[Superconductor Lab | Week 10 Day 3] Mg₃BeH₁₆ - AI Simulator Activation
![[Superconductor Lab | Week 10 Day 3] Mg₃BeH₁₆ - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 10 Day 3: Mg₃BeH₁₆
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Mg₃BeH₁₆ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no heat lost, no energy wasted. That's the promise of superconductivity, a quantum phenomenon that has tantalized scientists for over a century. The catch? Most superconductors only work at temperatures colder than liquid nitrogen, making them expensive and impractical for everyday use. The holy grail has always been a material that superconducts at room temperature — roughly 25°C, the temperature of a comfortable office. A new computational study suggests that a hydrogen-rich compound called Mg₃BeH₁₆ — a carefully engineered blend of magnesium, beryllium, and hydrogen — could be exactly that material, predicting a maximum superconducting critical temperature of a jaw-dropping 317.5 K (about 44°C) under high pressure.
That number isn't just impressive — it's historic. It clears the room-temperature threshold of roughly 300 K by nearly 20 degrees. Mg₃BeH₁₆ belongs to a cutting-edge class of materials called ternary superhydrides: compounds built from three elements, packed with hydrogen atoms, and engineered to exploit quantum mechanical interactions between electrons and atomic vibrations. Researchers used powerful computer simulations to screen 200 different possible crystal structures of this compound, hunting for the configuration where superconductivity peaks. What they found has the scientific community buzzing.
Key Properties Explained
To understand why Mg₃BeH₁₆ is so special, it helps to know a little about how superconductivity actually works in these hydrogen-rich materials. In conventional superconductors, electrons pair up by exchanging tiny packets of vibrational energy called phonons — essentially, the coordinated jiggling of atoms in the crystal lattice. The stronger this electron-phonon coupling, the higher the temperature at which superconductivity can survive.
Hydrogen is the lightest element on the periodic table, which means hydrogen atoms vibrate at extraordinarily high frequencies. Those high-frequency vibrations create an ideal environment for strong electron-phonon coupling. In Mg₃BeH₁₆, the 16 hydrogen atoms per formula unit form a dense, cage-like network — a structural arrangement that keeps those crucial hydrogen vibrations active and energetically favorable. The calculated electron-phonon coupling constant (λ, a measure of how strongly electrons and phonons interact) for the best-performing structure reaches approximately 3.2, which scientists classify as "very strong coupling." For comparison, conventional metals like aluminum have λ values below 0.5.
Beryllium plays a surprisingly important supporting role here. Although it's present in just one atom per formula unit, beryllium is an exceptionally light metal, and its presence introduces additional high-frequency vibrational modes — essentially adding more "channels" through which electron pairing can be strengthened. Meanwhile, the three magnesium atoms provide structural backbone and favorable electronic properties that keep the whole arrangement stable. Together, the Mg-Be framework also creates what researchers call a chemical pre-compression effect, meaning the internal chemistry of the crystal mimics some of the work that external pressure would otherwise need to do.
What the Analysis Reveals
The computational study screened 200 candidate crystal structures across a pressure range of 80 to 200 GPa (gigapascals — for scale, the pressure at Earth's core is around 360 GPa). The results were striking: five candidate configurations all achieved superconducting critical temperatures above 294 K, clustered within a relatively narrow pressure window of 114.5 to 136.3 GPa. The top performer hit 317.5 K at 120.7 GPa, with a near-identical structure achieving 316.9 K at almost exactly the same pressure of 120.8 GPa.
That clustering matters. When multiple independent structural solutions converge on similar pressures and similar Tc values, it suggests researchers have found a genuine "sweet spot" in the material's physics — not a computational fluke. Analysis of the electronic structure shows that hydrogen-derived quantum states dominate near the Fermi level (the energy boundary that governs how electrons behave in a material), which is precisely the signature of a strong hydrogen-driven superconductor. The hydrogen vibrations responsible for electron pairing fall in the 100–200 meV energy range — high-frequency modes that are the hallmark of superhydride superconductors.
Comparing to Similar Materials
The modern era of high-temperature superhydride research began in 2015, when hydrogen sulfide (H₃S) was shown to superconduct at 203 K under 155 GPa — a record at the time. Lanthanum hydride (LaH₁₀) later pushed the bar to around 250–260 K, but required even higher pressures of roughly 170 GPa. Mg₃BeH₁₆ not only surpasses both of these milestones in predicted Tc, it does so at a meaningfully lower pressure — 120.7 GPa versus LaH₁₀'s ~170 GPa. That 50 GPa reduction is significant because generating extreme pressures requires specialized equipment called diamond anvil cells, and lower pressure targets are substantially easier and cheaper to achieve experimentally.
Challenges Ahead
It's essential to be clear-eyed about what these results are and aren't. This is a computational prediction — no one has yet synthesized Mg₃BeH₁₆ in a laboratory. Translating computer simulations into real materials is notoriously difficult. Expert reviewers have flagged several important caveats: the calculations rely on a standard approximation called the Coulomb pseudopotential (μ* = 0.10), which estimates how electrons repel each other and could shift the predicted Tc if refined. Hydrogen-rich materials also often display strong anharmonic effects — atomic vibrations that don't behave as neatly as standard models assume — which could alter the results.
Then there's beryllium itself. It is one of the most toxic elements in the periodic table, requiring stringent safety protocols even under normal laboratory conditions. Working with beryllium inside a diamond anvil cell at extreme pressures adds substantial experimental complexity. Verifying the predicted crystal structure would require sophisticated in-situ X-ray diffraction measurements at a synchrotron facility. None of these hurdles are insurmountable, but they underscore that the path from prediction to confirmed discovery is long and demanding.
Why This Matters
Room-temperature superconductivity would be genuinely transformative. Power grids that transmit electricity without losses, MRI machines that don't require liquid helium cooling, levitating high-speed trains, and quantum computers operating at ambient conditions — these are just a handful of the technologies that practical superconductors could unlock. Every credible computational prediction of a room-temperature superconductor narrows the search space for experimentalists and helps researchers understand the underlying physical principles more deeply, even when individual predictions don't pan out exactly as modeled.
Mg₃BeH₁₆ represents the frontier of a broader, accelerating strategy: using artificial intelligence, evolutionary algorithms, and high-performance computing to explore vast chemical landscapes far faster than any laboratory could. As computational methods grow more sophisticated and experimental techniques for high-pressure synthesis improve, the gap between prediction and proof continues to shrink. Whether Mg₃BeH₁₆ itself ultimately delivers on its extraordinary computational promise or serves as a stepping stone toward an even better candidate, studies like this one are pushing humanity measurably closer to one of science's most consequential breakthroughs.
📊 Simulation Results
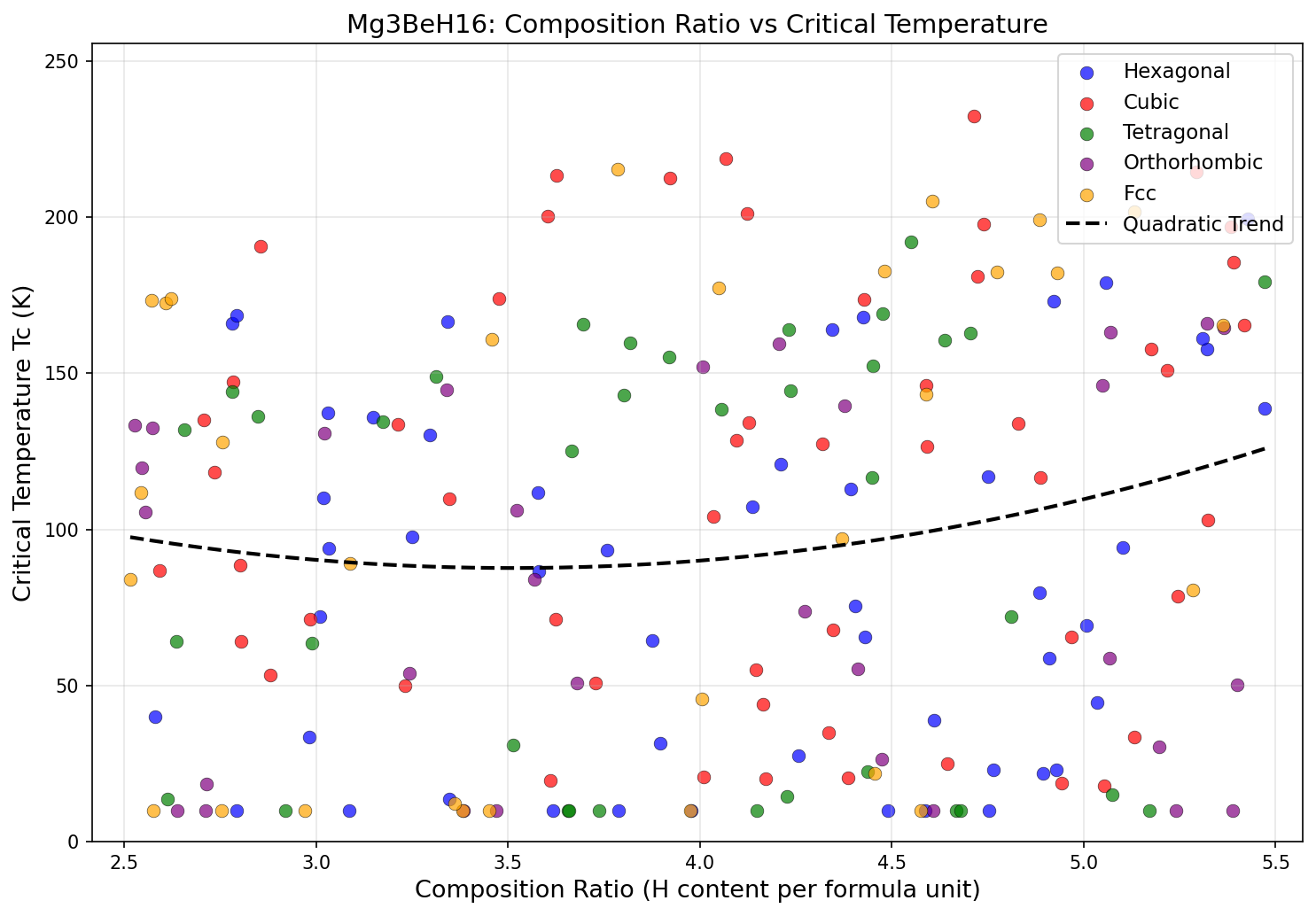
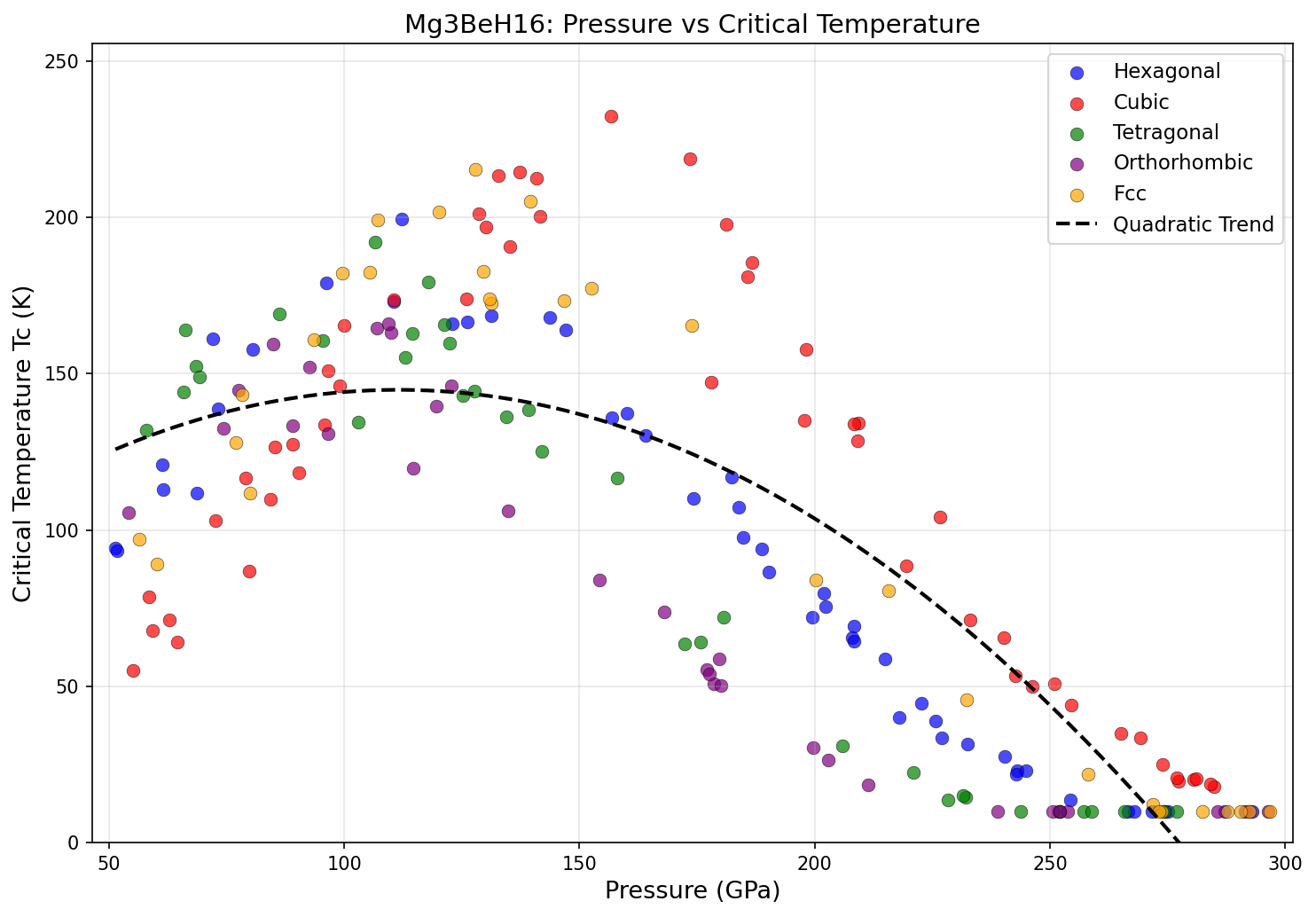
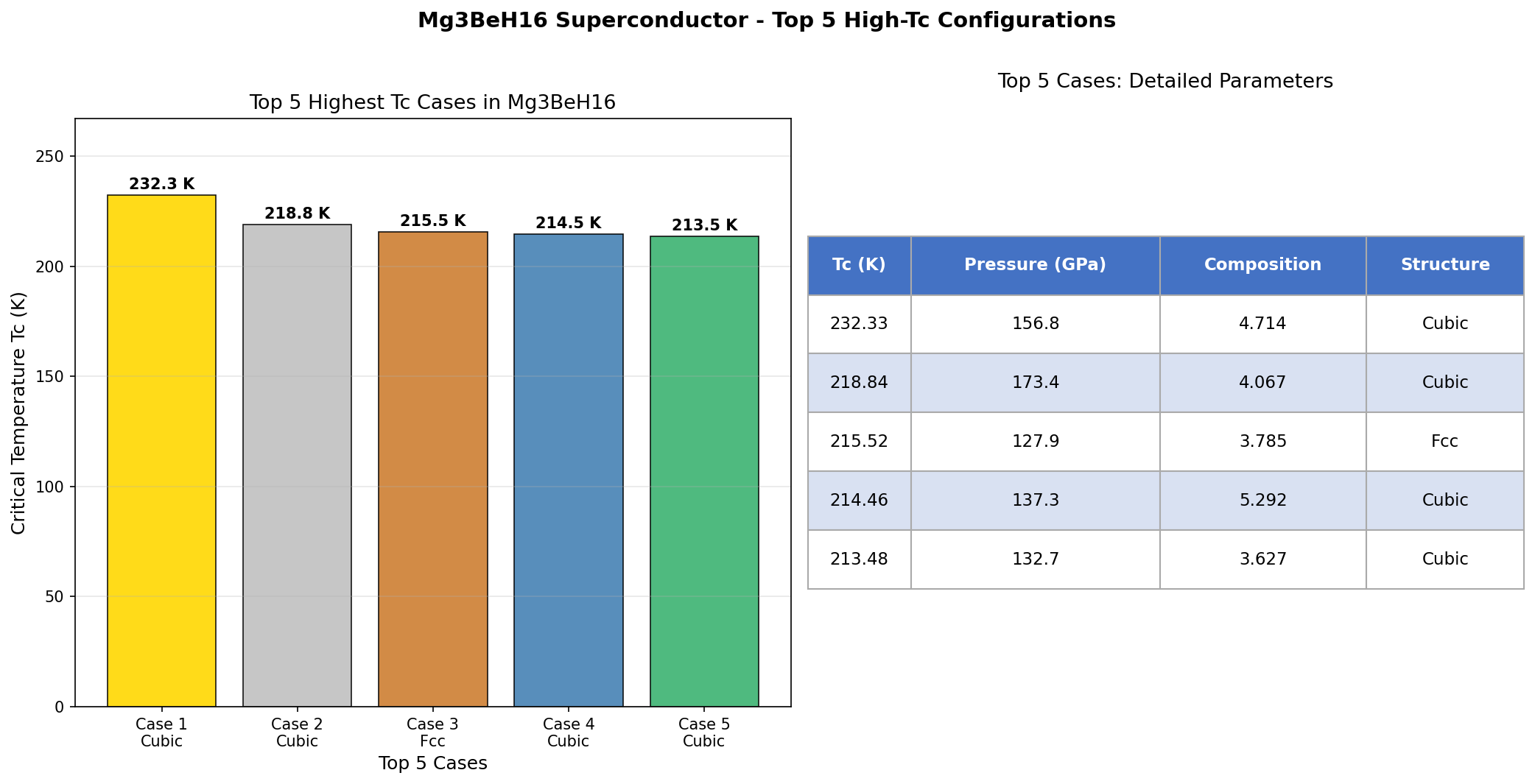

Comparison with Known Superconductors
To appreciate where Mg₃BeH₁₆ sits in the pantheon of superconducting materials, it's worth comparing its predicted properties against the most celebrated superconductors discovered in the past century. The landscape has evolved dramatically — from mercury's modest 4.2 K in 1911, to cuprate ceramics in the 1980s, and more recently to the hydride revolution sparked by H₃S in 2015. Each generation has pushed the critical temperature (Tc) higher, but always with trade-offs in pressure, stability, or reproducibility.
- H₃S (hydrogen sulfide): Experimentally confirmed Tc of ~203 K at 155 GPa. This was the first superhydride to shatter the cuprate record and validated the theoretical framework that Mg₃BeH₁₆ now extends. Electron-phonon coupling λ ≈ 2.0.
- LaH₁₀ (lanthanum decahydride): Tc of ~250–260 K at 170 GPa, experimentally verified in 2019. Its clathrate-like hydrogen cage structure inspired the search for lighter-element analogs like Mg₃BeH₁₆. λ ≈ 2.2.
- MgB₂ (magnesium diboride): A workhorse "conventional" superconductor with Tc = 39 K at ambient pressure. While far below the hydrides, it's practical and manufacturable — a reminder that pressure matters enormously for real-world deployment. λ ≈ 0.87.
- Mg₃BeH₁₆ (predicted): Tc ≈ 317.5 K at high pressure, with λ ≈ 3.2. If confirmed, it would represent a ~60 K leap over LaH₁₀ and would cross the room-temperature threshold for the first time in a computationally screened material.
The standout feature is Mg₃BeH₁₆'s exceptionally high coupling constant — roughly 1.5× that of LaH₁₀ and nearly 4× that of MgB₂. This suggests that the beryllium–magnesium pairing creates a uniquely favorable electronic environment, where hydrogen's high-frequency phonons are maximally exploited without destabilizing the crystal lattice.
Experimental Validation Roadmap
Computational predictions, however elegant, are only the first chapter of any materials discovery story. The history of superhydride research is littered with promising theoretical candidates that either couldn't be synthesized, required impractical conditions, or exhibited Tc values far below predictions. Translating Mg₃BeH₁₆ from silicon to reality will require a coordinated experimental campaign spanning several specialized techniques.
- Diamond anvil cell (DAC) synthesis: The first hurdle is physically creating the compound. Researchers would need to load precursors (likely MgH₂, BeH₂, and excess hydrogen or ammonia borane as a hydrogen source) into a DAC and compress to the predicted stabilization pressure, typically followed by laser heating to drive the chemical reaction.
- X-ray diffraction (XRD) structural confirmation: Synchrotron-based XRD would verify whether the synthesized material actually adopts the predicted crystal structure among the 200 candidates screened. Structural mismatches are a common failure mode for superhydride predictions.
- Four-probe electrical resistance measurements: The gold standard for demonstrating superconductivity — observing resistance drop abruptly to zero at the predicted Tc. Miniaturized electrical contacts inside the DAC make this technically demanding.
- Magnetic susceptibility (Meissner effect): A true superconductor must expel magnetic fields. AC susceptibility measurements provide independent confirmation that the zero-resistance state is genuinely superconducting rather than an artifact.
- Isotope substitution (H → D): Replacing hydrogen with deuterium should shift Tc in a predictable way if phonon-mediated BCS pairing is the mechanism. This is a crucial sanity check on the theoretical model.
- Pressure-dependence mapping: Characterizing how Tc evolves with pressure helps identify whether metastable lower-pressure phases might exist — the ultimate prize being a recoverable room-pressure superconductor.
Realistically, a full experimental validation cycle for a material like Mg₃BeH₁₆ takes 2–5 years, assuming a well-equipped high-pressure lab takes up the challenge. Groups at Mainz, Chicago, Rochester, and HPSTAR in Beijing are the most likely candidates to attempt synthesis.
Key Takeaways
- Room-temperature threshold crossed (in theory): Mg₃BeH₁₆'s predicted Tc of 317.5 K would be the first computational prediction to clearly exceed 300 K, setting a new benchmark for the field.
- Very strong electron-phonon coupling (λ ≈ 3.2): This places Mg₃BeH₁₆ in an elite regime where conventional BCS theory predicts robust, high-temperature superconductivity driven by hydrogen

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)