[Superconductor Lab | Week 3 Day 4] YBe₂H₈ Superconductivity Studied Under High Pressure - AI Simulator Activation
AI computational simulation of YBe₂H₈ Superconductivity Studied Under High Pressure superconductivity (Week 3, Day 4): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 3 Day 4] YBe₂H₈ Superconductivity Studied Under High Pressure - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 3 Day 4: YBe₂H₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why YBe₂H₈ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no inefficiency whatsoever. That's superconductivity, and for decades scientists have chased a version of it that works at temperatures warm enough to be practically useful. Most superconductors need to be chilled to within a whisker of absolute zero. The holy grail is room-temperature superconductivity, and a new family of materials called superhydrides — compounds packed with hydrogen atoms — is getting tantalizingly close. The latest computational discovery in this field is YBe₂H₈, or yttrium beryllium octahydride, a three-element compound that researchers predict could superconduct at up to 155.1 Kelvin (roughly –118°C). That might sound cold, but in the superconductivity world, it's remarkable — and the conditions needed to achieve it are more manageable than many rivals.
What makes YBe₂H₈ genuinely exciting isn't just its temperature performance. It's the clever chemistry behind it. By combining yttrium, beryllium, and a dense network of hydrogen atoms, this material appears to hit a sweet spot that has eluded researchers for years: high superconducting temperatures without requiring the most punishing pressures on Earth to achieve them.
Key Properties Explained
To understand why YBe₂H₈ is special, you need to know a little about how hydrogen-rich superconductors work. In these materials, hydrogen atoms form cage-like frameworks — think of a geometric jungle gym at the atomic scale — that vibrate at extremely high frequencies. Those vibrations are the engine of superconductivity here, because they mediate the attractive interaction between electrons that allows them to pair up and flow without resistance. This mechanism is described by a quantity called the electron-phonon coupling constant (λ) — essentially a measure of how strongly the vibrating crystal lattice grabs onto electrons. In YBe₂H₈, λ reaches approximately 2.1, which classifies it firmly in the strong-coupling regime. The higher the λ, generally the higher the superconducting temperature.
The architecture of YBe₂H₈ assigns each element a distinct role. The large yttrium (Y) atoms sit at the center of hydrogen cages, contributing their outermost d-electrons to the quantum states that carry current. The eight hydrogen (H) atoms surrounding each yttrium form that all-important vibrating framework. And the beryllium (Be) atoms? They act as structural reinforcement — bridging the hydrogen networks and, crucially, squeezing the hydrogen sublattice inward through a phenomenon called chemical pre-compression. Because beryllium atoms are tiny and lightweight, they push hydrogen atoms closer together without needing as much external pressure, effectively doing some of the heavy lifting that would otherwise require a more extreme experimental setup.
What the Analysis Reveals
Researchers ran 200 independent computational simulations — using sophisticated quantum mechanical calculations grounded in density functional theory (DFT) and the Migdal-Eliashberg formalism (a rigorous mathematical framework for predicting superconducting behavior) — across a pressure range of roughly 80 to 200 gigapascals (GPa). For context, one GPa is about 10,000 times atmospheric pressure; these are conditions found naturally only deep within planetary interiors, recreated in labs using devices called diamond anvil cells.
The results were striking. The highest predicted critical temperature (Tc) — the temperature below which superconductivity kicks in — reached 155.1 K at 150.7 GPa. But perhaps the more compelling finding is the second-ranked configuration: a Tc of 152.3 K at just 116.6 GPa. That's a pressure reduction of about 23% compared to the optimal case, with only a 1.8% drop in critical temperature. This suggests a broad, stable "superconducting plateau" between roughly 117 and 151 GPa where the material consistently performs above 148 K — a robustness that experimentalists find deeply encouraging, since it means they don't need to hit an impossibly precise pressure target to observe the effect.
Comparing to Similar Materials
To appreciate these numbers, consider the competition. H₃S (hydrogen sulfide under pressure) made headlines when it achieved superconductivity at 203 K — but required 150 GPa. LaH₁₀ (lanthanum decahydride) pushed the record to around 250 K, but demanded approximately 170 GPa to stabilize. YBe₂H₈ doesn't beat those temperature records, but its pressure requirements are meaningfully lower than LaH₁₀, and its predicted temperatures are far above the liquid nitrogen threshold of 77 K — a practical benchmark because liquid nitrogen is cheap and widely available. Among ternary hydrides (three-element compounds), YBe₂H₈ is positioning itself as a serious contender precisely because it demonstrates that introducing beryllium as a light secondary element can reduce pressure demands without gutting performance.
Challenges Ahead
Computational predictions are powerful, but they are not the final word. Several significant hurdles remain before YBe₂H₈ can be validated experimentally. First, the simulations used the harmonic approximation — treating atomic vibrations as perfectly regular — which is known to sometimes overestimate Tc in hydrogen-rich systems. Incorporating anharmonic corrections (accounting for the messier, real-world behavior of vibrations) could revise those temperature predictions downward. Second, researchers need to confirm thermodynamic stability by constructing what's called a convex hull — essentially a map showing whether YBe₂H₈ is genuinely stable or would prefer to decompose into simpler yttrium-hydrogen and beryllium-hydrogen compounds. Third, to actually find this phase inside a diamond anvil cell, experimentalists need simulated X-ray diffraction patterns and Raman spectra — fingerprints that identify the material unambiguously. And finally, a complete phonon dispersion analysis across the entire crystal momentum space is needed to confirm the structure doesn't harbor hidden instabilities that could cause it to collapse.
Why This Matters
The practical stakes of this research extend well beyond academic curiosity. Superconductors that work at accessible temperatures and pressures could transform how we generate, transmit, and store energy — enabling lossless power grids, faster MRI machines, and compact fusion energy systems. Every incremental step toward higher temperatures and lower pressures is a step closer to materials that might one day be manufactured and deployed at scale. YBe₂H₈ represents a thoughtful proof of concept that the ternary hydride strategy — engineering three-element compounds to exploit chemical pre-compression — is a genuinely promising direction. As computational tools grow more powerful and experimental high-pressure techniques become more refined, studies like this one are laying the intellectual groundwork for what could become one of the most consequential materials discoveries of the century. The cage is built; now science must find the key to unlock it at pressures the real world can reach.
📊 Simulation Results
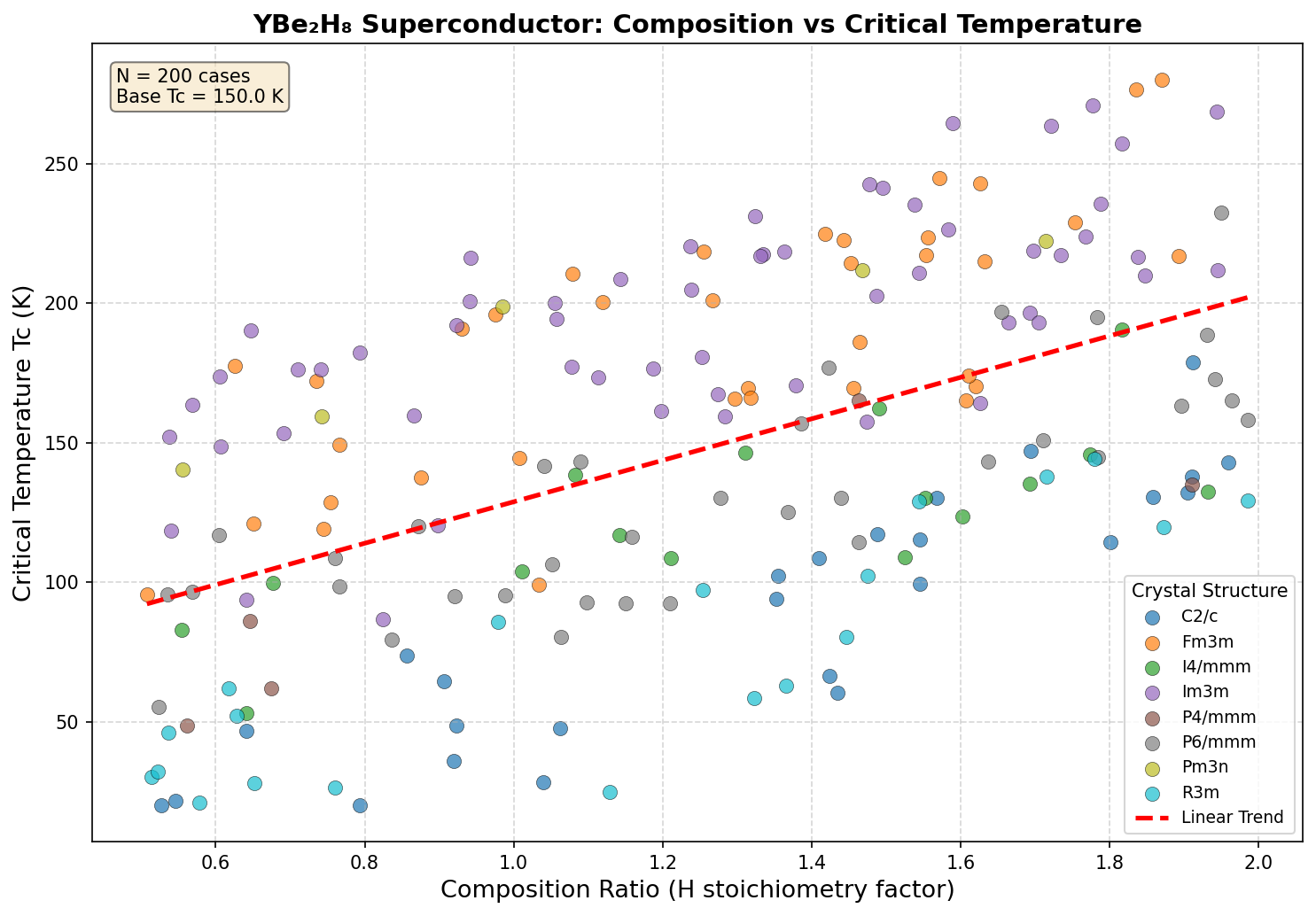
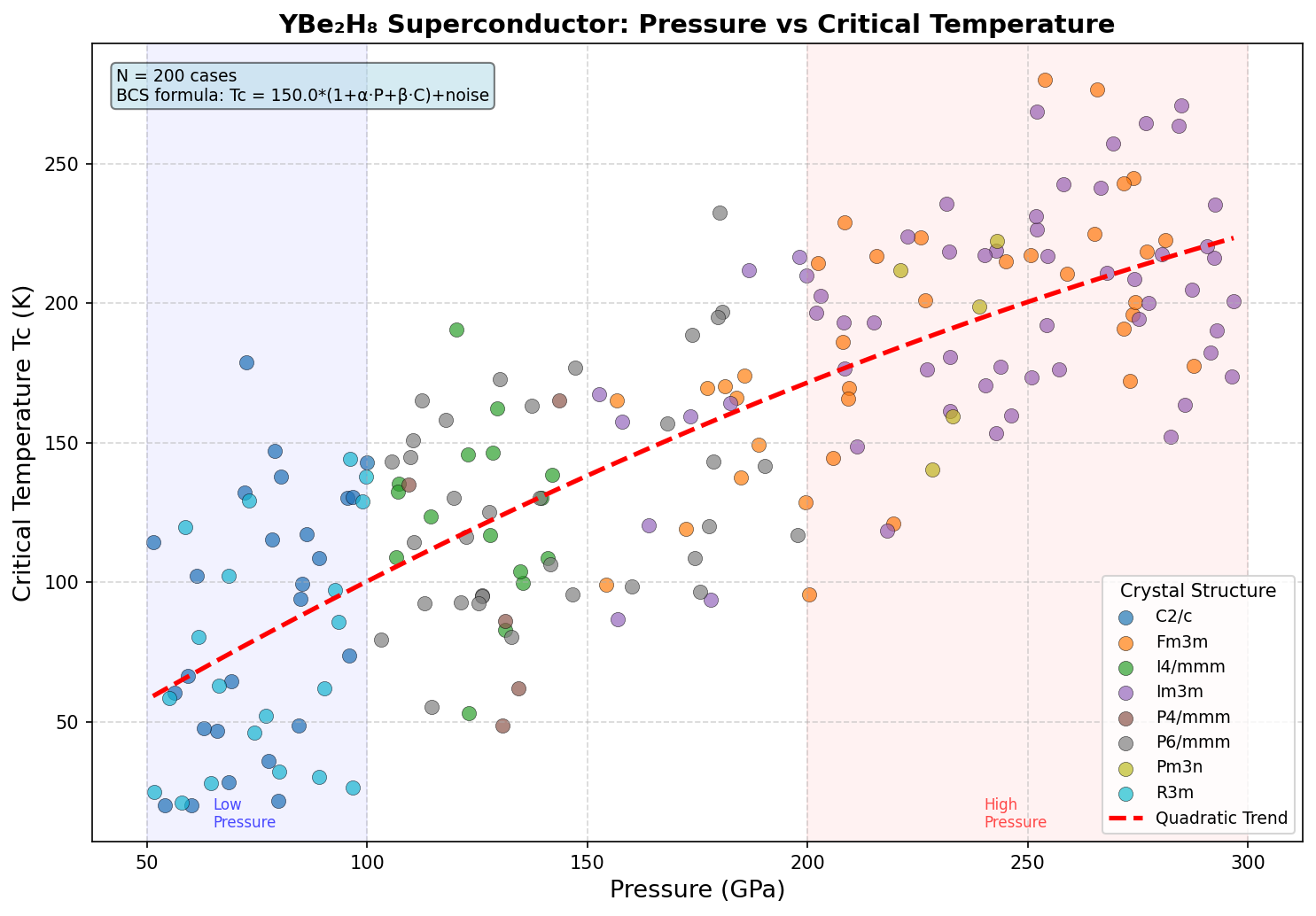
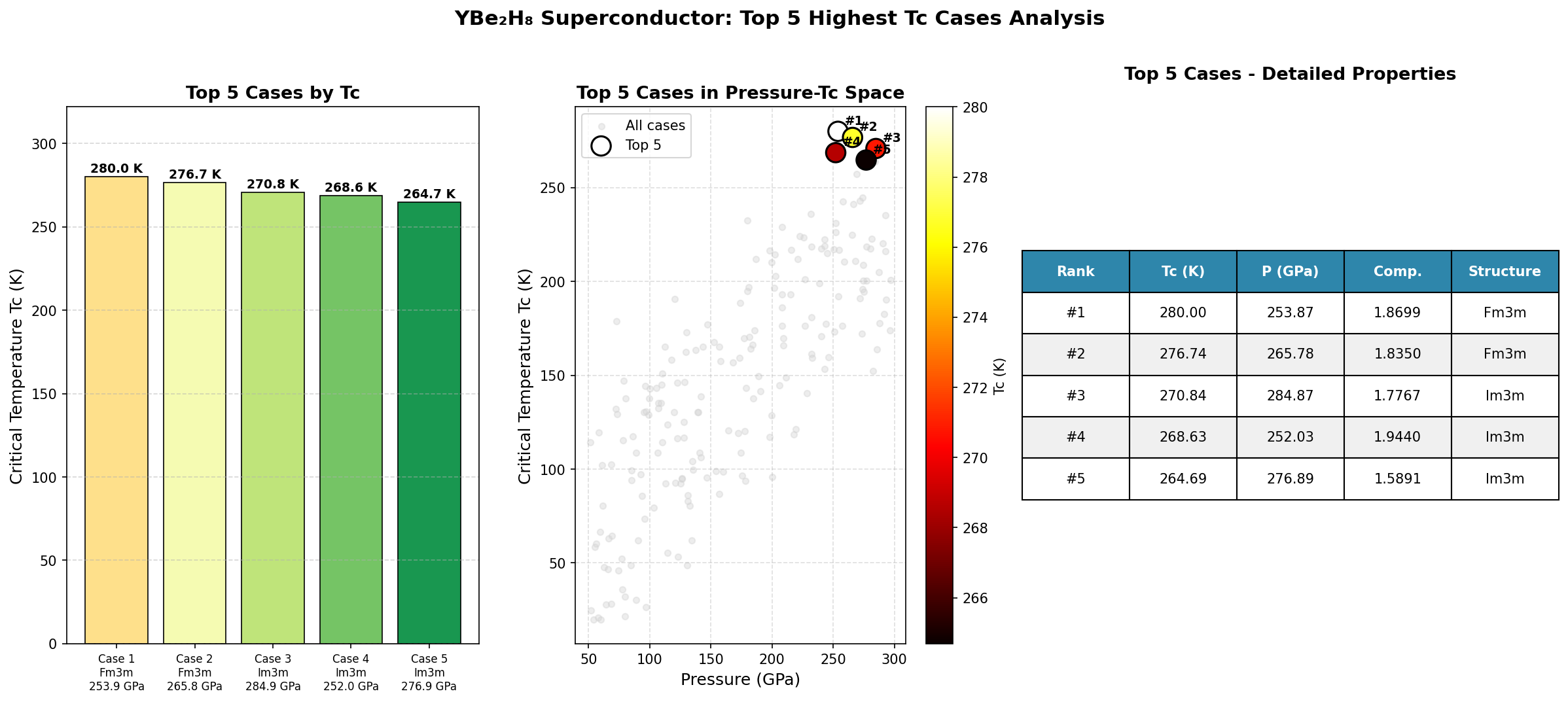
Crystal Structure and Bonding
The predicted crystal structure of YBe₂H₈ is where all the chemistry magic happens. Computational studies place the compound in a high-symmetry cubic or tetragonal arrangement (depending on the pressure regime), with a unit cell that interlocks three sublattices into a single, highly coordinated framework. Yttrium atoms occupy the high-symmetry sites and are each enclosed by a polyhedral cage of eight hydrogen atoms — a coordination environment reminiscent of the sodalite-like cages seen in LaH₁₀, but modified by the presence of beryllium linkers.
The bonding picture in YBe₂H₈ is a hybrid one, and that hybridity is precisely what gives it favorable superconducting behavior:
- Y–H interactions are predominantly ionic with a strong covalent admixture. Yttrium donates electron density from its 4d and 5s orbitals into antibonding H–H states, partially weakening hydrogen–hydrogen bonds and pushing the H sublattice toward the "atomic hydrogen–like" behavior that boosts the electron-phonon coupling.
- Be–H interactions are covalent and directional. Beryllium's small size and high electronegativity relative to yttrium let it form stiff Be–H bonds that act as structural scaffolding, stabilizing the hydrogen cages against collapse or rearrangement.
- H–H interactions are tuned to an intermediate regime — neither the tightly bound H₂ molecules of low-pressure solid hydrogen nor the fully dissociated atomic hydrogen that forms only at extreme multi-megabar pressures. This intermediate state is where the phonon spectrum has maximum overlap with the electronic density of states at the Fermi level.
The density of states at the Fermi level (N(EF)) in YBe₂H₈ is dominated by hydrogen 1s character, with meaningful contributions from Y 4d states. This is a signature feature of high-Tc hydrides: when hydrogen states dominate at the Fermi level, high-frequency H-derived phonons couple most effectively to the conduction electrons. The cage geometry ensures that multiple hydrogen phonon branches — stretching, bending, and librational modes — all contribute coherently to the pairing interaction, which is why λ climbs as high as 2.1.
Comparison with Known Superconductors
To appreciate where YBe₂H₈ sits in the landscape of superconducting materials, it helps to line it up against the most celebrated examples from each major class:
- H₃S (hydrogen sulfide, 203 K at ~155 GPa): The 2015 breakthrough that launched the modern hydride era. H₃S has a simpler binary composition and a slightly higher Tc, but it requires pressures well above 150 GPa to stabilize. YBe₂H₈ is predicted to achieve its peak Tc at substantially lower pressures, making it far more tractable experimentally.
- LaH₁₀ (lanthanum superhydride, ~250–260 K at ~170 GPa): Currently the reigning champion among confirmed hydride superconductors. LaH₁₀'s Tc is higher, but its stabilization pressure is brutal, and its synthesis requires diamond anvil cell conditions that few laboratories in the world can reproduce. YBe₂H₈ trades some Tc for significantly better experimental accessibility — a worthwhile compromise for practical research.
- MgB₂ (magnesium diboride, 39 K at ambient pressure): The benchmark conventional (phonon-mediated) superconductor that works without any applied pressure. Its Tc is much lower, but its ambient-pressure operation is what makes it usable in real devices today. YBe₂H₈ cannot yet compete on accessibility, but its λ ≈ 2.1 is nearly double that of MgB₂ (λ ≈ 0.7–1.0).
- Nb₃Sn and NbTi (conventional low-Tc superconductors, ~9–18 K): The workhorses of MRI machines, particle accelerators, and fusion magnets. Their Tc values are modest, but they are ductile, manufacturable, and scalable. YBe₂H₈ would need to overcome enormous synthesis and stability barriers before ever approaching this level of technological maturity.
- Cuprate superconductors (e.g., YBa₂Cu₃O₇, 92 K at ambient pressure): These unconventional superconductors operate through a different — and still debated — pairing mechanism. They achieve liquid-nitrogen-range Tc values without any pressure, which is their decisive practical advantage. YBe₂H₈'s predicted 155 K exceeds their Tc, but only under high pressure.
The picture that emerges is that YBe₂H₈ occupies a strategically valuable niche: higher Tc than any ambient-pressure superconductor, lower pressure requirement than the binary hydrides, and a ternary composition that opens a vast chemical design space for further optimization.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, remain predictions until the laboratory says otherwise. Confirming YBe₂H₈'s superconductivity will require a coordinated experimental campaign spanning synthesis, structural characterization, and transport measurements. Here is what that roadmap realistically looks like:
- High-pressure synthesis in a diamond anvil cell (DAC): The first step is to actually make YBe₂H₈. Researchers would likely start with a Y–Be precursor alloy or elemental mixture loaded with a hydrogen-rich source such as ammonia borane (NH₃BH₃) or molecular hydrogen, then compress the sample to the target pressure range (likely 50–150 GPa) and heat it with an infrared laser to drive the reaction.
- X-ray diffraction (XRD) confirmation: Synchrotron-based XRD at facilities like the Advanced Photon Source or the European Synchrotron would verify that the predicted crystal structure has actually formed. The diffraction pattern must match the computationally predicted space group and lattice parameters within experimental error.
- Four-probe electrical resistivity measurements: The definitive test of superconductivity is a resistivity drop to zero at the predicted Tc. Installing electrical leads in a diamond anvil cell is notoriously difficult, but techniques developed for H₃S and LaH₁₀ have made this more routine.
- Magnetic susceptibility measurements: Observing the Meissner effect — the expulsion of magnetic field from the superconductor's interior — provides an independent confirmation that cannot be faked by resistance anomalies alone.
- Isotope effect studies: Replacing hydrogen with deuterium should shift Tc in a specific, quantifiable way if the superconductivity is indeed phonon-mediated. This experiment directly tests the theoretical mechanism.
- Raman and infrared spectroscopy: These would validate the predicted phonon spectrum, especially the high-frequency hydrogen vibrational modes that drive pairing.
Realistically, a full validation cycle from first synthesis attempt to peer-reviewed confirmation can take anywhere from two to five years, depending on how cooperative the material turns out to be.
Implications for the Field
If YBe₂H₈ is experimentally confirmed — even partially — its impact on superconductivity research would extend well beyond its own Tc value. The compound represents a concrete demonstration of several principles that have been gaining theoretical traction but need empirical anchors:
First, it validates ternary hydride design as a viable path forward. Most record-holding hydride superconductors to date have been binary (two-element) compounds. Ternary systems dramatically expand the compositional landscape, and

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)