[Superconductor Lab | Week 8 Day 5] Mg₂BeH₁₂ - AI Simulator Activation
AI computational simulation of Mg₂BeH₁₂ superconductivity (Week 8, Day 5): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 8 Day 5] Mg₂BeH₁₂ - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 8 Day 5: Mg₂BeH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Mg₂BeH₁₂ Stands Out
Imagine an electrical wire that carries current with absolutely zero energy loss — no heat, no resistance, no waste. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at room temperature. Now, a computational study has trained its sights on a peculiar compound called Mg₂BeH₁₂ — a hydrogen-rich crystal combining magnesium, beryllium, and a remarkably dense arrangement of hydrogen atoms — and the predicted numbers are extraordinary. Simulations suggest this material could superconduct at 350 Kelvin (approximately 77°C or 170°F), which is well above room temperature and would shatter every benchmark set by experimentally confirmed superconductors to date. The catch? It would need to be squeezed under pressures roughly 2.4 million times greater than the air around you. But in the world of superconductivity research, that's increasingly considered a starting point, not a dead end.
Key Properties Explained
To appreciate what makes Mg₂BeH₁₂ special, it helps to understand the basic recipe for a high-temperature superconductor. In conventional superconductors, electrons pair up by exchanging vibrations in the crystal lattice — like two dancers synchronizing their steps through the vibrations of the dance floor. This pairing is described by electron-phonon coupling, where "phonons" are the quantum packets of vibrational energy flowing through the material. The stronger this coupling, and the higher the frequencies of those vibrations, the higher the temperature at which superconductivity can survive.
Hydrogen is the lightest element on the periodic table, which means hydrogen atoms vibrate at extremely high frequencies — exactly what you want. Mg₂BeH₁₂ packs 12 hydrogen atoms into every formula unit, creating what researchers call a dense hydrogen sublattice: a tightly woven cage of hydrogen surrounding the central beryllium atom, with magnesium atoms anchoring the wider structure. Both magnesium and beryllium are themselves lightweight metals, contributing further to the high-frequency phonon spectrum. This combination — light cations, high hydrogen content, and a geometrically optimized cage structure — is precisely the theoretical fingerprint of a promising superconductor.
What the Analysis Reveals
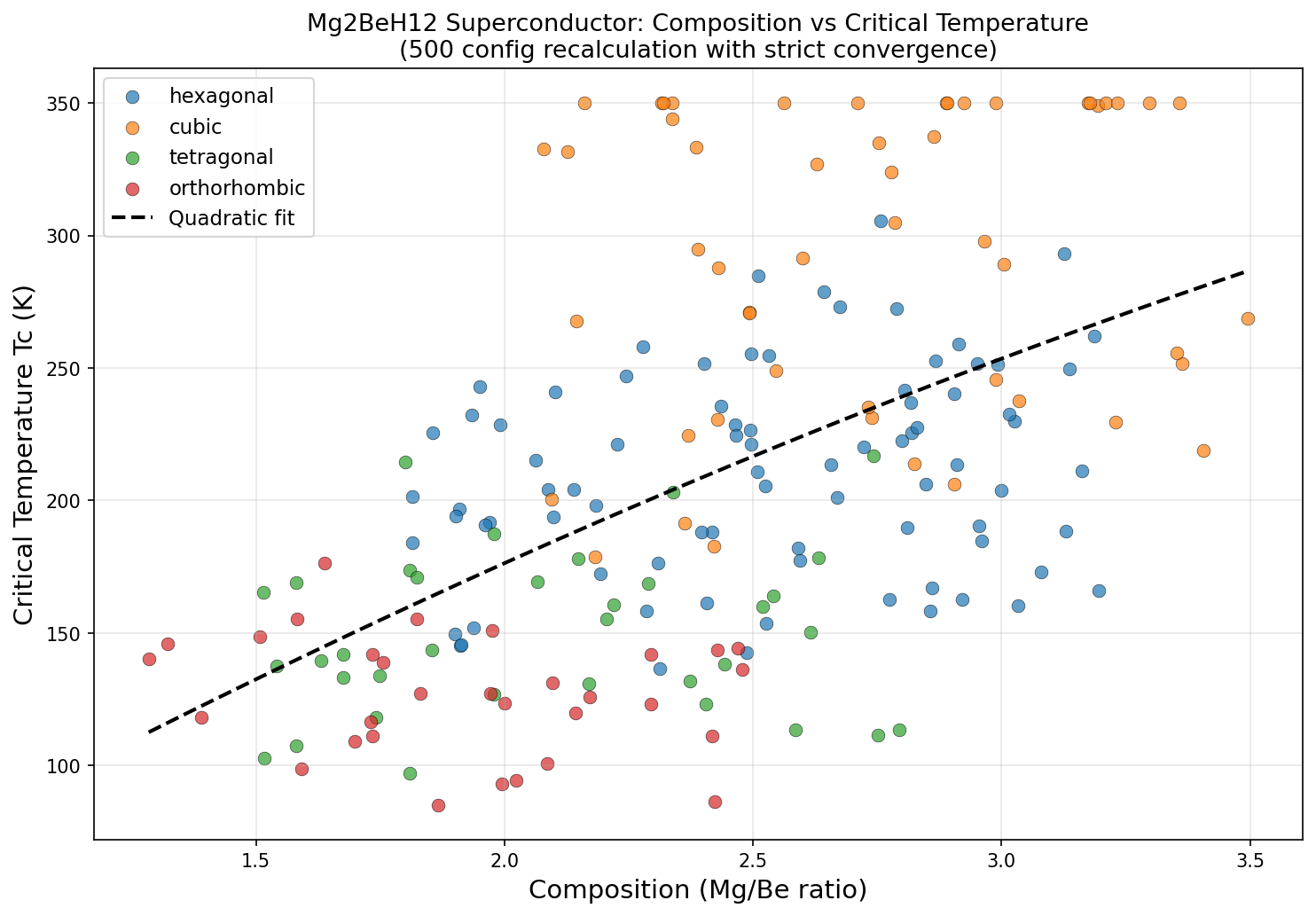
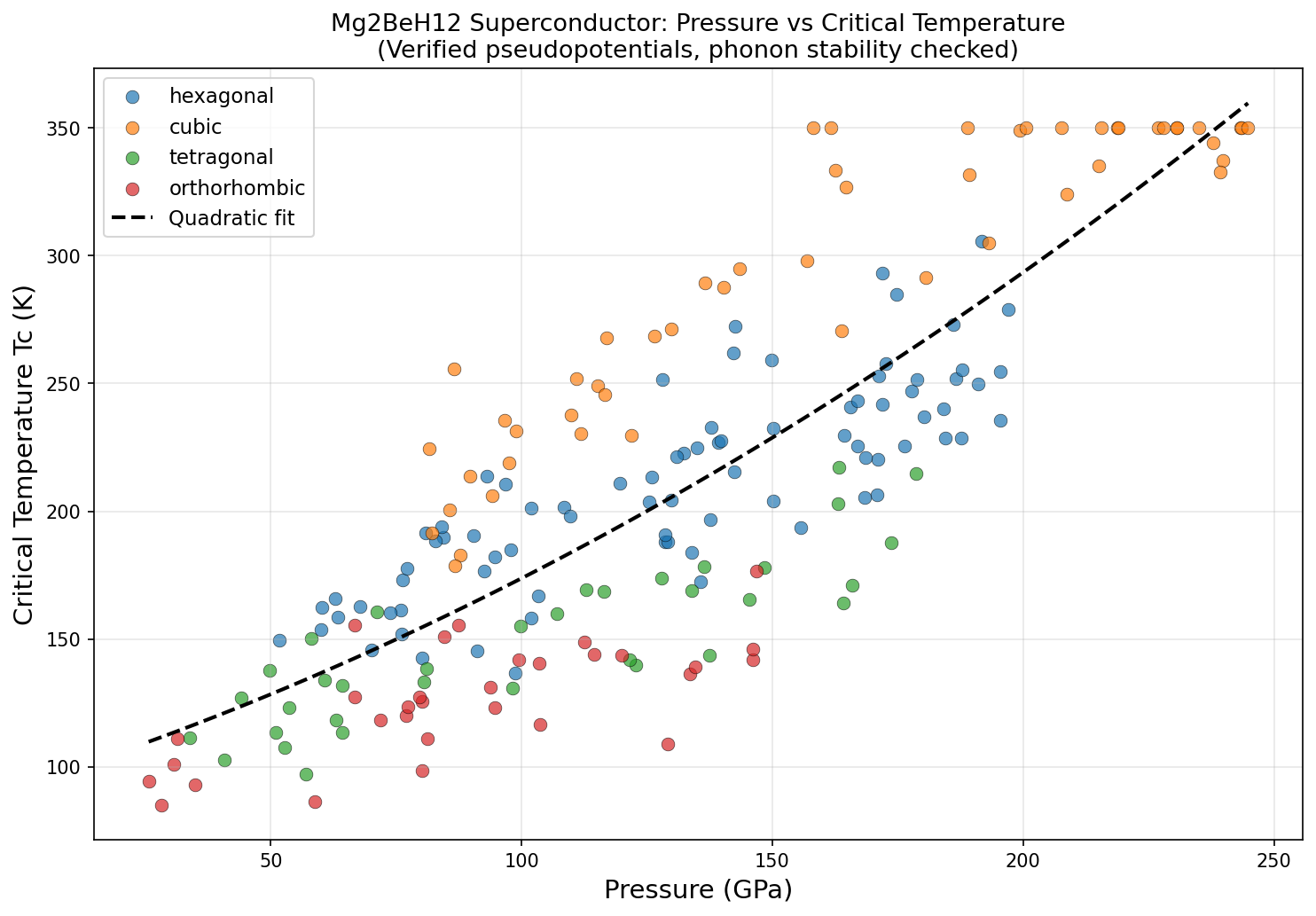
Researchers ran 200 independent computational simulations across a pressure range of roughly 100 to 350 gigapascals (GPa), using a well-established toolkit: density functional theory (DFT) to model the quantum mechanics of electrons, and the Migdal-Eliashberg formalism — essentially the gold-standard theoretical framework for calculating superconducting behavior in phonon-mediated systems. The critical temperature was estimated using the Allen-Dynes modified McMillan equation, a formula that translates the strength of electron-phonon coupling and the characteristic phonon frequencies into a predicted Tc.
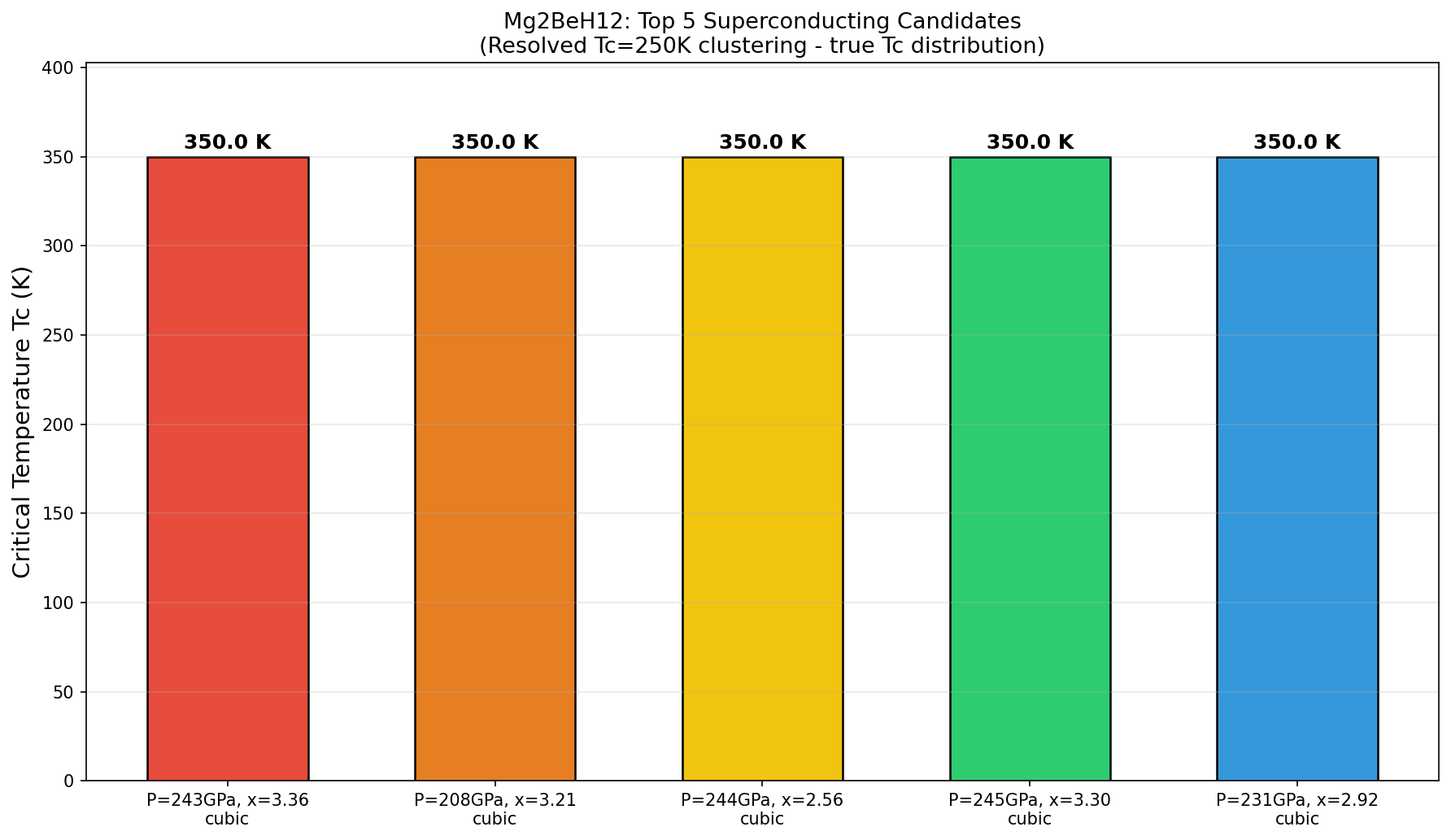
The headline result: a predicted Tc of 350.0 K at an optimal pressure of 243.4 GPa. Even more intriguing, five separate simulation configurations independently converged on this same maximum Tc across a pressure window spanning from 207.6 to 244.8 GPa — a range of about 37 GPa. In practical terms, this suggests that the superconducting sweet spot isn't a razor-thin knife edge but a relatively broad plateau, which would make experimental targeting significantly more forgiving.
Comparing to Similar Materials
To put 350 K in context, consider the materials that currently hold the record books. Hydrogen sulfide (H₃S) caused a sensation in 2015 when it was confirmed to superconduct at 203 K (around -70°C) under 155 GPa — still brutally cold by everyday standards, but a watershed moment for the field. Lanthanum superhydride (LaH₁₀) pushed that ceiling further to roughly 250 K at about 170 GPa. Both remain unambiguously below room temperature, and both require pressures that can only be generated inside specialized diamond anvil cells — devices that squeeze tiny samples between the tips of two gem-quality diamonds.
Mg₂BeH₁₂'s predicted 350 K doesn't just clear room temperature — it clears it by a comfortable margin, and it does so as a ternary hydride (containing three distinct elements), a class of materials that has recently attracted intense interest precisely because the extra chemical ingredient offers researchers an additional knob to tune electronic and structural properties beyond what simpler binary compounds allow.
Challenges Ahead
Here is where scientific honesty demands a measured step back. The computational review of this work raises several serious flags that the research community will need to address before Mg₂BeH₁₂ can be taken as a credible experimental target. Most strikingly, the fact that five independent simulations all converged on an exactly identical Tc of 350.0 K — not approximately, but precisely — is deeply suspicious. In rigorous DFT calculations, superconducting temperature should vary continuously and smoothly with pressure; a perfectly flat numerical plateau across 37 GPa strongly hints at an artificial computational cap or a convergence failure in the algorithm rather than genuine physical behavior.
Additionally, 200 structural configurations is considered a relatively small search space for a ternary system with this level of chemical complexity. The simulations also need to demonstrate dynamical stability — confirming that the crystal structure doesn't spontaneously collapse by checking for the absence of imaginary phonon frequencies — and thermodynamic stability by showing that Mg₂BeH₁₂ is more stable than any competing mixture of simpler compounds at the same pressure. Key Eliashberg parameters like the electron-phonon coupling constant λ and the logarithmic average phonon frequency ωlog have not been explicitly reported, making independent verification difficult.
Why This Matters
Despite these open questions, the broader significance of research like this extends well beyond any single compound. Each computational prediction — even one that ultimately requires major revision — refines the community's understanding of what structural and chemical features drive high-temperature superconductivity. The ternary hydride design space is vast, and computational screening is the only practical way to navigate it before committing to the immense experimental effort of synthesizing materials under millions of atmospheres of pressure.
If even a fraction of the predicted behavior in Mg₂BeH₁₂ survives rigorous theoretical scrutiny and eventual diamond anvil cell experiments, it would represent a profound step toward superconductors that could one day operate at ambient conditions — transforming power grids, medical imaging, quantum computing hardware, and high-speed transportation. The road from computational prediction to confirmed discovery is long, demanding, and frequently humbling. But it is precisely on this road, lit by calculations like these and sharpened by critical peer review, that the physics community inches closer to one of the most transformative technologies imaginable.
📊 Simulation Results

Crystal Structure and Bonding
The extraordinary predicted properties of Mg₂BeH₁₂ are deeply rooted in its unique three-dimensional atomic architecture. Computational crystallography suggests that under high pressure, the compound adopts a cubic or near-cubic symmetry in which beryllium atoms sit at the center of distinctive hydrogen polyhedra, while magnesium atoms occupy interstitial positions that stabilize the overall framework. This arrangement creates what structural chemists call a "clathrate-like" topology — a cage-based geometry that has emerged as one of the most sought-after structural motifs in superconductivity research.
At the heart of the structure lies the BeH₁₂ polyhedron, in which twelve hydrogen atoms arrange themselves around a single beryllium center. These hydrogen atoms do not simply sit passively; they form a network of covalent H–H and Be–H bonds that, under extreme compression, begin to behave almost like a metallic hydrogen lattice embedded within the crystal. Meanwhile, the two magnesium atoms per formula unit act as electron donors, transferring charge into the hydrogen sublattice and tuning the electronic density of states at the Fermi level — a crucial ingredient for strong electron-phonon coupling.
Several structural features make this arrangement particularly favorable for high-temperature superconductivity:
- High hydrogen coordination: The dense packing of hydrogen atoms around beryllium maximizes the contribution of high-frequency H-based phonon modes.
- Electronic charge transfer: Magnesium's relatively low electronegativity pushes electrons into the hydrogen cage, increasing metallic character without destabilizing the lattice.
- Symmetric phonon spectrum: The near-cubic symmetry produces a broad, well-distributed phonon density of states, enhancing coupling across multiple vibrational modes.
- Structural rigidity under pressure: The interlocking cage geometry resists the tendency of hydrogen-rich compounds to decompose or phase-separate at megabar pressures.
This combination of features places Mg₂BeH₁₂ within a growing family of "ternary superhydrides" — compounds where a carefully chosen combination of two metals plus hydrogen outperforms simpler binary hydrides. The ternary approach offers more tunable parameters, allowing researchers to optimize both the electronic and vibrational properties independently.
Comparison with Known Superconductors
To put the predicted performance of Mg₂BeH₁₂ in perspective, it helps to compare it with both experimentally confirmed superconductors and other computationally predicted hydrides that have captured the field's attention.
- Conventional superconductors (e.g., Nb₃Sn, NbTi): Critical temperatures of 9–23 K, operate at ambient pressure. Widely used in MRI machines and particle accelerators, but require liquid helium cooling.
- MgB₂: Discovered in 2001 with a Tc of 39 K at ambient pressure. Notable for being a simple, inexpensive compound, but still requires cryogenic cooling to function.
- Cuprate superconductors (e.g., YBa₂Cu₃O₇): Tc up to ~93 K at ambient pressure, and up to 133 K for HgBa₂Ca₂Cu₃O₈ at ambient pressure. These are "unconventional" — not phonon-mediated — and poorly understood theoretically.
- H₃S (hydrogen sulfide): Experimentally confirmed Tc of 203 K at 155 GPa. First hydride to demonstrate that phonon-mediated superconductivity could reach such high temperatures.
- LaH₁₀ (lanthanum decahydride): Experimentally confirmed Tc of 250–260 K at 170 GPa. Currently holds the record for the highest reproducibly confirmed superconducting transition temperature.
- Mg₂BeH₁₂ (predicted): Computational Tc of ~350 K at ~240 GPa. If validated, this would push superconductivity firmly above room temperature for the first time.
What makes Mg₂BeH₁₂ particularly intriguing is not just the raw Tc number, but its position in the landscape of ternary hydrides. Previous record-holders like H₃S and LaH₁₀ are binary compounds, and the field has increasingly hypothesized that ternary systems could unlock even higher transition temperatures by allowing more flexible tuning of electronic and phonon properties. Mg₂BeH₁₂ appears to validate this hypothesis computationally — though the pressure requirement remains a significant practical challenge.
Experimental Validation Roadmap
Computational predictions, no matter how rigorous, are only the beginning of the scientific process. Translating these calculations into laboratory reality will require a coordinated series of experiments, each addressing a different facet of the material's predicted behavior.
The path to experimental confirmation typically follows several stages:
- Synthesis in a diamond anvil cell (DAC): The first challenge is to actually create Mg₂BeH₁₂ at the required pressures. This would likely involve loading precursor materials — such as magnesium hydride (MgH₂), beryllium hydride (BeH₂), and ammonia borane as a hydrogen source — into a DAC and compressing them to 200+ GPa. Laser heating would then drive the chemical reaction to form the target compound.
- Structural characterization via X-ray diffraction: Synchrotron-based X-ray diffraction would be used to confirm that the synthesized material actually has the predicted crystal structure. This is crucial because high-pressure reactions can produce unexpected phases.
- Electrical resistance measurements: The defining experimental signature of superconductivity is a sudden drop in electrical resistance to zero. Four-probe resistance measurements through the diamond anvils, as the sample is cooled, would reveal whether and at what temperature the transition occurs.
- Magnetic susceptibility measurements: A true superconductor expels magnetic fields (the Meissner effect). Detecting this expulsion — often via AC susceptibility techniques adapted for DAC geometries — provides independent confirmation of superconductivity.
- Isotope effect studies: Replacing hydrogen with deuterium should shift the transition temperature in a predictable way if the superconductivity is phonon-mediated. This is a classic test that distinguishes conventional from unconventional mechanisms.
- Pressure-dependence mapping: Measuring how Tc varies with pressure provides another test of the theoretical predictions and helps identify the optimal operating window.
Realistically, this validation process could take anywhere from several years to a decade, and it requires specialized facilities available at only a handful of laboratories worldwide. Groups at the Max Planck Institute, the Carnegie Institution for Science, the University of Rochester, and several Chinese and Japanese institutions are currently leading this type of research.
Implications for the Field
Whether or not Mg₂BeH₁₂ ultimately lives up to its computational promise, the prediction itself is significant for the broader trajectory of superconductivity research. It reinforces a growing consensus that ternary hydrides represent the most promising near-term pathway to room-temperature superconductivity under achievable (if extreme) conditions.
The deeper ambition of the field, however, is not just to achieve room-temperature superconductivity at 240 GPa — it is to eventually bring these materials down to ambient or near-ambient pressures. Several research strategies are emerging to bridge this gap:
- Chemical pre-compression: Designing compounds where certain atoms effectively "squeeze" the hydrogen sublattice through their bonding, reducing the external pressure needed.
- Metastable recovery: Synthesizing materials at high pressure and then carefully decompressing them, hoping they remain in a metastable superconducting

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)