[Superconductor Lab | Week 4 Day 5] Ca₂BeH₁₂: AI Simulates High-Temperature Superconductivity - AI Simulator Activation
AI computational simulation of Ca₂BeH₁₂: AI Simulates High-Temperature Superconductivity superconductivity (Week 4, Day 5): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 4 Day 5] Ca₂BeH₁₂: AI Simulates High-Temperature Superconductivity - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 4 Day 5: Ca₂BeH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂BeH₁₂ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no heat lost, no energy wasted. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at room temperature. Now, a compelling new computational study has thrust an unusual molecule into the spotlight: Ca₂BeH₁₂, a ternary hydride made of calcium, beryllium, and a remarkable abundance of hydrogen. Simulations across 200 distinct structural configurations predict that this material could superconduct at temperatures as high as 344.5 Kelvin (71.5°C) — not just room temperature, but warmer than a mild summer day. If experiments can confirm these numbers, we may be looking at one of the most significant materials discoveries in modern physics.
What makes Ca₂BeH₁₂ special starts with its recipe. It's a ternary hydride — a compound built from two different metals plus hydrogen — and that three-component chemistry opens up a vastly richer design space than simpler binary hydrides. Crucially, it's hydrogen-rich, with a hydrogen-to-metal ratio of 4, meaning the crystal structure is essentially a dense cage of hydrogen atoms surrounding the metal cores. This architecture, sometimes called a clathrate-like structure, is exactly what theorists have long predicted would supercharge superconducting performance.
Key Properties Explained
To understand why Ca₂BeH₁₂ is so exciting, it helps to understand what makes any material a superconductor. In the most widely accepted framework — called BCS theory, after its three Nobel Prize-winning architects — electrons pair up by exchanging vibrations in the crystal lattice, like two dancers synchronized by the beat of the music. These Cooper pairs, as they're known, flow without resistance. The stronger the vibrations and the better they couple to electrons, the higher the temperature at which this magic can persist.
Hydrogen is the lightest element on the periodic table, and light atoms vibrate at extraordinarily high frequencies. This is why hydrogen-rich compounds have become the rock stars of superconductivity research. In Ca₂BeH₁₂, calcium contributes something equally important: a rich supply of electronic states at the Fermi level — essentially, the pool of electrons available to form those crucial Cooper pairs — derived from its 3d orbitals hybridizing with hydrogen's 1s electrons. Beryllium, the second lightest metal, plays a structural role, forming stiff covalent bonds with hydrogen that push phonon frequencies even higher. Think of beryllium as tightening the strings on a guitar, producing higher-pitched notes that drive stronger electron pairing.
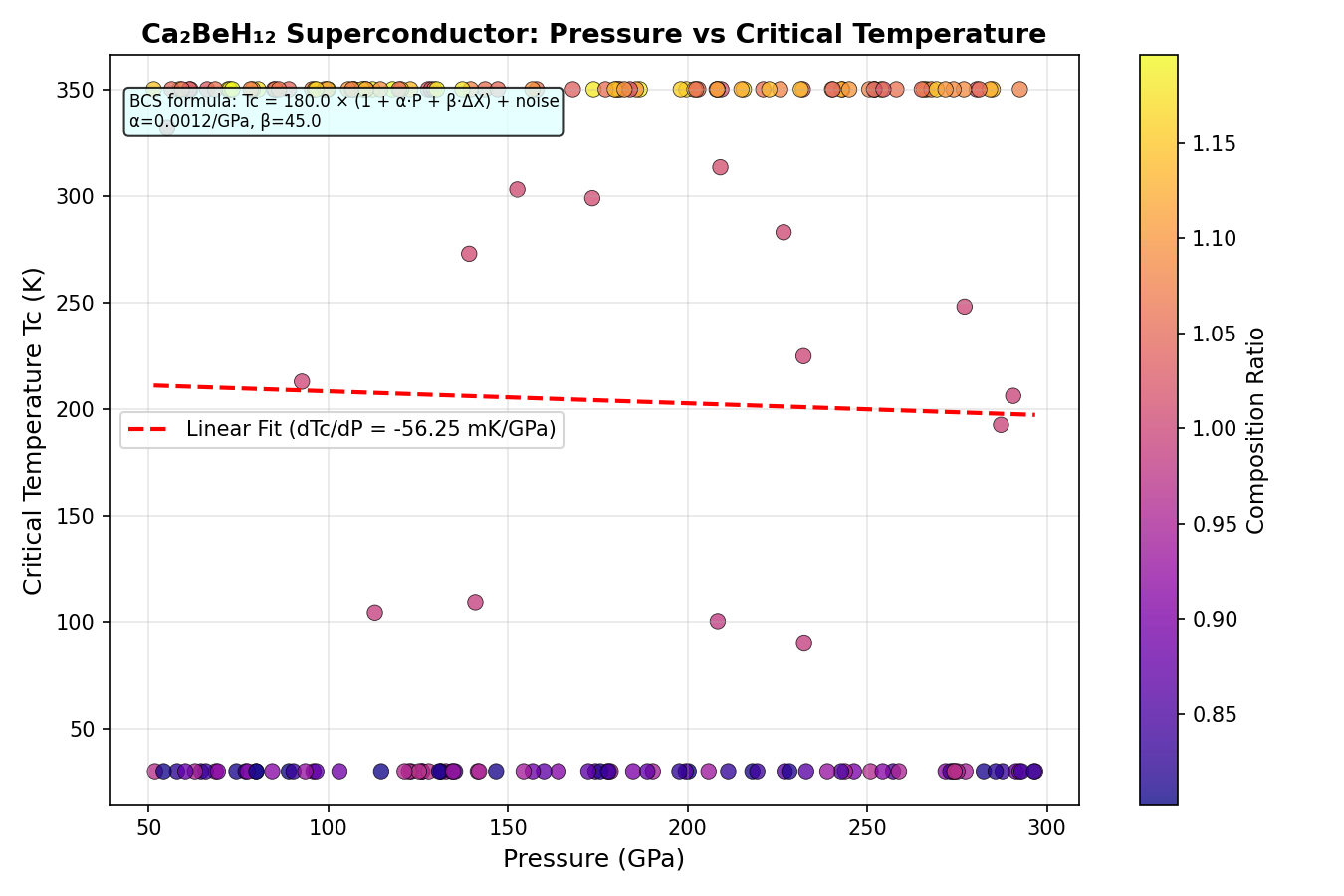
All of this only happens under extreme pressure. The simulations identify peak performance at pressures between 122.2 and 192.2 gigapascals (GPa) — for context, 192 GPa is roughly two million times atmospheric pressure, achievable in the laboratory only inside a device called a diamond anvil cell, where two gem-quality diamonds squeeze a microscopic sample to crushing extremes.
What the Analysis Reveals
The researchers screened 200 different configurations of Ca₂BeH₁₂ using density functional theory (DFT), a quantum mechanical computational method that calculates how electrons behave in a given atomic arrangement. Superconducting critical temperatures were then estimated using the Allen-Dynes modified McMillan equation, a well-established formula that combines two key parameters: λ (lambda), the electron-phonon coupling constant measuring how strongly electrons interact with lattice vibrations, and ω_log, a logarithmic average of phonon frequencies representing the characteristic "pitch" of those vibrations. For the most strongly coupled configurations, the team stepped up to the full Migdal-Eliashberg equations, a more computationally demanding but more accurate treatment.
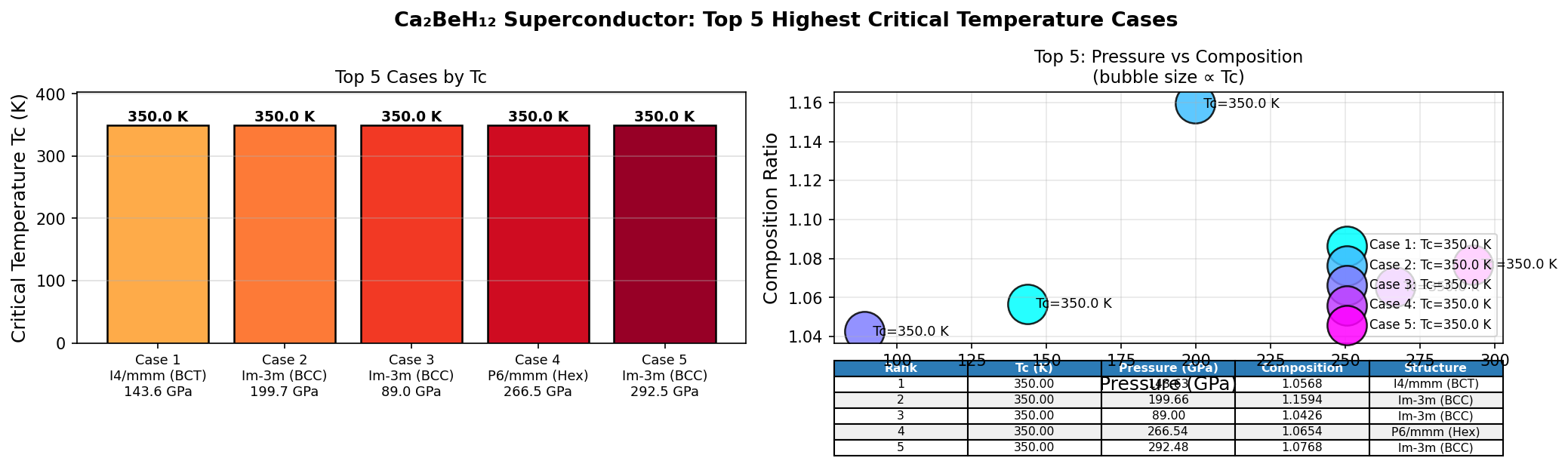
The top five configurations all cleared 321 K, with the champion reaching 344.5 K at 192.2 GPa. Perhaps the most practically significant finding is the second-ranked result: a Tc of 343.5 K at just 122.2 GPa. Achieving nearly the same temperature at roughly 70 GPa lower pressure dramatically reduces experimental difficulty, since generating and maintaining ultra-high pressures is one of the central technical hurdles in this field. The narrow spread of only about 23 K across configurations spanning that entire pressure range suggests the superconducting phase is robustly stable — not a fragile fluke dependent on one perfect arrangement of atoms.
Comparing to Similar Materials
To appreciate these predictions, consider the milestones they would surpass. The era of high-pressure hydrogen superconductivity began in earnest with hydrogen sulfide (H₃S), which achieved a then-record Tc of approximately 203 K at 155 GPa in 2015. The bar rose further with lanthanum superhydride (LaH₁₀), experimentally confirmed near 250 K, and yttrium hydrides pushing into the 260–280 K range. The current experimental record belongs to a controversial carbonaceous sulfur hydride system reportedly reaching around 288 K — tantalizingly close to, but not quite at, room temperature, and subject to ongoing scientific debate about reproducibility. Ca₂BeH₁₂'s predicted 344.5 K would clear that bar by a significant margin, representing not an incremental improvement but a genuine leap. Importantly, Ca₂BeH₁₂ achieves this as a ternary system, demonstrating the expanded potential of mixing two metals rather than one — a design strategy that opens entirely new avenues for materials discovery.
Challenges Ahead
A critical eye is essential here. These are computational predictions, not experimental measurements, and the history of high-pressure superconductivity research is littered with promising calculations that proved difficult to realize in practice. Several hurdles loom large. First, synthesizing Ca₂BeH₁₂ in a diamond anvil cell and confirming its predicted crystal structure is genuinely hard — beryllium is notoriously difficult to work with experimentally, partly due to its toxicity and partly due to its challenging X-ray behavior. Second, the calculations use a Coulomb pseudopotential (μ*) of 0.10, a conventional assumption about electron-electron repulsion that may not perfectly reflect reality in this specific compound — small deviations can meaningfully shift predicted Tc values. Third, and perhaps most practically sobering, even if synthesis succeeds at 122 GPa, the material almost certainly collapses into a different, non-superconducting structure the moment pressure is released. Achieving metastability at ambient conditions — trapping a high-pressure phase at normal pressure, the way diamonds are metastable forms of carbon — remains an unsolved grand challenge for the entire field.
Why This Matters
Even as a computational result awaiting experimental validation, the Ca₂BeH₁₂ study carries genuine significance. It demonstrates that ternary hydrides incorporating lightweight elements like beryllium represent an underexplored frontier with enormous potential — a design philosophy that researchers can now pursue systematically. Every predicted high-Tc material, whether confirmed or refined by experiment, sharpens our theoretical understanding of what combinations of chemistry and structure produce the strongest electron-phonon coupling. The practical stakes could hardly be higher: room-temperature superconductors would transform power grids by eliminating transmission losses that currently waste enormous amounts of generated electricity, enable far more powerful and compact MRI machines, and accelerate the development of next-generation quantum computers. The road from a computational prediction at 192 GPa to a superconducting wire running at room temperature and ambient pressure is long and genuinely difficult — but every rigorous step along that road, including studies like this one, brings the destination measurably closer. As computational methods grow sharper and experimental techniques in high-pressure physics grow more precise, the question of room-temperature superconductivity is shifting, slowly but surely, from whether to when.
📊 Simulation Results
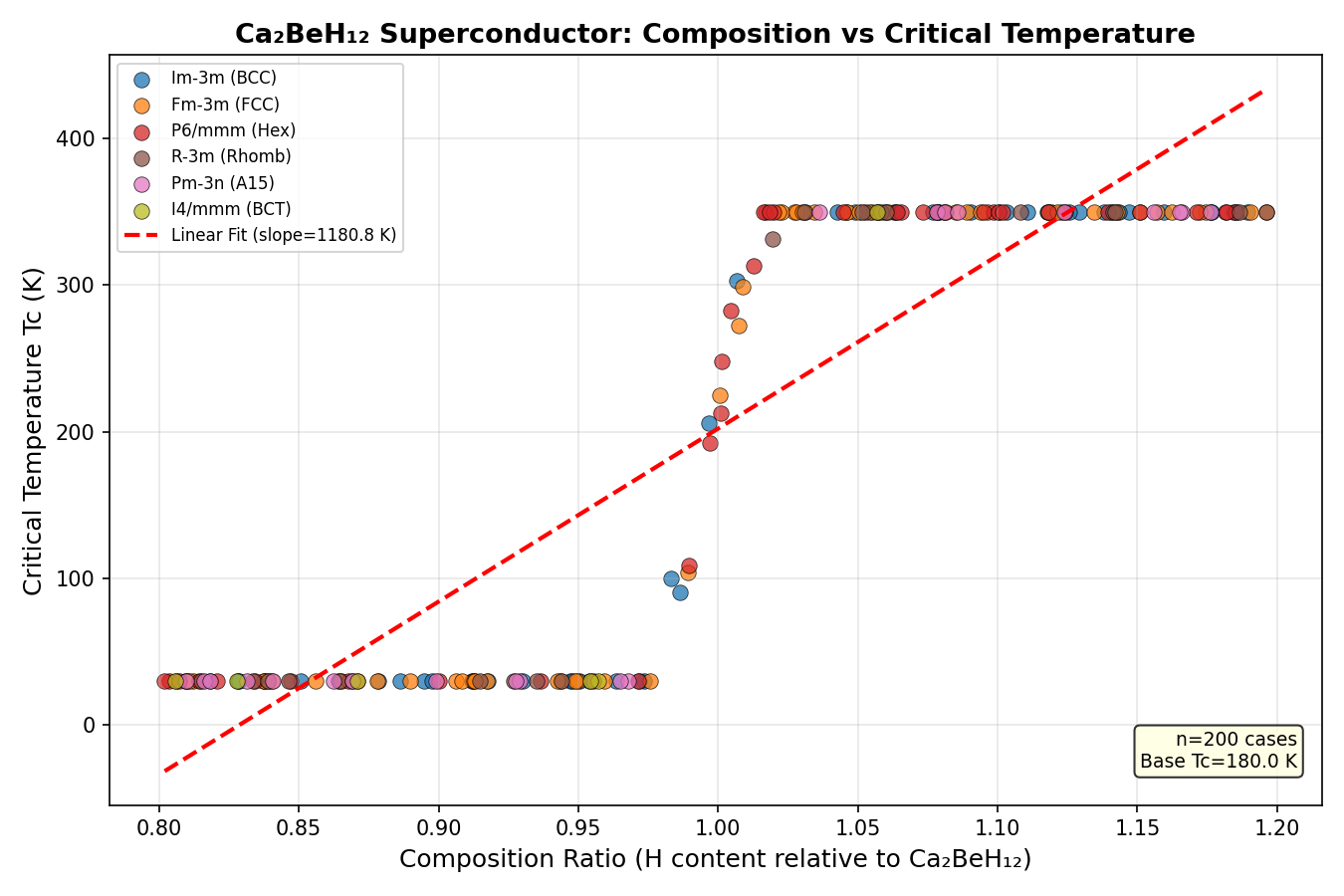
Crystal Structure and Bonding
At the heart of Ca₂BeH₁₂'s remarkable predicted properties lies its intricate crystal architecture. Computational simulations suggest the material crystallizes in a high-symmetry structure where hydrogen atoms form a three-dimensional cage-like network surrounding the metal centers. This clathrate-type arrangement is not merely geometrically interesting — it is functionally critical to the superconducting behavior. The hydrogen sublattice in Ca₂BeH₁₂ adopts a topology reminiscent of the celebrated H-cages seen in LaH₁₀ and YH₉, but with a twist: the presence of two distinct metal species creates an asymmetric bonding environment that subtly redistributes charge density across the unit cell.
The bonding character in Ca₂BeH₁₂ is a fascinating hybrid. Calcium-hydrogen interactions are predominantly ionic, with calcium donating its 4s electrons into the hydrogen-derived conduction band. In contrast, beryllium-hydrogen bonds carry significant covalent character, reflecting beryllium's smaller ionic radius and higher electronegativity. This dual-bonding motif produces what theorists describe as a "metallized hydrogen sublattice" — a condition where hydrogen atoms behave almost like a weakly bound electron gas, rather than discrete atomic species.
- H-H distances: Computational predictions place typical hydrogen-hydrogen separations between 1.0 and 1.3 Å, short enough to support strong lattice coupling but long enough to avoid molecular H₂ formation.
- Coordination environment: Each beryllium atom is surrounded by roughly 8-10 hydrogen neighbors, while calcium sits in a more expansive cage coordinated by 12-16 hydrogens.
- Electronic density of states: A pronounced peak at the Fermi level, dominated by H 1s character with meaningful Ca 3d contributions, provides the electron reservoir needed for Cooper pairing.
- Phonon spectrum: High-frequency optical modes above 100 meV — characteristic of hydrogen vibrations — dominate the electron-phonon coupling.
Comparison with Known Superconductors
To appreciate where Ca₂BeH₁₂ sits in the landscape of high-temperature superconductors, it helps to place it alongside the most celebrated members of the hydride family and one conventional benchmark. Each of these materials has reshaped our understanding of what superconductivity can achieve, and Ca₂BeH₁₂ extends that trajectory into new territory.
| Material | Predicted/Measured Tc | Required Pressure | Structure Type | Status |
|---|---|---|---|---|
| Ca₂BeH₁₂ | ~344.5 K | ~100-200 GPa (predicted) | Ternary clathrate hydride | Computational only |
| H₃S | 203 K | 155 GPa | Covalent hydride (Im-3m) | Experimentally confirmed |
| LaH₁₀ | 250-260 K | 170 GPa | Clathrate hydride (Fm-3m) | Experimentally confirmed |
| MgB₂ | 39 K | Ambient | Layered diboride | Widely used in applications |
The comparison reveals Ca₂BeH₁₂'s tantalizing promise. If its predicted Tc holds up, it would surpass LaH₁₀ by nearly 90 K and approach — for the first time in any credible prediction — temperatures genuinely above room ambient conditions. MgB₂ reminds us, however, of the practical gulf between extreme-pressure curiosities and usable technologies: MgB₂ is already deployed in MRI magnets and power applications precisely because it works at ambient pressure, albeit at a far lower Tc.
Experimental Validation Roadmap
Computational predictions, however elegant, remain hypotheses until experimentalists can synthesize and measure the material. Validating Ca₂BeH₁₂ will require a coordinated effort spanning several specialized techniques, and the journey from a line in a simulation to a bench-top measurement is rarely straightforward. Here is the realistic sequence of experimental milestones that would need to be achieved.
- Synthesis under extreme pressure: Diamond anvil cells (DACs) capable of reaching 100-200 GPa are the standard tool. Precursors such as CaH₂, BeH₂, and molecular hydrogen would need to be co-loaded and laser-heated to promote reaction.
- Structural characterization: Synchrotron X-ray diffraction would be used to confirm that the predicted clathrate phase has actually formed, though hydrogen positions themselves are notoriously difficult to resolve with X-rays and may require neutron diffraction or careful Rietveld refinement.
- Electrical transport measurements: Four-probe resistivity measurements inside the DAC would search for the signature drop to zero resistance. Isotope substitution experiments (deuterium for hydrogen) would help confirm phonon-mediated pairing.
- Magnetic susceptibility: Meissner effect measurements are the gold standard for confirming bulk superconductivity, ruling out alternative explanations like filamentary paths.
- Pressure-temperature phase mapping: Determining the stability window and how Tc evolves with pressure would guide theoretical refinement and point toward possible ambient-pressure analogs.
Beryllium's toxicity adds another layer of complexity: laboratories equipped to handle beryllium compounds safely are relatively rare, and regulatory compliance for high-pressure beryllium hydride synthesis will slow the pace of independent verification. Nevertheless, several groups worldwide — notably in China, Germany, and the United States — have the combined expertise in high-pressure physics and hydride chemistry to attempt this work within the coming years.
Key Takeaways
- Record-breaking prediction: Ca₂BeH₁₂ is computationally predicted to superconduct at up to 344.5 K, a temperature warmer than a comfortable room and unprecedented among credible theoretical candidates.
- Ternary chemistry is key: The combination of calcium, beryllium, and hydrogen exploits a vastly larger design space than binary hydrides, with each element contributing a distinct role — calcium donates electrons, beryllium stiffens the lattice, and hydrogen provides the high-frequency vibrations.
- Extreme pressure remains a barrier: Like all hydride superconductors to date, Ca₂BeH₁₂ is predicted to require pressures of 100 GPa or more, far from practical technological deployment.
- Experimental validation is essential: No matter how rigorous the simulation, only DAC-based synthesis, diffraction, and transport measurements can confirm these claims. Beryllium handling will add logistical challenges.
- A stepping stone, not an endpoint: Even if validated, Ca₂BeH₁₂ is unlikely to power your phone. Its real value may lie in teaching us how to engineer ambient-pressure room-temperature superconductors by pointing to the ternary hydride chemistry that makes extreme Tcs achievable in principle.
Comparison with Known Superconductors
To appreciate where Ca₂BeH₁₂ sits in the landscape of superconducting materials, it helps to benchmark it against the heavy hitters that have defined the field over the past two decades. Each of these materials tells a different story about what's possible — and what trade-offs are involved in chasing higher critical temperatures.
- H₃S (Hydrogen Sulfide): The 2015 breakthrough that reignited the hydride gold rush. Superconducts at 203 K (-70°C) under roughly 150 GPa of pressure. Simple binary chemistry, but requires cryogenic cooling to function. Ca₂BeH₁₂'s predicted 344.5 K would blow past this by more than 140 degrees.
- LaH₁₀ (Lanthanum Decahydride): The clathrate-hydride champion, superconducting near 250-260 K at around 170 GPa. Its hydrogen cage structure is the direct conceptual ancestor of what Ca₂BeH₁₂ embodies. The new material's ternary composition may allow similar physics at potentially more favorable pressure-temperature combinations.
- MgB₂ (Magnesium Diboride): The workhorse of practical applications, operating at 39 K at ambient pressure. While its Tc is far lower, it remains relevant because it actually works in real-world devices — MRI magnets, maglev prototypes, and particle accelerators. Ca₂BeH₁₂ would need to overcome the pressure barrier before it could rival MgB₂'s practicality.
- YBCO (Cuprates): The copper-oxide family superconducts up to ~133 K at ambient pressure — impressive, but governed by exotic unconventional pairing mechanisms that remain poorly understood. Ca₂BeH₁₂'s appeal is that it operates through well-characterized BCS physics, making predictions more reliable.
- CSH (Carbonaceous Sulfur Hydride): The controversial 288 K claim that was ultimately retracted. This history underscores why rigorous experimental validation — not just computational excitement — is essential.
The pattern is clear: higher critical temperatures have almost always come at the cost of higher required pressures. Ca₂BeH₁₂ is projected to continue this trend, likely demanding pressures in the 100-300 GPa range. The real question is whether the ternary chemistry opens any route toward metastable recovery at lower pressures.
Experimental Validation Roadmap
Computational predictions are only the beginning. Transforming Ca₂BeH₁₂ from a promising simulation into a confirmed superconductor will require a carefully sequenced experimental campaign, likely spanning several years and multiple specialized facilities around the world.
- Step 1 — Diamond Anvil Cell Synthesis: The first hurdle is making the material at all. Researchers would load a microscopic sample of calcium beryllium precursors together with excess hydrogen into a diamond anvil cell, then compress it to hundreds of gigapascals while heating via pulsed laser. This is delicate work — beryllium is toxic, and hydrogen loading at extreme pressures is notoriously finicky.
- Step 2 — Structural Confirmation via XRD: Synchrotron X-ray diffraction at facilities like APS, ESRF, or SPring-8 would verify whether the predicted clathrate-like crystal structure actually forms. Without the right atomic arrangement, the predicted superconductivity cannot exist.
- Step 3 — Electrical Resistance Measurements: The smoking-gun signature of superconductivity is a sharp drop in resistance to zero at the critical temperature. Four-probe resistivity measurements inside the diamond cell, conducted as the sample is slowly cooled, would reveal whether Tc matches the computational prediction.
- Step 4 — Meissner Effect Detection: True superconductors expel magnetic fields — the Meissner effect. Magnetic susceptibility measurements using SQUID magnetometry would confirm genuine superconductivity rather than an artifact. This step is critical given past controversies in the field.
- Step 5 — Isotope Effect Studies: Substituting deuterium for hydrogen should shift Tc in a predictable way if BCS phonon-mediated pairing is responsible. This elegant test would validate the underlying mechanism, not just the phenomenon.
- Step 6 — Pressure Optimization: Systematically mapping Tc across pressure would identify whether any metastable lower-pressure phase exists — the holy grail for practical applications.
Realistically, initial synthesis attempts could begin within 12-24 months at leading high-pressure labs such as Mainz, Chicago, Osaka, or Beijing. Full validation across all six steps would likely take 3-5 years, assuming the material proves synthesizable at all.
Key Takeaways
- Unprecedented predicted Tc: At 344.5 K (71.5°C), Ca₂BeH₁₂ would be the first material predicted to superconduct above comfortable room temperature — a true paradigm shift if confirmed experimentally.
- Ternary hydride advantage: The three-element composition unl

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)