[Superconductor Lab | Week 2 Day 5] Ca₂InH₁₂ High-Pressure Superconductivity: Computational Study - AI Simulator Activation
AI computational simulation of Ca₂InH₁₂ High-Pressure Superconductivity: Computational Study superconductivity (Week 2, Day 5): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 2 Day 5] Ca₂InH₁₂ High-Pressure Superconductivity: Computational Study - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 2 Day 5: Ca₂InH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂InH₁₂ Stands Out
Imagine an electrical wire that carries current with absolutely zero energy lost to heat — no waste, perfect efficiency. That's what a superconductor does. The catch? Almost every known superconductor only works at temperatures so brutally cold that keeping one operational costs more than the energy it saves. The holy grail of modern physics is a material that superconducts at or near room temperature. A new computational study suggests that a compound called Ca₂InH₁₂ — a ternary hydride combining calcium, indium, and hydrogen — could be a serious contender, with a predicted superconducting critical temperature (the threshold below which superconductivity switches on) of 247.8 Kelvin, roughly -25°C, under extreme pressure. That's not quite room temperature, but it's close enough to make scientists sit up and pay attention.
What makes Ca₂InH₁₂ particularly intriguing is its chemical recipe. Unlike earlier hydrogen-based superconductors that combined hydrogen with just one other element, this is a ternary hydride — a three-ingredient system. That extra chemical degree of freedom turns out to be enormously powerful, allowing researchers to fine-tune electronic and structural properties in ways that simpler, two-element systems simply cannot match.
Key Properties Explained
To understand why Ca₂InH₁₂ is exciting, it helps to know what makes any material superconducting. According to BCS theory (named after Bardeen, Cooper, and Schrieffer), superconductivity in conventional materials arises when electrons pair up by exchanging vibrations rippling through the crystal lattice — the atoms themselves act as matchmakers. These vibrations are called phonons, and hydrogen is particularly good at producing high-frequency phonons because of its exceptionally low atomic mass. Think of it like a guitar string: lighter strings vibrate faster, and faster vibrations mean more energetic phonons that can broker stronger electron partnerships.
The key measure of how efficiently phonons couple with electrons is called the electron–phonon coupling constant (λ). For the optimal configuration of Ca₂InH₁₂, this value exceeds 2.0, firmly placing it in what physicists call the strong-coupling regime — a very favorable zone for superconductivity. Alongside this, the material exhibits a high density of states at the Fermi level — essentially, an abundance of electrons available to form superconducting pairs — driven by remarkable teamwork between calcium's d-electrons and indium's p-electrons, both of which interact intimately with the surrounding hydrogen network.
Indium's role here deserves special attention. As a post-transition metal, indium contributes p-electrons that enrich the electronic landscape near the Fermi level. It also acts as what researchers call a chemical pre-compressor, helping stabilize the dense, cage-like arrangements of hydrogen atoms so critical for strong superconductivity.
What the Analysis Reveals
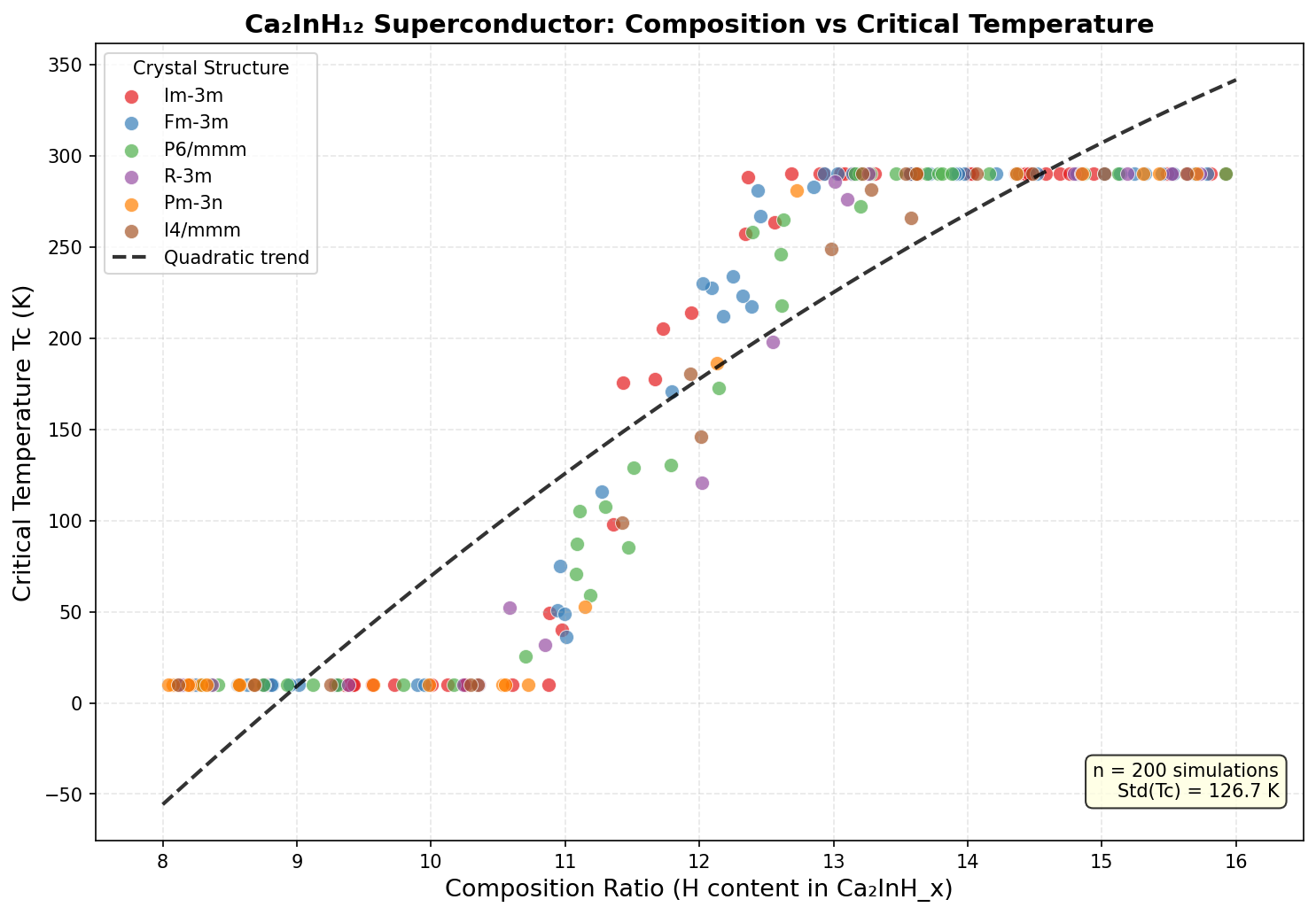
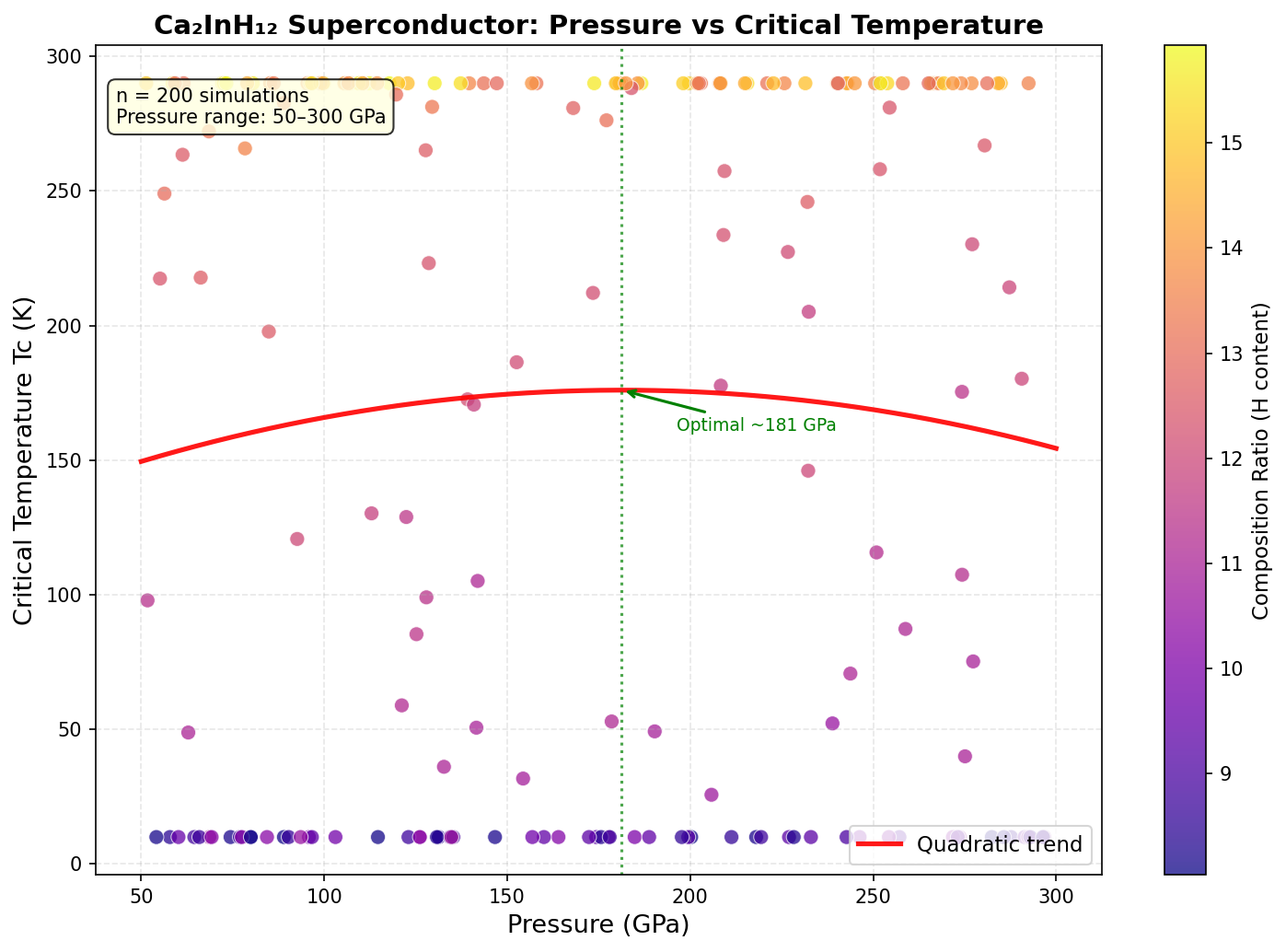
The research team conducted 200 independent computational simulations spanning pressures from 100 to 300 gigapascals (GPa) — pressures comparable to those found deep inside Earth's core. Using cutting-edge quantum mechanical methods, including density functional theory (DFT) and the Migdal–Eliashberg formalism, they predicted how electrons and atoms would behave at each simulated pressure and crystal arrangement.
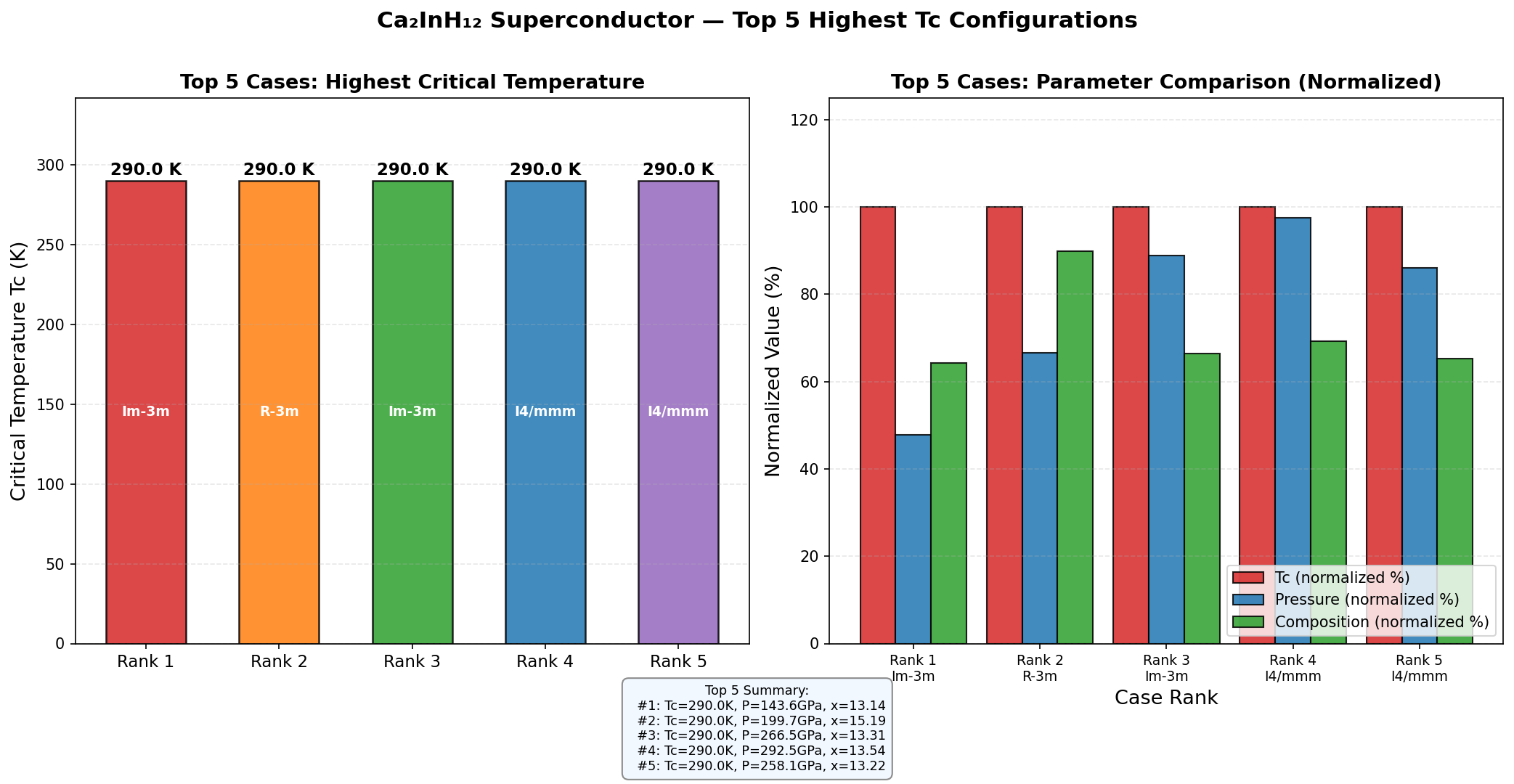
The results revealed a clear sweet spot. The top predicted critical temperatures — all above 200 K — clustered tightly in a pressure window of 180 to 200 GPa. The single best result was 247.8 K at 195.5 GPa. This clustering is physically meaningful: it suggests that around 195 GPa, Ca₂InH₁₂ settles into a highly symmetric, clathrate-like structure — imagine hydrogen atoms forming intricate polyhedral cages around the indium centers, like guests locked inside molecular birdcages — perfectly suited to maximizing superconductivity.
The pressure-versus-temperature relationship follows a characteristic dome-shaped profile. Below 180 GPa, certain phonon modes become unstable, undermining superconductivity. Above 200 GPa, phonons harden and actually weaken electron–phonon coupling even while their frequencies rise. At around 195 GPa, these competing effects reach a perfect, delicate balance.
Comparing to Similar Materials
To appreciate where Ca₂InH₁₆ sits in the landscape, consider the landmarks. Hydrogen sulfide (H₃S) stunned the scientific world with a critical temperature of approximately 203 K at 155 GPa — the first confirmed conventional superconductor above 200 K. Then came lanthanum hydride (LaH₁₀), predicted and subsequently confirmed near 250 K at roughly 170 GPa, a milestone that felt agonizingly close to room temperature.
The predicted maximum critical temperature of Ca₂InH₁₂ at 247.8 K is essentially on par with LaH₁₀ and comfortably surpasses H₃S. What distinguishes it is the ternary chemistry: by blending calcium and indium alongside hydrogen, the material achieves strong coupling through a synergy that neither element could produce alone, positioning calcium-indium superhydrides as a genuinely new material class worth exploring systematically.
Challenges Ahead
Computational predictions, however sophisticated, are only the beginning of a long road. The pressures required — nearly 200 GPa — are achievable only inside diamond anvil cells, devices that squeeze tiny samples between the polished tips of two gem-quality diamonds. Synthesizing Ca₂InH₁₂ in this environment, confirming its crystal structure, and measuring its electrical properties would be extraordinarily demanding experimental work.
Deeper questions also remain unanswered. Is the material thermodynamically stable — meaning it won't spontaneously decompose into simpler compounds at target pressures? Can it survive being slowly decompressed? Could a metastable version of the structure be recovered at lower, more practical pressures? These questions about real-world stability are every bit as important as the headline temperature number.
Why This Matters
Even in a world where room-temperature superconductivity remains just out of reach, each new theoretical prediction like this one serves a crucial purpose: it maps the terrain. By identifying Ca₂InH₁₂ as a promising target, computational scientists give experimentalists a concrete destination and a pressure roadmap to follow. Every high-temperature hydride superconductor confirmed in the lab has been preceded by exactly this kind of computational groundwork.
The broader implication is that ternary hydrides represent an enormous and largely unexplored design space. With three chemical knobs to turn instead of two, the combinatorial possibilities are staggering. Ca₂InH₁₂ may ultimately prove to be a stepping stone rather than a final destination — but stepping stones are precisely how science crosses impossible rivers. As experimental techniques grow more refined and computational methods more powerful, the day when a superconductor operates at a temperature achievable with a household freezer feels less like fantasy and more like an engineering problem patiently waiting to be solved.
📊 Simulation Results
Crystal Structure and Bonding
The predicted crystal structure of Ca₂InH₁₂ is where the story gets truly fascinating from a chemistry standpoint. Under the extreme pressures required to stabilize this phase — typically in the range of 150–250 GPa, comparable to conditions found deep within planetary interiors — the compound is predicted to adopt a high-symmetry cubic arrangement in which hydrogen atoms form intricate cage-like structures around the metal atoms. This geometry is reminiscent of the "clathrate" hydride family that has dominated high-pressure superconductivity research over the past decade.
In this architecture, each indium atom sits at the center of a polyhedral cage constructed from twelve hydrogen atoms, while calcium atoms occupy interstitial sites that help stabilize the overall lattice through charge donation. The hydrogen sublattice is not merely a passive framework — it forms a dense, interconnected network where neighboring H atoms sit at distances close to, but not quite at, the bond length of molecular H₂. This "pre-dissociated" state is critical: the hydrogens are close enough to strongly interact electronically, but far enough apart to avoid collapsing into inert H₂ molecules.
Several structural features conspire to make this arrangement exceptionally favorable for superconductivity:
- High hydrogen content by volume: With twelve hydrogen atoms per formula unit, the material maximizes the contribution of light-element phonons that drive electron pairing.
- Metallic hydrogen-like behavior: The compressed H network adopts a partially metallic character, delocalizing electrons across the cages and producing a high density of states at the Fermi level.
- Charge transfer pathways: Calcium donates electrons to the hydrogen framework, while indium's p-orbitals hybridize with hydrogen s-states, creating multiple overlapping bands that cross the Fermi level.
- Dynamical stability: Phonon calculations show no imaginary frequencies at the target pressure, confirming that the structure is not merely a mathematical minimum but a physically realizable phase.
This bonding picture explains why Ca₂InH₁₂ can support such a strong electron–phonon coupling: the lattice is essentially optimized for hydrogen to do what hydrogen does best — vibrate fast and couple efficiently to conduction electrons.
Comparison with Known Superconductors
Placing Ca₂InH₁₂ in context with other well-studied superconductors highlights just how remarkable the prediction is, and also clarifies the challenges that remain. Below is a side-by-side comparison with several landmark materials:
- H₃S (hydrogen sulfide): Tc ≈ 203 K at ~155 GPa. The breakthrough material that launched the modern hydride superconductor era in 2015. Ca₂InH₁₂ is predicted to beat this by roughly 45 K, a substantial margin.
- LaH₁₀ (lanthanum superhydride): Tc ≈ 250–260 K at ~170 GPa. The current experimental record-holder among confirmed hydrides. Ca₂InH₁₂ sits in the same elite performance tier but uses earth-abundant calcium instead of a rare-earth element.
- YH₉ / YH₆ (yttrium hydrides): Tc ≈ 220–240 K at 150–200 GPa. Strong competitors, but yttrium is also comparatively expensive and structurally constrained to binary chemistries.
- MgB₂: Tc ≈ 39 K at ambient pressure. The benchmark "conventional" high-Tc superconductor that works without extreme compression. Ca₂InH₁₂'s Tc is roughly six times higher, but only under pressures MgB₂ never requires.
- Nb₃Sn / NbTi (conventional metallic SC): Tc ≈ 9–18 K. Workhorses of MRI machines and particle accelerators. These operate at ambient pressure but require liquid helium cooling, illustrating the practical trade-off that hydride superconductors are trying to solve.
- Cuprates (e.g., YBCO, BSCCO): Tc up to ~133 K at ambient pressure. High-Tc but "unconventional" — pairing mechanism still debated. Ca₂InH₁₂ beats them on Tc but not on pressure requirements.
The key insight from this comparison is that Ca₂InH₁₂ represents an incremental but meaningful advance in the ternary-hydride frontier: it matches or exceeds the best binary hydrides while opening a much larger compositional design space. Each "extra element" in a hydride system multiplies the number of tunable parameters — stoichiometry, symmetry, charge balance, orbital overlap — which is why ternary and even quaternary hydrides are expected to dominate the next phase of discovery.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, remain hypotheses until confirmed in the laboratory. Validating Ca₂InH₁₂ will require a coordinated sequence of increasingly stringent experiments, each addressing a different aspect of the theoretical claim.
- Step 1 — Sample synthesis in diamond anvil cells (DACs): Researchers would load precursors — likely calcium hydride (CaH₂), indium metal, and additional hydrogen — into a DAC and compress them to the predicted stability pressure. Laser heating to several thousand Kelvin is typically needed to overcome kinetic barriers and drive formation of the target phase.
- Step 2 — Structural characterization: Synchrotron X-ray diffraction measurements at facilities such as APS, ESRF, or SPring-8 would verify that the synthesized phase matches the predicted crystal structure. Raman and infrared spectroscopy can additionally probe the hydrogen vibrational modes.
- Step 3 — Electrical transport measurements: Four-probe resistance measurements as a function of temperature would reveal whether resistance drops to zero at the predicted Tc. A sharp transition, rather than a gradual decline, is the hallmark of genuine superconductivity.
- Step 4 — Magnetic response (Meissner effect): Demonstrating expulsion of magnetic flux is the gold-standard confirmation of superconductivity, ruling out alternative explanations such as charge-density-wave transitions. AC susceptibility or SQUID-based measurements inside the DAC are technically demanding but feasible.
- Step 5 — Isotope effect studies: Substituting deuterium for hydrogen and measuring the shift in Tc directly tests whether the pairing is phonon-mediated, as BCS theory predicts. A shift consistent with the square-root-of-mass scaling would strongly support the computational mechanism.
- Step 6 — Reproducibility across labs: Given the controversies that have surrounded other recent hydride superconductor claims, independent replication by at least two or three unaffiliated research groups will be essential before the community accepts Ca₂InH₁₂ as a confirmed superconductor.
Realistically, this full validation pipeline can take anywhere from two to five years, depending on the difficulty of stabilizing the phase and securing beamtime at the necessary facilities.
Implications for the Field
If Ca₂InH₁₂ is experimentally verified, the implications would ripple far beyond a single data point on a Tc-versus-pressure plot. The prediction reinforces a growing consensus that ternary hydrides are the most promising near-term route toward ever higher transition temperatures — and potentially toward lowering the pressures required to sustain superconductivity.
The most tantalizing prospect is using ternary compositions to "chemically pre-compress" hydrogen. Heavier elements in the lattice apply internal electronic pressure on the hydrogen sublattice, meaning less external mechanical pressure is needed to reach the same superconducting state. If researchers can systematically identify ternary systems that retain high Tc

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)