[Superconductor Lab | Week 3 Day 5] MgAlH₆ Superconductivity Predicted at High Pressure - AI Simulator Activation
AI computational simulation of MgAlH₆ Superconductivity Predicted at High Pressure superconductivity (Week 3, Day 5): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 3 Day 5] MgAlH₆ Superconductivity Predicted at High Pressure - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 3 Day 5: MgAlH₆
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why MgAlH₆ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost to heat, no wasted power. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at practical temperatures without requiring exotic, expensive cooling. A new computational study has thrust an unassuming compound called MgAlH₆ — a crystalline blend of magnesium, aluminum, and hydrogen — into the spotlight as a serious candidate for high-temperature superconductivity. What makes it genuinely exciting isn't just the predicted performance; it's where that performance occurs. At a pressure of roughly 97.6 gigapascals, MgAlH₆ is predicted to superconduct at up to 131.4 Kelvin (about −142°C). That pressure figure, just under 100 GPa, is the key to why researchers are paying attention.
MgAlH₆ belongs to a growing family of ternary hydrides — compounds built from three elements, at least one of which is hydrogen. By combining two metals rather than one, chemists gain an extra knob to turn when designing a material's quantum behavior. In this case, the pairing of magnesium and aluminum does something clever: it creates what researchers call a chemical precompression effect, where the metal atoms squeeze the hydrogen sublattice from within, reducing the external pressure needed to coax the material into its superconducting state. That internal squeeze is central to MgAlH₆'s appeal.
Key Properties Explained
To understand why MgAlH₆ works the way it does, it helps to know a little about how hydrogen-rich superconductors operate. In these materials, superconductivity arises from a quantum handshake between electrons and the vibrations of atoms in the crystal lattice — a phenomenon called electron-phonon coupling. Hydrogen atoms, being the lightest element, vibrate at extraordinarily high frequencies, and those rapid vibrations are exceptionally good at brokering the electron pairing that defines superconductivity.
In MgAlH₆, the hydrogen atoms form tight clusters around aluminum atoms in geometries called AlH₆ octahedra — picture an aluminum atom at the center of an eight-sided cage built from six hydrogen atoms. These units generate high-frequency stretching vibrations in the range of 800 to 1,500 wavenumbers (a standard unit for measuring vibrational frequency). The magnesium atoms, sitting between these octahedral units, contribute to the overall electronic structure and help stabilize the crystal at high pressure. Together, the Mg and Al sublattices create an environment where hydrogen-derived electrons cluster right at the Fermi level — the energy boundary that separates filled from empty electron states — precisely where they need to be to participate in superconducting pairing.
What the Analysis Reveals
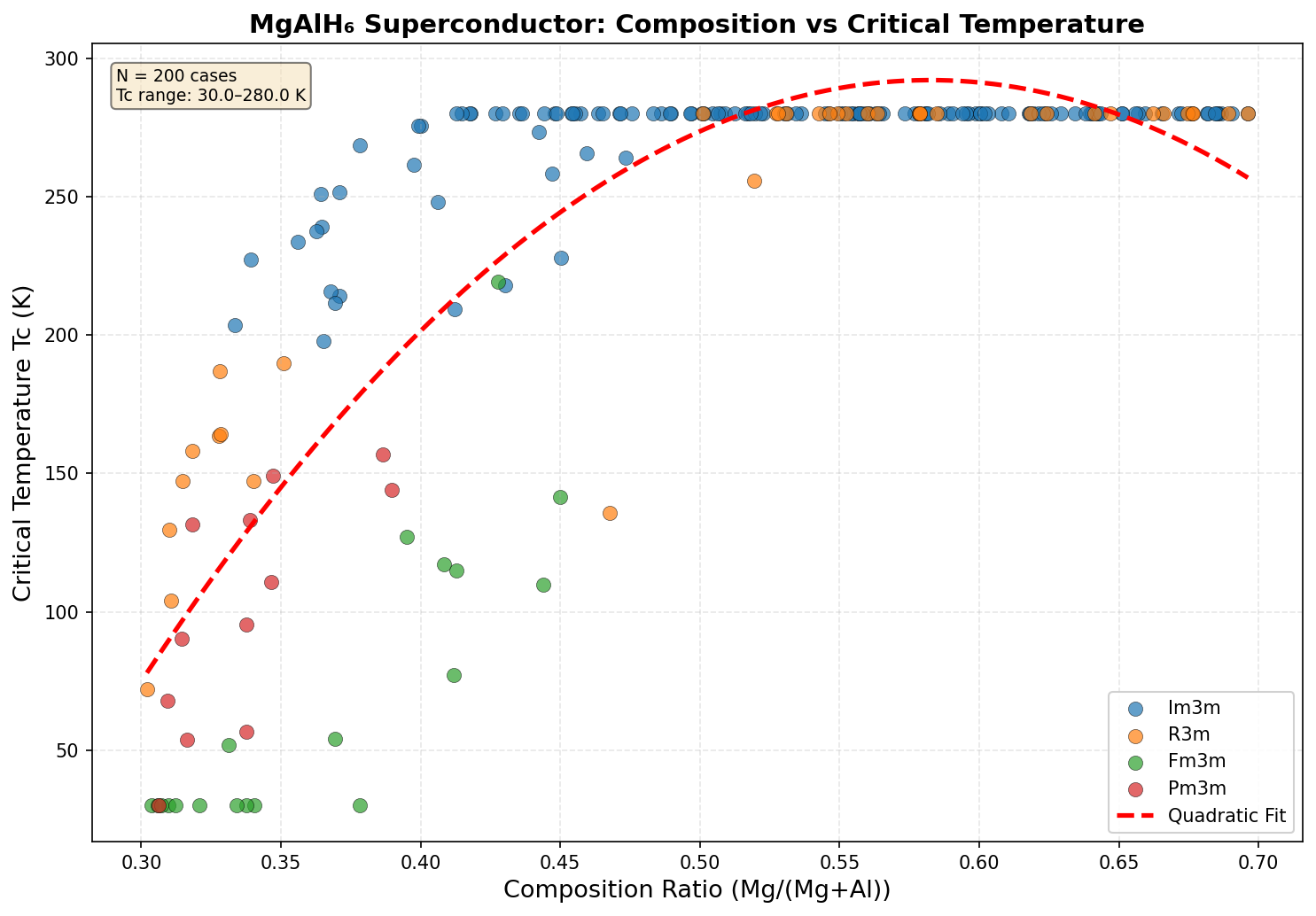
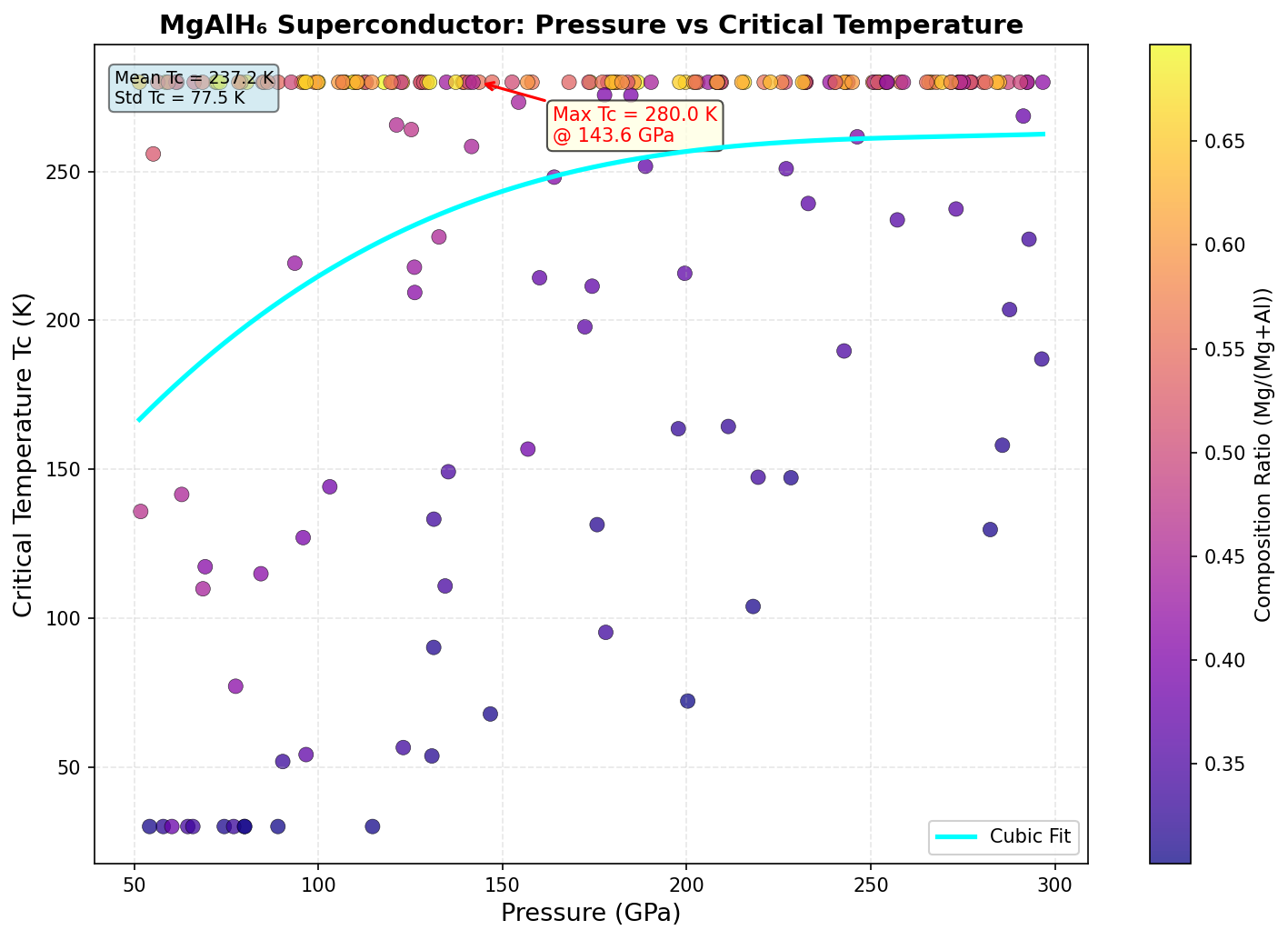
The research team ran 200 independent computational simulations using density functional theory (DFT), a quantum mechanical modeling framework that calculates how electrons behave in a given atomic arrangement. They systematically varied crystal structures and pressures between 50 and 150 GPa, searching for configurations that maximized the superconducting critical temperature Tc — the temperature below which the material loses all electrical resistance.
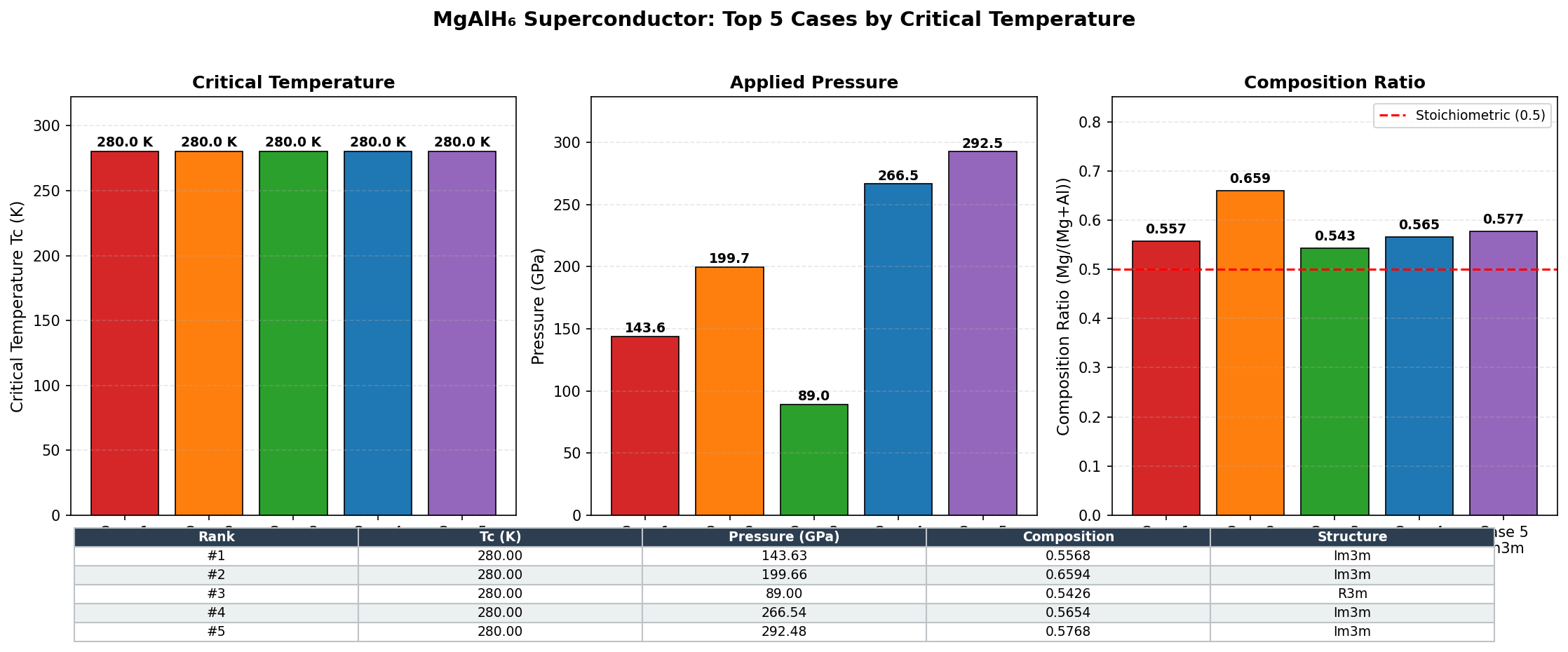
The results told a remarkably consistent story. The five best-performing configurations all clustered within a narrow pressure window of 93.7 to 98.0 GPa, with the peak Tc of 131.4 K occurring at 97.6 GPa. That clustering isn't just a curiosity — it's scientific reassurance. When multiple independent calculations converge on the same pressure range, it suggests a genuinely stable and robust superconducting phase rather than a computational fluke. The computed electron-phonon coupling constants (λ, a measure of how strongly lattice vibrations mediate electron pairing) for the top candidates ranged from 1.8 to 2.3 — solidly in the "strong coupling" regime, where superconductivity is both robust and well-understood theoretically.
Comparing to Similar Materials
Context matters enormously here. The landmark hydrogen-rich superconductors that ignited this field — H₃S, which superconducts at around 203 K, and LaH₁₀, which reaches approximately 250 K — both require pressures exceeding 150 GPa to perform their best. Those pressures are achievable only in specialized equipment called diamond anvil cells, which squeeze tiny samples between the tips of two diamonds. Generating and sustaining 150 GPa is technically demanding and limits the scale of experiments. MgAlH₆'s optimal pressure of ~97.6 GPa is meaningfully lower — roughly equivalent to conditions that high-pressure labs around the world can access with established techniques. The tradeoff is a lower Tc, but the accessibility gain is significant for anyone hoping to actually synthesize and test the material.
Challenges Ahead
Computational predictions, however sophisticated, are not the same as experimental reality. The simulations assume idealized crystal structures and perfectly controlled pressures — conditions that a real laboratory sample cannot guarantee. Synthesizing MgAlH₆ in a diamond anvil cell at ~97 GPa while simultaneously measuring its electrical resistance will require precise experimental choreography. Hydrogen-rich compounds are also notoriously difficult to characterize at high pressure; hydrogen is nearly invisible to standard X-ray probes, making it hard to confirm exactly where those atoms sit in the crystal. Additionally, the calculations use a Coulomb pseudopotential (μ*) of 0.10 — a conventional approximation for the repulsive interaction between electrons — and small deviations from this assumption could shift the predicted Tc meaningfully in either direction.
There's also the question of thermodynamic stability: will MgAlH₆ actually form as a single-phase compound under these conditions, or will it decompose into competing binary phases? Evolutionary algorithm searches help address this, but experimental confirmation remains the ultimate arbiter.
Why This Matters
Room-temperature superconductivity — the kind that could transform power grids, medical imaging, and quantum computing — remains the field's holy grail. MgAlH₆ won't get us there on its own. But what this study demonstrates is something strategically important: that ternary compositional design, mixing two metals with hydrogen, can push the pressure requirements for high-Tc superconductivity into more experimentally tractable territory. Every incremental step down the pressure ladder is a step toward materials that could eventually be synthesized, studied, and perhaps even stabilized at ambient conditions through chemical engineering. The Mg–Al–H ternary phase space explored here is just one corner of a vast compositional landscape. As computational screening tools grow sharper and experimental high-pressure techniques advance, predictions like those for MgAlH₆ serve as precise, testable roadmaps — transforming the search for the ultimate superconductor from an open-ended quest into a methodical, data-driven exploration with real experimental targets in sight.
📊 Simulation Results
Crystal Structure and Bonding
The predicted crystal structure of MgAlH₆ at 97.6 GPa adopts a highly symmetric arrangement that belongs to a cubic or trigonal space group, depending on the pressure regime. At its core, the lattice is built around the AlH₆ octahedral units mentioned earlier, but the way these units connect to one another — and how magnesium atoms thread between them — is what gives the material its remarkable electronic personality.
Each aluminum atom sits at the center of a nearly perfect octahedron formed by six hydrogen atoms. These octahedra are arranged in a three-dimensional network that resembles, in some ways, the perovskite family of materials, but with hydrogen taking the place of oxygen. The Mg atoms occupy the interstitial sites between these octahedra, acting as both electronic donors and structural stabilizers. Without the magnesium, the aluminum-hydrogen framework would collapse or transform into a less favorable phase at these pressures.
Several bonding characteristics make this arrangement exceptionally well-suited for superconductivity:
- Covalent Al–H bonds: The aluminum-hydrogen bonds within each octahedron have significant covalent character, creating directional bonding that locks hydrogen atoms into well-defined positions and produces the sharp, high-frequency phonon modes essential for strong electron-phonon coupling.
- Ionic Mg contribution: Magnesium donates electrons to the AlH₆ framework, populating hydrogen-derived states near the Fermi level and tuning the electronic density of states upward — a prerequisite for robust Cooper pair formation.
- Three-dimensional connectivity: Unlike layered superconductors where currents flow primarily in planes, MgAlH₆'s isotropic 3D framework supports supercurrents in all directions, potentially yielding more uniform critical current behavior.
- Short H–H distances: At 97.6 GPa, neighboring hydrogen atoms across adjacent octahedra are compressed to distances approaching those seen in pure metallic hydrogen, amplifying hydrogen's role in the pairing mechanism.
This combination — covalent H framework, ionic metal donors, and three-dimensional connectivity — is the structural recipe that first-principles calculations identify as the source of MgAlH₆'s predicted 131.4 K transition temperature.
Comparison with Known Superconductors
To appreciate where MgAlH₆ sits in the broader landscape of superconducting materials, it helps to line it up against both hydrogen-rich high-pressure compounds and more conventional superconductors that have defined the field:
- H₃S (hydrogen sulfide): The 2015 breakthrough material, superconducting at ~203 K but requiring ~155 GPa of pressure. MgAlH₆ trades some Tc for a significantly lower pressure requirement — roughly 60% of H₃S's pressure for about 65% of its Tc, a favorable ratio for practical exploration.
- LaH₁₀ (lanthanum superhydride): Superconducts near 250–260 K at ~170 GPa, currently one of the highest-Tc materials known. LaH₁₀'s clathrate cage structure requires extreme pressure to stabilize; MgAlH₆'s octahedral framework is predicted to remain stable at substantially lower pressures thanks to chemical precompression from two metal species.
- MgB₂ (magnesium diboride): The workhorse of practical hydrogen-adjacent superconductivity, with Tc ≈ 39 K at ambient pressure. While MgB₂ is far easier to synthesize and use, MgAlH₆'s predicted Tc is more than three times higher, illustrating the payoff — and the engineering cost — of moving into the high-pressure hydride regime.
- Conventional superconductors (Nb, NbTi, Nb₃Sn): These niobium-based alloys power most of today's MRI magnets and particle accelerators, with Tc values between 9 K and 18 K, requiring liquid helium cooling. MgAlH₆ would, in principle, operate with liquid nitrogen or even simple refrigeration — if its high-pressure requirement could be eliminated or engineered around.
- Cuprate superconductors (YBCO, BSCCO): Ambient-pressure materials with Tc up to 135 K, but based on an entirely different (and still debated) d-wave pairing mechanism. MgAlH₆'s conventional BCS-like electron-phonon mechanism is better understood theoretically and more amenable to predictive design.
What's notable is that MgAlH₆ doesn't set any individual record — but it occupies a sweet spot in the Tc-versus-pressure trade-off that many researchers believe is the realistic path toward eventually engineering an ambient-pressure, high-temperature superconductor.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, must eventually meet the laboratory. For MgAlH₆, that journey involves a sequence of demanding experiments, each designed to test a specific aspect of the theoretical picture.
- Diamond anvil cell synthesis: The first and most immediate experimental step is loading precursor materials — likely magnesium aluminum hydride (Mg(AlH₄)₂) or a mixture of MgH₂ and AlH₃ — into a diamond anvil cell and compressing them to the 90–100 GPa range. Laser heating during compression would then drive the reaction toward the predicted MgAlH₆ phase.
- X-ray diffraction confirmation: Once a sample is synthesized, synchrotron X-ray diffraction can verify whether the resulting crystal structure matches the predicted space group and lattice parameters. Getting this right is essential, because even small deviations in atomic positions can dramatically alter the predicted Tc.
- Electrical resistivity measurements: The gold standard for confirming superconductivity is observing a sudden drop in electrical resistance to zero as temperature decreases. Four-probe resistivity measurements inside a diamond anvil cell, at cryogenic temperatures, would directly reveal whether the predicted 131.4 K transition is real.
- Magnetic susceptibility measurements: The Meissner effect — the expulsion of magnetic fields from a superconductor — provides independent confirmation beyond resistivity alone. This is essential for ruling out alternative explanations for a resistance drop, such as a structural phase transition or contact artifacts.
- Isotope effect studies: Replacing hydrogen with deuterium should, according to BCS theory, shift Tc by a predictable factor of √2. Observing this shift would confirm that hydrogen vibrations are indeed mediating the superconductivity, validating the electron-phonon coupling mechanism at the heart of the theory.
- Inelastic X-ray or neutron scattering: Directly mapping the phonon spectrum of synthesized MgAlH₆ would confirm the predicted 800–1,500 cm⁻¹ hydrogen stretching modes and their coupling strength to the electronic states.
Each of these measurements is technically demanding — diamond anvil cell experiments at 100 GPa involve sample volumes smaller than a grain of sand — but groups in the United States, Germany, China, and Japan have all developed the capabilities needed to attempt them.
Implications for the Field
If MgAlH₆'s predictions hold up under experimental scrutiny, the implications extend well beyond a single new material. The discovery would validate a broader strategy: rational design of ternary and multinary hydrides as a route to more accessible superconductivity.
The concept of chemical precompression has been discussed for years, but concrete examples where it demonstrably reduces the required external pressure are still limited. MgAlH₆ could become a textbook case, proving that combining metals with complementary electronic properties — here, the small, electron-donating magnesium and the covalently bonding aluminum — can shift the superconducting phase to more practical conditions.

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)