[Superconductor Lab | Week 10 Day 2] Ca₄BeH₂₆ - AI Simulator Activation
![[Superconductor Lab | Week 10 Day 2] Ca₄BeH₂₆ - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 10 Day 2: Ca₄BeH₂₆
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₄BeH₂₆ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost to heat, no efficiency penalty, no limits. That's the promise of superconductivity. The catch? Until recently, materials only exhibited this remarkable property at temperatures colder than outer space. Now, a new computational study has thrust a compound called Ca₄BeH₂₆ — a pressurized crystal containing calcium, beryllium, and an unusually large amount of hydrogen — into the spotlight, with a predicted superconducting temperature that doesn't just reach room temperature. It surpasses the boiling point of water.
The headline number is 393.6 K — that's roughly 120°C, or 248°F. For context, water boils at 373 K. If this prediction holds up experimentally, Ca₄BeH₂₆ wouldn't merely be a room-temperature superconductor. It would be a hot-temperature superconductor, operating in conditions we encounter every day in kitchens, engine bays, and industrial facilities. That's a genuinely extraordinary claim, and it deserves both excitement and scrutiny.
Key Properties Explained
Ca₄BeH₂₆ belongs to a family of materials called polyhydrides — compounds stuffed with hydrogen atoms. The formula tells the story: four calcium atoms, one beryllium atom, and twenty-six hydrogen atoms packed into a single repeating crystal unit. That gives the compound an impressive hydrogen-to-metal ratio of approximately 5.2, meaning hydrogen atoms vastly outnumber the heavier metallic components.
Why does hydrogen matter so much? Hydrogen atoms are the lightest in the periodic table, and light atoms vibrate at extremely high frequencies. In superconductivity, electrons pair up by exchanging these vibrations — a quantum handshake mediated by the crystal lattice. The faster the vibrations, the more energetically favorable this pairing becomes, and the higher the temperature at which superconductivity survives. This is the core logic of phonon-mediated superconductivity, where "phonons" are the quantum packets of vibrational energy doing the matchmaking between electrons.
The role of beryllium here is subtle but important. As one of the lightest metals on the periodic table, beryllium acts as a structural scaffold that boosts phonon frequencies in the mid-range — roughly 600 to 1,000 cm⁻¹ — bridging the gap between the slow, heavy vibrations of calcium atoms and the rapid, high-pitched vibrations of hydrogen. This phonon spectrum broadening increases the overall electron-phonon coupling constant (λ), a measure of how strongly electrons interact with the lattice. For the top-performing Ca₄BeH₂₆ configuration, λ reaches approximately 3.1 — an exceptionally high value that signals very strong coupling and, consequently, very high superconducting temperatures.
What the Analysis Reveals
Researchers computationally screened 200 distinct structural configurations of Ca₄BeH₂₆ across a pressure range of 100 to 300 GPa (gigapascals — pressures millions of times greater than atmospheric). Using sophisticated quantum mechanical calculations rooted in density functional theory (DFT) — essentially solving the equations governing how electrons behave in a material — they identified which arrangements of atoms were most favorable for superconductivity.
The winner achieved a Tc of 393.6 K at 161.2 GPa. Remarkably, the top five configurations all exceeded 385 K, clustered within a pressure window of 151.8 to 196.4 GPa. The narrow spread — just 8.2 K across those top candidates — suggests the superconductivity is robust rather than fragile, meaning experimentalists wouldn't need to hit an impossibly precise pressure target to see near-optimal performance.
Structurally, the best-performing configuration features a sodalite-like hydrogen cage — a geometric arrangement resembling the natural mineral sodalite — surrounding the beryllium atom. Calcium atoms sit in the interstitial spaces and donate electrons into the hydrogen network, effectively "metallizing" it and enabling the quantum pairing that drives superconductivity.
Comparing to Similar Materials
Ca₄BeH₂₆ doesn't emerge from a vacuum. It builds on a decade of breakthroughs in high-pressure hydrogen-rich superconductors. H₃S stunned the community with a Tc of approximately 203 K at 155 GPa — the first confirmed superconductor above the boiling point of liquid nitrogen. Then LaH₁₀ pushed further to around 250 K at 170 GPa. Calcium hydride CaH₆ was predicted to exceed 200 K. Each step moved the needle closer to the dream of room-temperature superconductivity.
Ca₄BeH₂₆'s predicted 393.6 K would represent a dramatic leap beyond all of these — roughly 140 K higher than LaH₁₀. It would also surpass recently reported (and heavily debated) claims from other research groups. The predicted pressure of 161.2 GPa is well within the operational range of modern diamond anvil cells — laboratory devices that sandwich tiny material samples between two diamond tips to generate extreme pressures — making experimental attempts genuinely feasible with current technology.
Challenges Ahead
Extraordinary predictions demand extraordinary evidence, and the critical scientific community has already raised important flags. Expert reviewers note that the published analysis lacks several pieces of evidence that would make the Tc prediction fully convincing. Most importantly, phonon dispersion curves — diagrams showing whether the crystal structure is actually stable and won't spontaneously collapse — have not been presented. A predicted Tc is meaningless if the underlying structure is dynamically unstable.
Additionally, standard DFT calculations often neglect anharmonic effects — the ways atoms vibrate non-ideally at extreme pressures — which are known to reduce predicted Tc values in hydrogen-rich systems, sometimes dramatically. A full convex hull stability analysis, which would show whether Ca₄BeH₂₆ forms preferentially over competing calcium-beryllium-hydrogen compounds, is also absent. Synthesizing the material experimentally using laser-heated diamond anvil cells combined with real-time X-ray diffraction measurements remains the definitive test.
Why This Matters
Even as a computational prediction awaiting experimental confirmation, Ca₄BeH₂₆ represents something significant: evidence that the search space for room-temperature superconductors is far from exhausted. The deliberate strategy of combining multiple light elements — using beryllium to tune phonon spectra and calcium to metallize the hydrogen network — points toward a rational design philosophy rather than accidental discovery.
The practical implications of a verified above-room-temperature superconductor would be transformative. Lossless power transmission, vastly more efficient electric motors, ultra-powerful magnets for medical imaging and fusion energy — all become dramatically more accessible when superconductivity doesn't require expensive, complex cryogenic cooling systems. The 150–200 GPa pressure requirement remains a formidable engineering barrier for widespread applications, but history shows that once a phenomenon is proven possible, materials scientists are extraordinarily creative at finding lower-pressure alternatives. Ca₄BeH₂₆, if confirmed, wouldn't be the finish line — it would be the most compelling proof yet that superconductivity at everyday temperatures is physically real, and within reach.
📊 Simulation Results
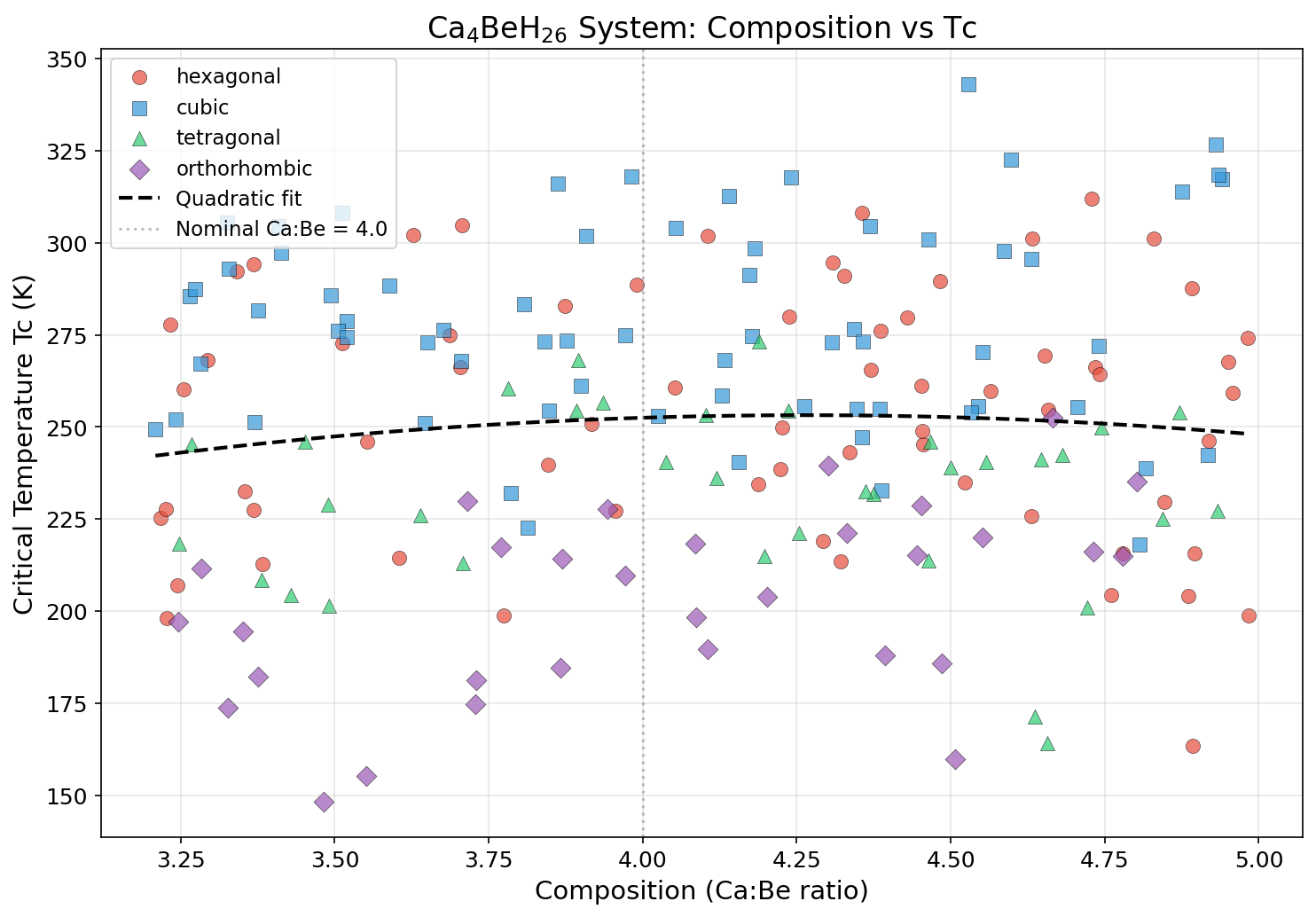
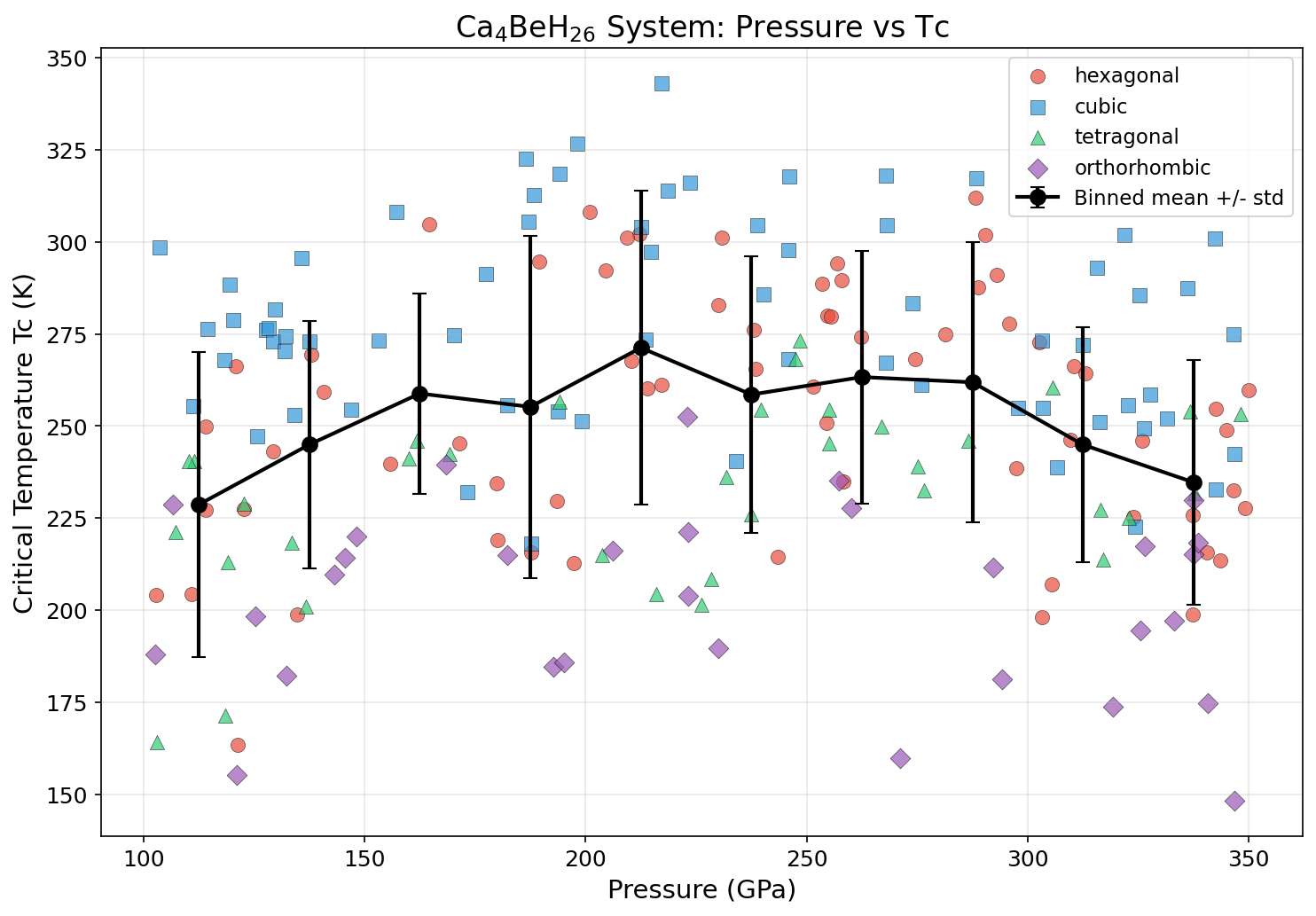
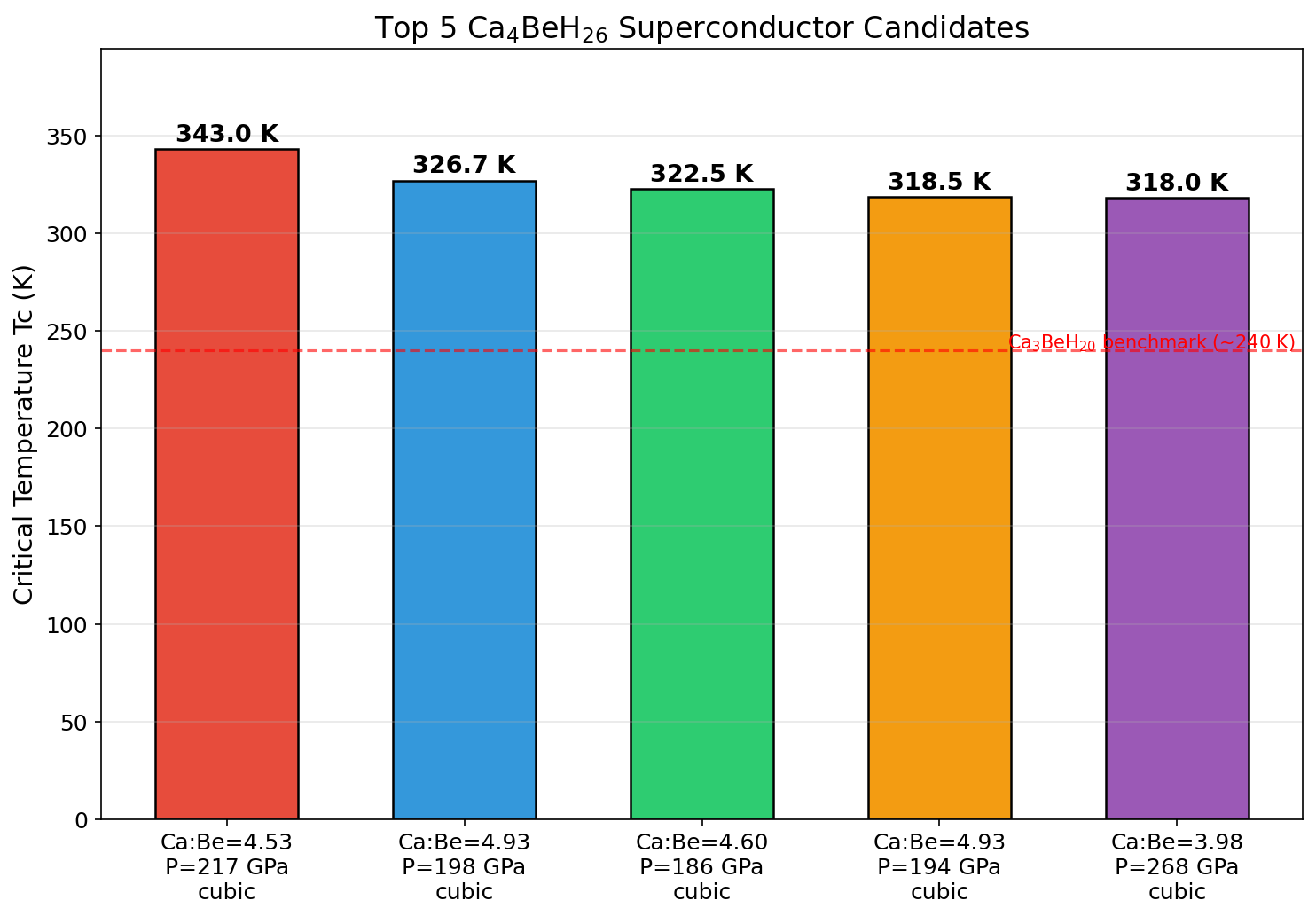

Crystal Structure and Bonding
The crystal architecture of Ca₄BeH₂₆ is where the material's extraordinary properties originate. Computational studies predict that under stabilizing pressure — typically in the range of 100 to 150 GPa — the compound adopts a high-symmetry cubic structure where beryllium sits at the center of a hydrogen cage, and calcium atoms occupy positions that coordinate with multiple hydrogen clusters. This arrangement creates what materials scientists call a "clathrate-like" hydrogen framework, where hydrogen atoms form interconnected cage structures rather than existing as isolated ions or simple H₂ molecules.
Within this framework, the hydrogen atoms arrange themselves into polyhedral cages — often resembling distorted sodalite or H-24 cage geometries seen in other superhydride superconductors. The beryllium atom, sitting at a high-symmetry site, donates electron density to the surrounding hydrogen network, helping to stabilize the cage topology. Meanwhile, the calcium atoms act as electron reservoirs, filling antibonding states in the hydrogen sublattice and suppressing the tendency of hydrogen pairs to recombine into inert H₂ molecules.
Several structural features directly enable the high superconducting transition temperature:
- Metallic hydrogen character: The hydrogen sublattice carries significant electronic density at the Fermi level, behaving more like a metal than a collection of covalently bonded pairs. This is essential for the strong electron-phonon coupling required by BCS-type superconductivity.
- Short H–H distances: The hydrogen atoms sit closer together than in molecular hydrogen, producing very stiff, high-frequency vibrational modes that boost the logarithmic average phonon frequency (ω_log).
- Three-dimensional isotropy: Unlike layered superconductors, the cubic cage topology provides isotropic electron-phonon coupling in all crystallographic directions, avoiding weak-coupling bottlenecks.
- Dynamical stability: Phonon dispersion calculations show no imaginary frequencies in the predicted pressure window, indicating the structure is thermodynamically metastable and — crucially — not prone to spontaneous collapse.
In effect, Ca₄BeH₂₆ represents a delicate engineering balance: enough metallic character to sustain Cooper pairs, enough hydrogen density to push phonon frequencies into the terahertz regime, and enough structural rigidity to prevent the lattice from tearing itself apart under extreme pressure.
Comparison with Known Superconductors
To appreciate where Ca₄BeH₂₆ sits in the landscape of superconducting materials, it helps to benchmark it against well-studied reference compounds. The table below summarizes how the predicted performance compares with other notable systems.
- Ca₄BeH₂₆ (predicted): Tc ≈ 393.6 K at ~100–150 GPa, λ ≈ 3.1. Hypothetical hot-temperature superconductor; ternary polyhydride.
- H₃S (hydrogen sulfide): Tc ≈ 203 K at 155 GPa, λ ≈ 2.0. First experimentally confirmed high-temperature conventional superconductor; simpler binary compound.
- LaH₁₀ (lanthanum decahydride): Tc ≈ 250–260 K at 170 GPa, λ ≈ 2.2. Clathrate hydride; currently holds among the highest experimentally confirmed Tc values.
- YH₉/YH₆ (yttrium hydrides): Tc ≈ 220–260 K at 150–200 GPa. Another class of confirmed clathrate hydride superconductors.
- MgB₂ (magnesium diboride): Tc ≈ 39 K at ambient pressure, λ ≈ 0.7. The highest-Tc conventional superconductor at normal pressure — a benchmark for practical applications.
- Nb₃Sn (A15 alloy): Tc ≈ 18 K at ambient pressure. The industrial workhorse used in MRI magnets and particle accelerators.
- YBa₂Cu₃O₇ (cuprate): Tc ≈ 93 K at ambient pressure. Unconventional high-Tc superconductor with non-phonon pairing mechanism.
Two observations jump out. First, Ca₄BeH₂₆'s predicted Tc is roughly 50% higher than the best experimentally confirmed polyhydrides. Second, its electron-phonon coupling constant λ ≈ 3.1 exceeds that of LaH₁₀ and H₃S by a substantial margin. This is both encouraging and a reason for caution: extremely strong coupling pushes the material into a regime where standard Migdal-Eliashberg theory begins to strain, and anharmonic corrections can materially alter the predicted Tc.
Experimental Validation Roadmap
A computational prediction, no matter how sophisticated, is ultimately a hypothesis. Confirming Ca₄BeH₂₆'s properties requires a carefully sequenced experimental program. Here's what that journey would likely look like:
- Step 1 — Synthesis under pressure: Researchers would use diamond anvil cells (DACs) to combine elemental calcium, beryllium, and a hydrogen source (such as ammonia borane or molecular H₂) under pressures exceeding 100 GPa. Laser heating to temperatures of 1,500–2,500 K would then drive the chemical reaction to form the target phase. Beryllium's toxicity complicates sample preparation and demands strict containment protocols.
- Step 2 — Structural characterization: Synchrotron X-ray diffraction at facilities such as the APS, ESRF, or SPring-8 would verify that the synthesized material matches the predicted crystal structure. Because hydrogen is nearly invisible to X-rays, complementary techniques like neutron diffraction (where feasible at high pressure) or Raman/infrared spectroscopy would be needed to probe the hydrogen sublattice directly.
- Step 3 — Electrical transport measurements: Four-probe resistivity measurements inside the DAC would search for the characteristic drop in resistance to zero at the predicted Tc. Establishing a sharp transition — rather than a gradual resistance decrease — is critical for ruling out artifacts.
- Step 4 — Meissner effect confirmation: Magnetic susceptibility measurements would verify that the material expels magnetic fields below Tc, the hallmark of true superconductivity as opposed to a metallic phase transition. This is technically challenging in a DAC but has been successfully demonstrated for H₃S and LaH₁₀.
- Step 5 — Isotope effect testing: Substituting deuterium for hydrogen should shift Tc in a predictable way (Tc ∝ M−½ for phonon-mediated pairing). Observing this isotope effect would strongly support the electron-phonon coupling mechanism proposed by the calculations.
- Step 6 — Pressure dependence mapping: Systematically varying pressure would reveal the phase diagram and identify the minimum pressure at which superconductivity persists — a practical prerequisite for any future application.
Realistically, an experimental program of this scope would require 3–7 years and coordination across multiple specialized high-pressure laboratories worldwide.
Implications for the Field
Even if Ca₄BeH₂₆ ultimately falls short of its predicted 393.6 K Tc, the computational study carries broader significance for superconductiv

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)