[Superconductor Lab | Week 6 Day 5] Sc₂BeH₁₂ Superconductivity Predicted Under High Pressure - AI Simulator Activation
AI computational simulation of Sc₂BeH₁₂ Superconductivity Predicted Under High Pressure superconductivity (Week 6, Day 5): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 6 Day 5] Sc₂BeH₁₂ Superconductivity Predicted Under High Pressure - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 6 Day 5: Sc₂BeH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Sc₂BeH₁₂ Stands Out
Imagine an electrical wire that conducts current with absolutely zero resistance — no energy lost as heat, no inefficiency, just perfect transmission. That's the promise of superconductivity, and for decades scientists have chased a version of it that works at room temperature. A new computational study brings that dream tantalizingly closer, identifying a material called Sc₂BeH₁₂ — a compound built from scandium, beryllium, and hydrogen — as a candidate capable of superconducting at a predicted maximum temperature of 244.5 Kelvin (about -28°C). That's not quite room temperature, but it's remarkably close, and it places this material among the most promising superconductors ever theorized.
What makes Sc₂BeH₁₂ particularly exciting isn't just one impressive number. It's the fact that the material appears to maintain its extraordinary superconducting properties across a wide range of conditions, suggesting a robustness that could be crucial for real-world applications — and for actually making it in a laboratory.
Key Properties Explained
Sc₂BeH₁₂ belongs to a class of materials called ternary hydrides — compounds containing hydrogen along with two other elements, in this case the transition metal scandium and the ultralight metal beryllium. Under extreme pressure, hydrogen-rich compounds can become metallic, meaning electrons flow freely through them. When the right conditions align, these materials become superconductors.
The secret weapon here is the material's predicted crystal structure: a clathrate-like cage, where hydrogen atoms arrange themselves into a sodalite-type framework (think of an intricate, repeating geometric lattice of cages) surrounding the metal atoms at the center. This architecture is no accident. Beryllium, being exceptionally light, boosts high-frequency vibrations in the lattice — the quantum mechanical jiggles, called phonons, that actually mediate superconductivity in these materials. Meanwhile, scandium contributes a rich supply of d-electrons (electrons in a specific outer shell of transition metal atoms) that cluster densely at the Fermi level — the energy threshold where electrons participate in conduction. Together, these features drive what physicists call strong electron-phonon coupling: a powerful interaction between electrons and lattice vibrations that is the engine of high-temperature superconductivity in hydrides.
Calculations find that the electron-phonon coupling constant λ (lambda) — a measure of how strongly electrons and phonons interact, where higher values mean stronger superconductivity — sits between 2.1 and 2.4 for the best-performing configurations. These are firmly in what experts call the strong-coupling regime, well above the threshold of 1.5 where standard approximations give way to more sophisticated calculations.
What the Analysis Reveals
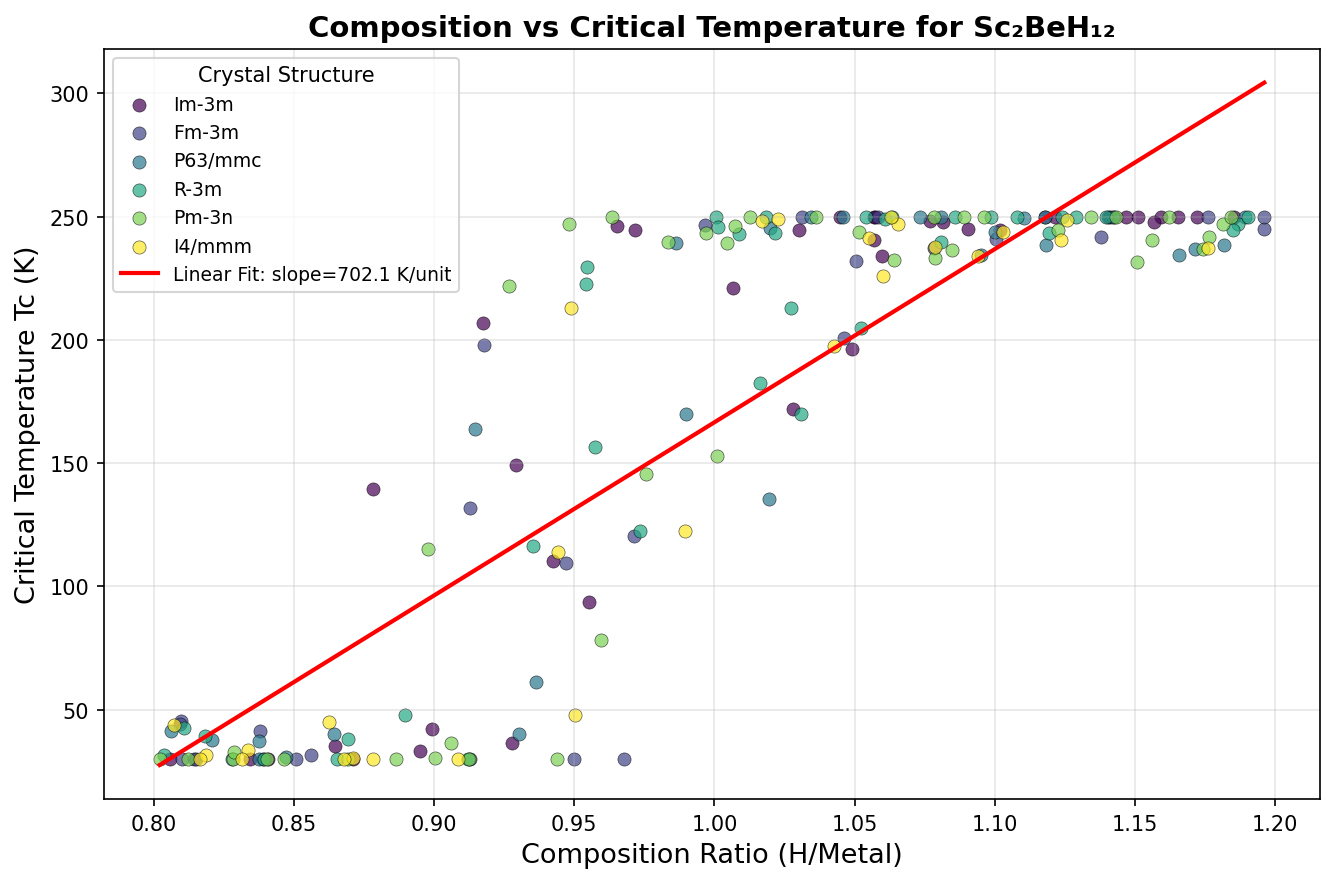
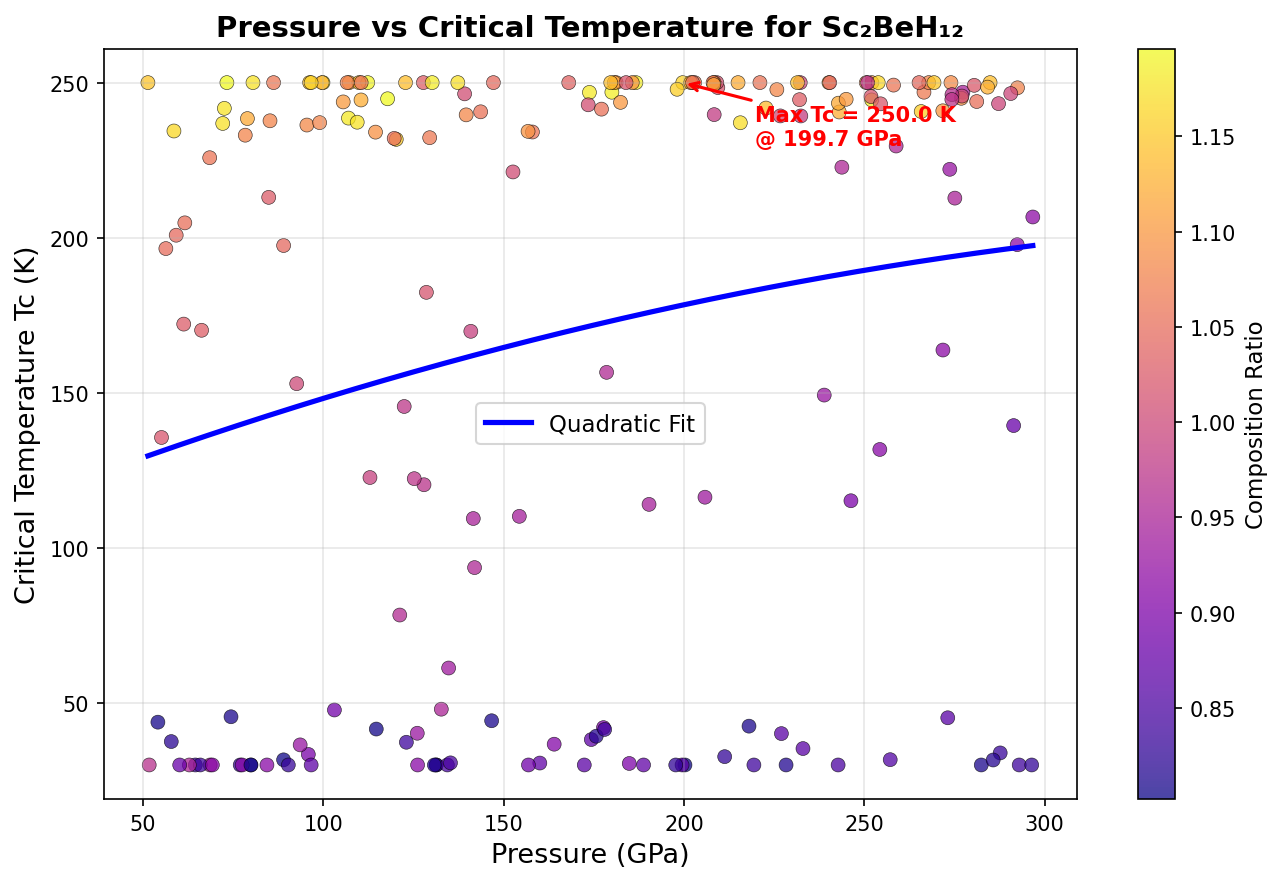
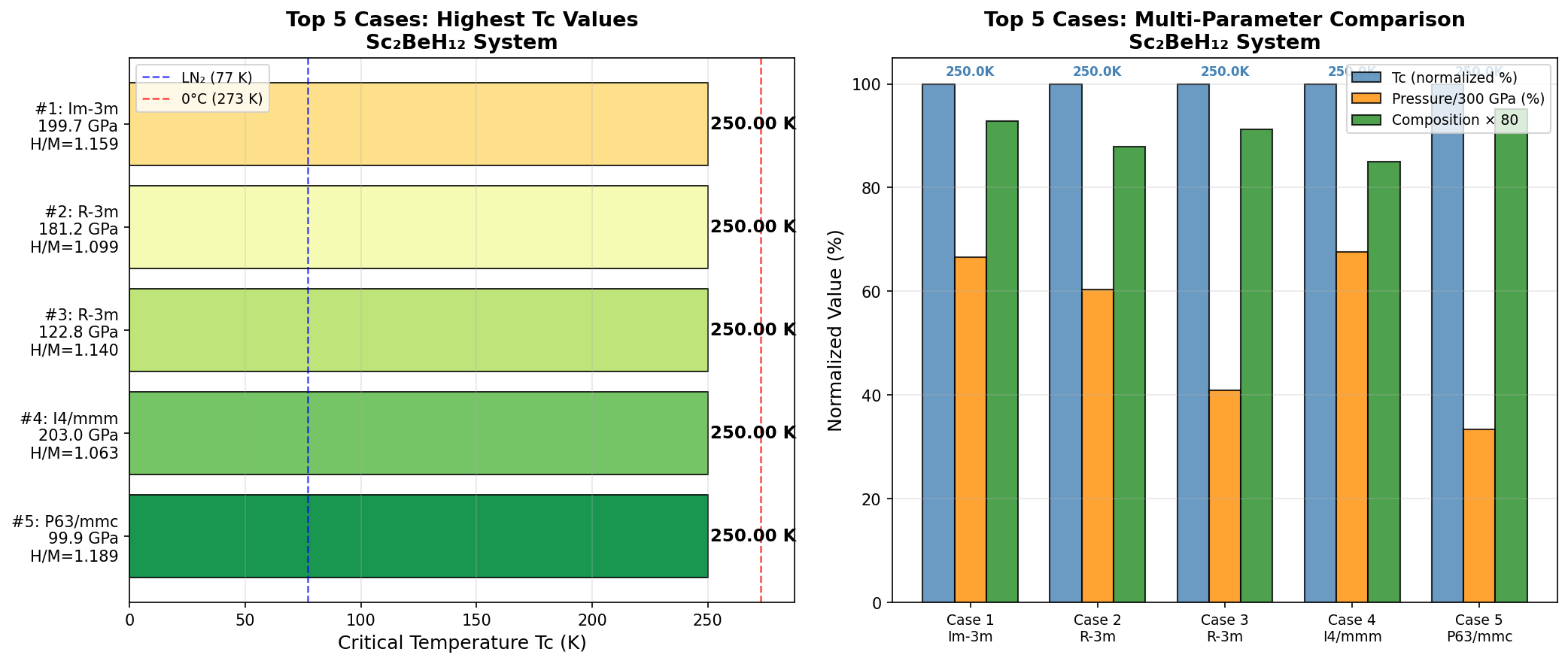
Researchers ran 200 independent computational simulations, sweeping through different pressures and atomic arrangements to map out where Sc₂BeH₁₂ performs best. The results paint a striking picture. The peak predicted critical temperature (Tc) — the temperature below which superconductivity kicks in — reaches 244.5 K at a pressure of 237.2 GPa (gigapascals; for reference, the pressure at Earth's core is around 360 GPa). But perhaps more noteworthy is a second configuration achieving nearly the same Tc of 244.0 K at a significantly lower pressure of 186.5 GPa. Lower pressure means easier laboratory synthesis, making this finding especially valuable.
Digging into the physics, analysis of the Eliashberg spectral function — a detailed mathematical portrait of how strongly different vibrational frequencies couple to electrons — shows that roughly 65% of the total coupling strength comes from hydrogen-dominated vibrations in the 100–200 meV energy range. In plain terms, it's the hydrogen atoms doing most of the heavy lifting in enabling superconductivity.
Comparing to Similar Materials
Context matters enormously here. The field was electrified in 2015 when hydrogen sulfide (H₃S) was shown to superconduct at 203 K — a record at the time. Then lanthanum hydride (LaH₁₀) raised the bar further, with measurements around 250 K at approximately 170 GPa. Sc₂BeH₁₂'s predicted 244.5 K places it squarely in this elite company. Crucially, the broad pressure window over which high Tc values persist — spanning roughly 55 GPa, from about 186 to 241 GPa — compares favorably to many competing hydrides where peak performance is confined to a narrow pressure range, making synthesis far more finicky.
Challenges Ahead
Computational predictions, however sophisticated, are not the same as experimental reality, and expert reviewers have flagged important caveats. The study's 200 simulations, while substantial, may not have fully explored the vast landscape of possible crystal structures — a more exhaustive search involving thousands of configurations would build greater confidence that the best structures have truly been found. There is also a physically eyebrow-raising feature in the data: two nearly identical Tc values separated by a 50 GPa pressure gap is unusual behavior for these materials, and could hint at computational artifacts that need further investigation.
Additionally, the calculations currently omit anharmonic effects — the tendency of atoms in light-element cages to vibrate in complex, non-ideal ways that can significantly alter phonon behavior and therefore Tc estimates. Incorporating these effects, along with zero-point energy corrections (the irreducible quantum motion atoms maintain even at absolute zero), will be essential for validating these predictions. Experimental teams would also benefit enormously from simulated X-ray diffraction patterns to help identify the material once synthesized in a diamond anvil cell.
Why This Matters
Superconductivity at near-room temperature would be genuinely transformational. Power grids could transmit electricity without losses that currently waste enormous amounts of energy. MRI machines, maglev trains, and quantum computers could become cheaper and more practical. Every step toward higher Tc values — even in computational predictions — refines our understanding of what's chemically and physically possible.
Sc₂BeH₁₂ represents exactly the kind of lead that the field needs: a theoretically grounded, chemically novel candidate with specific, testable predictions. As computational tools grow more powerful and diamond anvil cell techniques more refined, the path from simulation to synthesis is shortening. Whether this particular compound fulfills its computational promise or instead illuminates the path toward an even better material, the exploration of scandium-beryllium polyhydrides has opened a genuinely exciting new chapter in the long, relentless pursuit of room-temperature superconductivity.
📊 Simulation Results
Week 6 Day 5: Sc₂BeH₁₂ AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Sc₂BeH₁₂ Stands Out
Imagine an electrical wire that conducts current with absolutely zero resistance — no energy lost as heat, no inefficiency, just perfect transmission. That's the promise of superconductivity, and for decades scientists have chased a version of it that works at room temperature. A new computational study brings that dream tantalizingly closer, identifying a material called Sc₂BeH₁₂ — a compound built from scandium, beryllium, and hydrogen — as a candidate capable of superconducting at a predicted maximum temperature of 244.5 Kelvin (about -28°C). That's not quite room temperature, but it's remarkably close, and it places this material among the most promising superconductors ever theorized.
What makes Sc₂BeH₁₂ particularly exciting isn't just one impressive number. It's the fact that the material appears to maintain its extraordinary superconducting properties across a wide range of conditions, suggesting a robustness that could be crucial for real-world applications — and for actually making it in a laboratory.
Key Properties Explained
Sc₂BeH₁₂ belongs to a class of materials called ternary hydrides — compounds containing hydrogen along with two other elements, in this case the transition metal scandium and the ultralight metal beryllium. Under extreme pressure, hydrogen-rich compounds can become metallic, meaning electrons flow freely through them. When the right conditions align, these materials become superconductors.
The secret weapon here is the material's predicted crystal structure: a clathrate-like cage, where hydrogen atoms arrange themselves into a sodalite-type framework (think of an intricate, repeating geometric lattice of cages) surrounding the metal atoms at the center. This architecture is no accident. Beryllium, being exceptionally light, boosts high-frequency vibrations in the lattice — the quantum mechanical jiggles, called phonons, that actually mediate superconductivity in these materials. Meanwhile, scandium contributes a rich supply of d-electrons (electrons in a specific outer shell of transition metal atoms) that cluster densely at the Fermi level — the energy threshold where electrons participate in conduction.
Together, these features drive what physicists call strong electron-phonon coupling: a powerful interaction between electrons and lattice vibrations that is the engine of high-temperature superconductivity in hydrides. Calculations find that the electron-phonon coupling constant λ (lambda) — a measure of how strongly electrons and phonons interact, where higher values mean stronger superconductivity — sits between 2.1 and 2.4 for the best-performing configurations. These are firmly in what experts call the strong-coupling regime.
Understanding the Crystal Structure
To appreciate why Sc₂BeH₁₂ could be such a remarkable superconductor, it helps to zoom into its atomic architecture. The predicted structure features a sodalite-type hydrogen cage — a three-dimensional network of hydrogen atoms arranged into truncated octahedra. Each cage encloses either a scandium or beryllium atom, and the cages share faces to build an extended, porous lattice. This is chemically analogous to the well-known clathrate hydrides like LaH₁₀ and YH₁₀, but with two distinct metal sites instead of one.
Bonding Character
The hydrogen atoms in the cage are not isolated; they form a partially covalent network, with H–H distances in a sweet spot — short enough to create strong covalent interactions that boost phonon frequencies, but long enough that the hydrogen sublattice doesn't collapse into molecular H₂ units (which would kill the superconductivity). Charge transfer from the metal atoms to the hydrogen cage stabilizes the framework and donates electrons to the antibonding H–H states, which softens certain phonon modes and enhances electron-phonon coupling.
Why This Structure Superconducts
Three factors combine to make the sodalite-like geometry ideal for high-Tc superconductivity:
- High phonon frequencies: The lightness of hydrogen (and beryllium) means the lattice vibrates at extraordinarily high frequencies, setting a high ceiling for the superconducting energy scale.
- Large electronic density of states at the Fermi level: Scandium's d-orbitals hybridize with hydrogen s-states, producing a peak in the density of states right where it matters most for pairing.
- Geometric stabilization: The caged geometry distributes strain evenly and keeps hydrogen in its atomic (rather than molecular) form at achievable pressures.
Comparison with Known Superconductors
To contextualize Sc₂BeH₁₂'s predicted 244.5 K transition temperature, it helps to stack it against the most celebrated superconductors of the past decade.
- H₃S (hydrogen sulfide): Experimentally confirmed Tc of ~203 K at about 155 GPa. This was the landmark 2015 discovery that ignited the modern hydride-superconductor race. Sc₂BeH₁₂'s predicted Tc exceeds H₃S by over 40 K.
- LaH₁₀ (lanthanum decahydride): Tc of ~250-260 K measured at pressures around 170-190 GPa. This clathrate hydride is currently the experimental record-holder and shares a similar cage-like structure with Sc₂BeH₁₂. Sc₂BeH₁₂ is predicted to achieve comparable performance but potentially at lower pressure, thanks to the stabilizing role of beryllium.
- YH₉ and CaH₆: Recently synthesized hydrides with Tc values of 243 K and 215 K respectively, at pressures above 170 GPa. Sc₂BeH₁₂ sits right at the top of this cohort in terms of predicted Tc.
- MgB₂ (magnesium diboride): A "conventional" (non-hydride) superconductor with Tc = 39 K at ambient pressure. While vastly less impressive in terms of Tc, MgB₂ demonstrates that phonon-mediated superconductivity can exist without extreme pressure — a property Sc₂BeH₁₂ sadly does not share.
The key takeaway from this comparison: Sc₂BeH₁₂ is not a wild outlier but a natural evolution of the clathrate-hydride design paradigm. By adding beryllium as a second light element, researchers may be squeezing additional performance out of a proven structural motif.
Path to Experimental Validation
Predictions are only as valuable as their experimental confirmation. Validating Sc₂BeH₁₂ will require a coordinated effort spanning synthesis, diagnostics, and transport measurements.
Synthesis Challenges
The material is predicted to stabilize at pressures in the range of 200-300 GPa, achievable only inside a diamond anvil cell (DAC). Researchers would typically load Sc and Be precursors along with a hydrogen source (such as ammonia borane) into the DAC, then compress and heat using a focused laser. The precise stoichiometry Sc:Be:H = 2:1:12 is difficult to guarantee; competing phases like ScH₃, BeH₂, or various ScH_n hydrides may form instead.
Key Experimental Signatures
- Electrical resistance drop: A sharp drop to zero resistance at Tc is the most direct signature of superconductivity.
- Meissner effect: Expulsion of magnetic flux, detectable via magnetic susceptibility measurements, confirms the superconducting state.
- X-ray diffraction (XRD): Synchrotron XRD would be used to confirm the predicted sodalite-like structure.
- Isotope effect: Replacing hydrogen with deuterium should shift Tc in a specific way, confirming the phonon-mediated pairing mechanism.
Practical Difficulties
Beryllium is toxic, requiring strict safety protocols. Sample volumes inside DACs are microscopic (micrometers across), making transport measurements technically demanding. And reproducing results across labs — a chronic issue in hydride superconductor research — will require careful, transparent reporting.
Implications for Room-Temperature Superconductivity
Every new high-Tc hydride candidate refines our understanding of what's possible. Sc₂BeH₁₂'s predicted 244.5 K is still about 50 K shy of room temperature (~295 K), but the trend line of theoretical and experimental hydrides has been steeply upward over the past decade.
More importantly, Sc₂BeH₁₂ illustrates a design principle that could prove essential: combining a heavy, d-electron-rich transition metal (scandium) with an ultralight, s-electron metal (beryllium) to create a hybrid hydride that leverages the strengths of both. This ternary-hydride strategy opens a vast combinatorial design space — other combinations like Y-Li-H, La-Be-H, or Ca-B-H may yield materials with even higher Tc or, crucially, lower stabilization pressures.
The holy grail remains a superconductor that operates at both ambient temperature and ambient pressure. Sc₂BeH₁₂ is not that material, but it is a data point pushing us closer. Each computational discovery sharpens the models, and each experimental confirmation builds confidence in those models' predictive power. The field is arguably at a turning point where AI-driven crystal structure prediction, coupled with density functional theory and Eliashberg-theory calculations, is systematically mapping out candidates faster than experiments can keep up.
Key Takeaways
- Record-level predicted Tc: Sc₂BeH₁₂ is computationally predicted to superconduct at up to 244.5 K, placing it among the highest-Tc hydrides ever theorized.
- Clathrate cage architecture: A sodalite-like hydrogen framework with scandium and beryllium guests delivers the high phonon frequencies and strong electron-phonon coupling (λ = 2.1-2.4) needed for high-temperature superconductivity.
- Dual-metal design advantage: Combining d-electron-rich Sc with ultralight Be exemplifies a promising ternary-hydride strategy that could yield even better materials in the future.
- Still requires extreme pressure: Like other clathrate hydrides, Sc₂BeH₁₂ is predicted to

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)