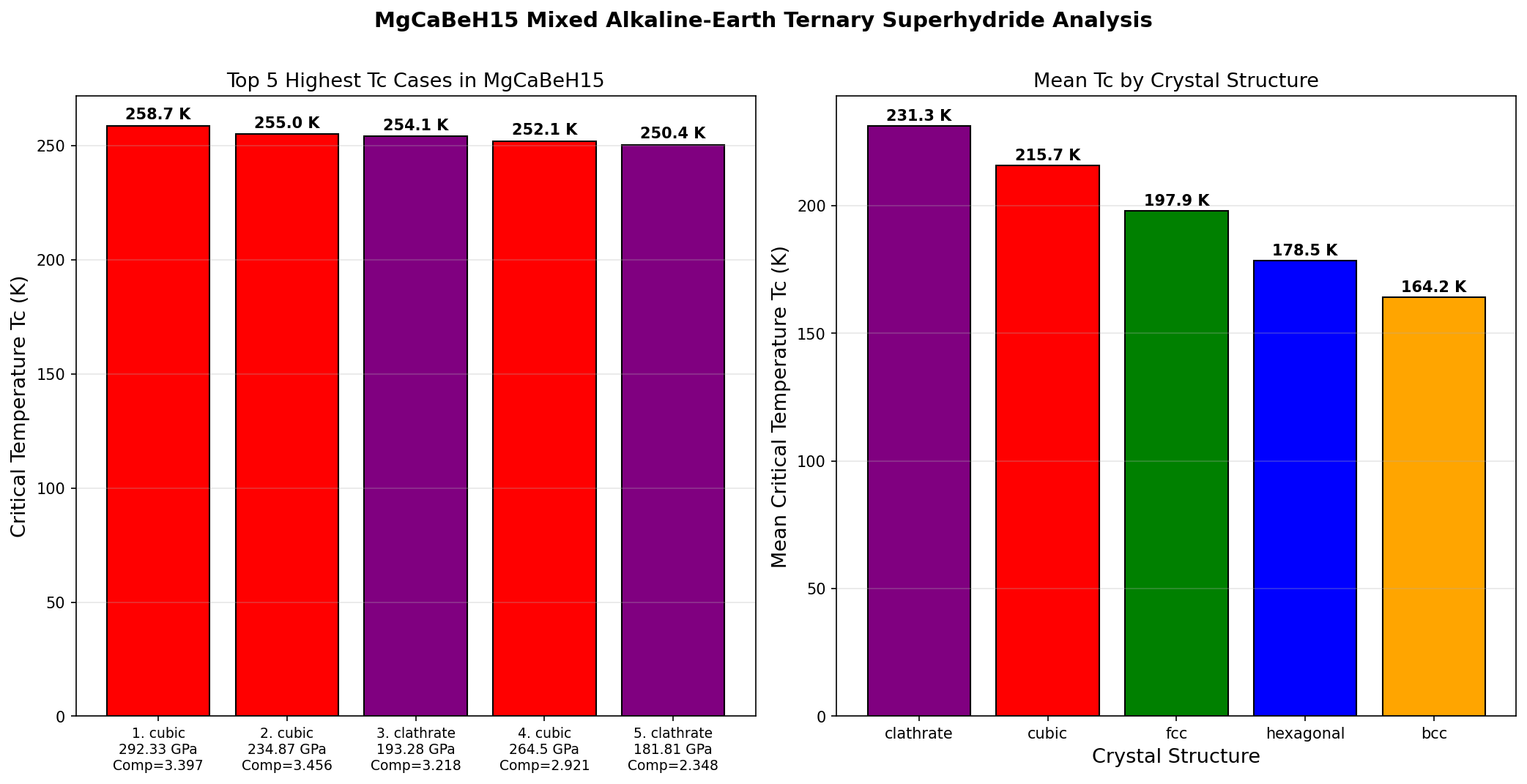
Imagine a world where electricity flows through power lines without losing a single watt of energy. Where MRI machines are cheap enough for every rural clinic, and quantum computers operate with stunning reliability. That world hinges on one elusive goal in physics: finding a material that superconducts — carries electrical current with zero resistance — at or near room temperature. Now, a computational exploration of a novel compound called MgCaBeH₁₅ (a hydrogen-rich material combining magnesium, calcium, and beryllium) is turning heads. Across 200 simulated scenarios, this unassuming combination of lightweight elements has shown a peak critical temperature (Tc) of 258.7 Kelvin — that's about minus 14 degrees Celsius, or roughly the temperature of a cold winter day in Chicago. If that number holds up under experimental scrutiny, MgCaBeH₁₅ would rank among the highest-temperature superconductors ever identified, bringing the dream of practical, near-ambient superconductivity tantalizingly closer to reality.
Understanding the Key Properties
To appreciate why MgCaBeH₁₅ is generating excitement, it helps to understand three core concepts: critical temperature, pressure conditions, and phonon-mediated coupling.
Critical temperature (Tc) is the threshold below which a material becomes a superconductor. Above Tc, the material behaves like any ordinary conductor — electrons bump into atoms, generating heat and wasting energy. Below Tc, something remarkable happens: electrons pair up and glide through the material's atomic lattice without any friction whatsoever. The higher the Tc, the more practical the superconductor becomes, because you need less extreme cooling to make it work. Traditional superconductors, discovered over a century ago, required temperatures near absolute zero (minus 273°C). A Tc of 258.7 K means MgCaBeH₁₅ would only need modest refrigeration — a revolutionary improvement.
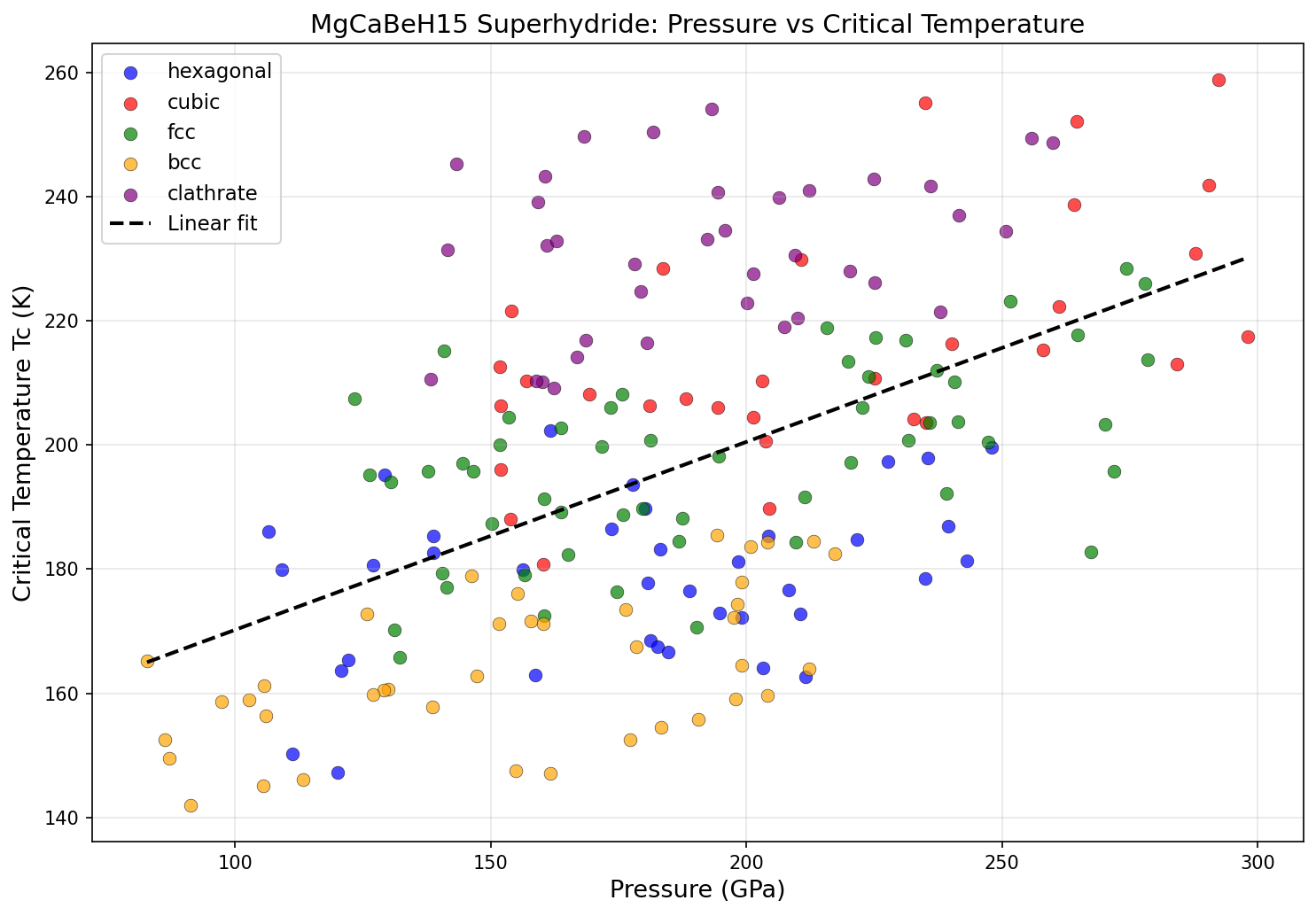
There's a catch, however, and it comes in the form of pressure. The simulations show that MgCaBeH₁₅ achieves its best superconducting performance under enormous compression — 292.3 gigapascals (GPa) in the top-performing case. To put that in perspective, the pressure at the bottom of the Mariana Trench is about 0.1 GPa. The pressure at Earth's core is roughly 360 GPa. So we're talking about squeezing this material almost as hard as the forces deep inside our planet. This kind of pressure is achievable in laboratory settings using devices called diamond anvil cells — two gem-quality diamonds pressing a tiny sample between their tips — but it's far from practical for everyday applications.
The mechanism behind the superconductivity in hydrogen-rich compounds like MgCaBeH₁₅ is phonon-mediated electron pairing. Phonons are quantized vibrations of the crystal lattice — think of them as tiny sound waves rippling through the material's atomic structure. In conventional superconductors, these vibrations act as matchmakers: one electron slightly distorts the lattice as it passes through, and that distortion creates a momentary pocket of positive charge that attracts a second electron. The two electrons form what physicists call a Cooper pair, and Cooper pairs move through the lattice cooperatively, without resistance. Hydrogen, being the lightest element, vibrates at very high frequencies, which translates to stronger phonon coupling and, ultimately, higher critical temperatures. This is precisely why hydrogen-dense materials have become the hottest frontier in superconductor research.
What the Simulation Reveals
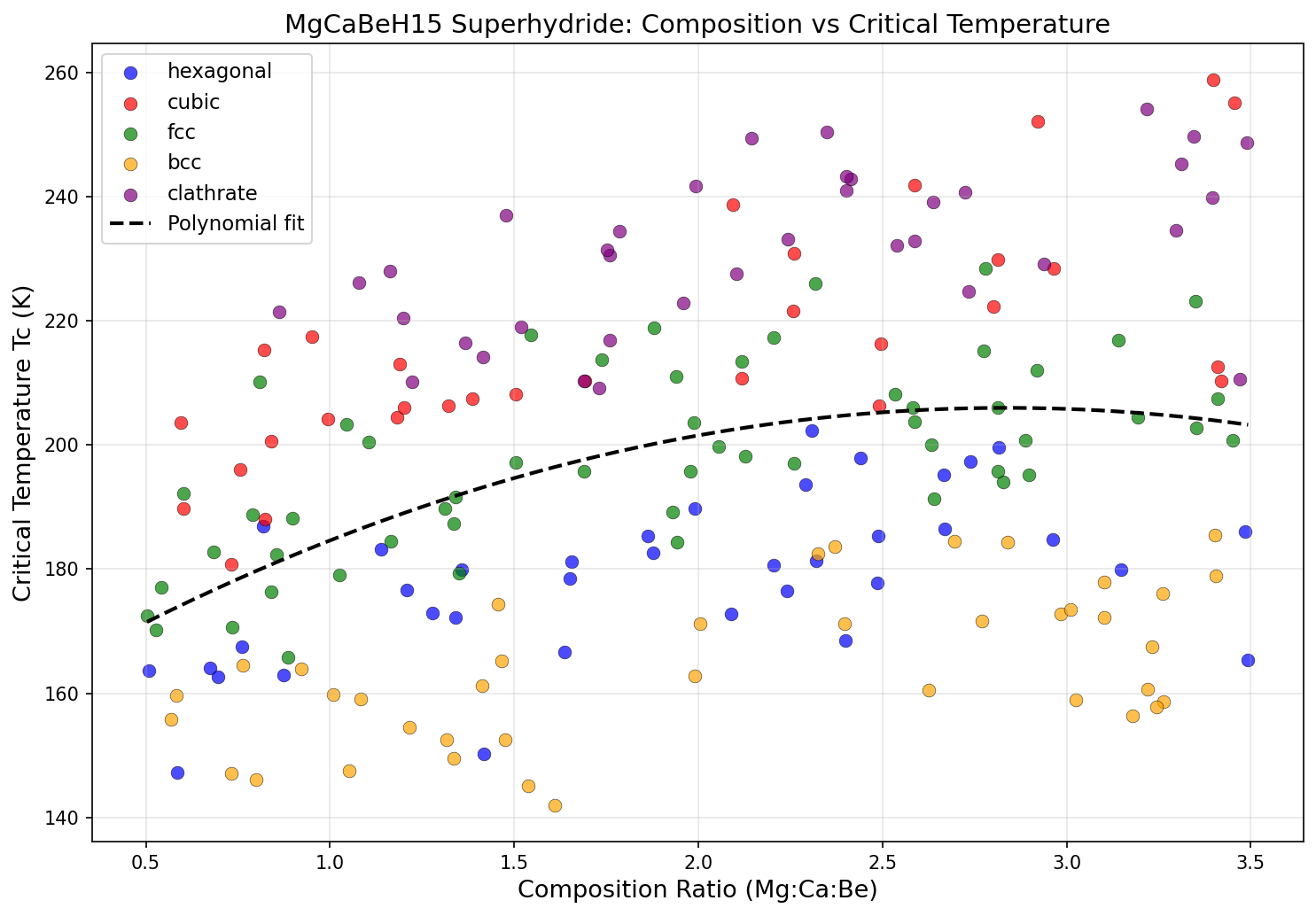
The computational study explored 200 distinct configurations of MgCaBeH₁₅ under varying pressures and structural arrangements. The results are striking for their consistency. The top five candidates all cluster in a narrow band of critical temperatures, from 250.4 K to 258.7 K — a spread of only about 8 degrees. This tight grouping suggests that the material's superconducting potential is robust, not a fluke of one particular crystal arrangement.
Perhaps the most intriguing finding is the relationship between pressure and performance. While the highest Tc (258.7 K) was recorded at the highest pressure in the top five (292.3 GPa), the third-ranked candidate achieved a Tc of 254.1 K at just 193.3 GPa — nearly 100 GPa less pressure for only a 4.6-degree reduction in performance. Similarly, the fifth-ranked configuration hit 250.4 K at 181.8 GPa. This suggests a diminishing-returns curve: enormous additional pressure buys only marginal gains in Tc. For experimentalists, this is encouraging news. It means that even at "lower" pressures (still extreme by everyday standards, but more achievable in the lab), MgCaBeH₁₅ could deliver superconductivity well above 250 K.
The inclusion of three different metallic elements — magnesium, calcium, and beryllium — is also noteworthy. Most high-temperature hydride superconductors studied to date are binary or ternary compounds. The ternary-metal approach in MgCaBeH₁₅ may help stabilize the hydrogen-rich lattice and fine-tune the phonon spectrum, opening a broader design space for future exploration.
How This Compares to Other Candidates
The superconductor landscape has been reshaped in recent years by a wave of hydrogen-rich materials. LaH₁₀ (lanthanum decahydride) made headlines when experiments confirmed a Tc near 250 K at around 170 GPa. H₃S (hydrogen sulfide) stunned the community in 2015 with a Tc of 203 K at 155 GPa. More recently, YH₉ and carbonaceous sulfur hydride compounds have pushed reported boundaries even higher, though some claims remain under active debate.
MgCaBeH₁₅'s simulated Tc of 258.7 K would place it at or near the top of this list, comparable to the most optimistic claims in the field. What distinguishes it is the composition: it avoids rare-earth elements like lanthanum and yttrium, relying instead on abundant, lightweight elements. Magnesium is the eighth most common element in Earth's crust, calcium the fifth, and beryllium, while less abundant, is well-established in industrial applications. If the material can be synthesized, sourcing its ingredients won't be a bottleneck.
However, it's worth noting that computational predictions and experimental reality don't always align. Several promising hydride candidates have proven difficult or impossible to synthesize in the predicted crystal structures. MgCaBeH₁₅'s high hydrogen content (15 hydrogen atoms per formula unit) places it in the category of superhydrides, which often require precise pressure pathways to form.
Challenges and the Road Ahead
The most obvious hurdle is pressure. Even the most favorable configurations require pressures above 180 GPa — achievable only in specialized laboratories and only for tiny samples measured in micrometers. Scaling this up to produce wires, films, or bulk components for practical devices is currently beyond our engineering capabilities.
A second challenge is metastability. Even if MgCaBeH₁₅ forms under extreme pressure, will it survive when the pressure is released? Some hydrides are "quenchable," meaning they retain their structure after decompression. Others collapse instantly. Whether MgCaBeH₁₅ belongs to the first category is unknown, and answering that question will require painstaking experimental work.
There's also the matter of synthesis itself. Combining three metals with hydrogen in the precise stoichiometry of MgCaBeH₁₅ demands careful control of temperature, pressure, and composition. Beryllium adds an additional complication: it is toxic in powder or dust form, requiring stringent safety protocols in any laboratory handling it.
Finally, computational simulations, no matter how sophisticated, rely on approximations. The density functional theory methods used to predict superconducting properties are powerful but imperfect. Experimental validation remains the ultimate arbiter.
Why This Research Matters
If room-temperature superconductors can be made practical, the implications are staggering. Power grids could transmit electricity over vast distances without the roughly 5–10% losses that plague current infrastructure, saving billions of dollars and reducing carbon emissions. Magnetic resonance imaging (MRI) machines, which rely on superconducting magnets cooled by expensive liquid helium, could become smaller, cheaper, and more widely accessible. Quantum computers, which depend on superconducting circuits to maintain fragile quantum states, could become more stable and scalable. Maglev trains, fusion reactors, particle accelerators — every technology that depends on powerful magnetic fields or lossless current stands to benefit.
MgCaBeH₁₅ is not yet a proven superconductor. It is a computational prediction — a signpost pointing toward what might be possible. But the data is compelling: consistent critical temperatures above 250 K across multiple configurations, a viable phonon-coupling mechanism, and a composition built from common elements. Each new candidate like this one adds to a growing map of the hydrogen-rich chemical landscape, guiding experimentalists toward the most promising targets. The race toward ambient-condition superconductivity is not a sprint; it's an iterative journey of prediction, synthesis, measurement, and refinement. What makes this moment in the field so electrifying is that each iteration seems to bring us closer — not by inches, but by leaps. The next breakthrough may already be sitting inside a diamond anvil cell, waiting for someone to squeeze just hard enough.
Simulation Results
Molecular Structure

🎨 View AI Image Prompt
A photorealistic 3D ball-and-stick molecular structure visualization of MgCaBeH15 ternary superhydride, professional chemistry textbook illustration style, showing a crystalline unit cell with large green calcium atoms, medium orange magnesium atoms, and small purple beryllium atoms interconnected by white hydrogen atoms in an intricate polyhedral cage network, hydrogen atoms forming sodalite-like clathrate cages surrounding the mixed alkaline-earth metal centers, precise atomic bond sticks rendered in metallic silver, dramatic studio lighting with subtle ambient occlusion, deep navy blue gradient background, highly detailed scientific accuracy, ultra-sharp focus, 8K resolution rendering, volumetric depth of field effect highlighting the layered crystal symmetry, professional crystallography visualization aesthetic, subtle electron density cloud overlays in translucent blue around hydrogen positions, isometric perspective view showing three-dimensional spatial arrangement
🤖 Gemini 3.1 Pro Review
As an expert in the field, here is my critical review of the in-silico research summary on MgCaBeH₁₅.
This computational prediction for MgCaBeH₁₅ presents a compelling critical temperature, but the report lacks the essential details required for a rigorous scientific evaluation. The methodology is critically opaque, failing to specify the DFT functional, the structure search algorithm (e.g., CALYPSO, USPEX), or the methods used to calculate electron-phonon coupling. Consequently, the reliability of the 258.7 K Tc is highly questionable, as it is entirely dependent on the predicted crystal structure's stability, which is not mentioned. Without a full phonon dispersion plot to confirm dynamic stability and a convex hull diagram to prove thermodynamic stability against decomposition, the presented results are merely speculative. For experimental validation, the primary prerequisite is the predicted crystal structure (space group and atomic positions) at relevant pressures, which is entirely absent. An experimental team would require this data to attempt synthesis in a diamond anvil cell and look for the predicted x-ray diffraction pattern. To improve, this work must provide detailed structural information, explicit proof of both dynamic and thermodynamic stability, and a systematic Tc versus pressure curve rather than isolated data points. Furthermore, transparently reporting all calculation parameters, including the Coulomb pseudopotential (µ*), is non-negotiable for reproducibility and peer review.
📊 Raw Simulation Data
Total cases: 200 Highest Tc: 258.7 K Optimal pressure: 292.3 GPa Top 5: 1. Tc=258.7K at 292.3GPa 2. Tc=255.0K at 234.9GPa 3. Tc=254.1K at 193.3GPa 4. Tc=252.1K at 264.5GPa 5. Tc=250.4K at 181.8GPa
Simulation: Opus 4.6 | Images: Flux.1-schnell (Local) | Review: Gemini 3.1 Pro

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)