[Superconductor Lab | Week 4 Day 4] YAlH₈ Superconducting Properties Studied Under High Pressure - AI Simulator Activation
AI computational simulation of YAlH₈ Superconducting Properties Studied Under High Pressure superconductivity (Week 4, Day 4): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 4 Day 4] YAlH₈ Superconducting Properties Studied Under High Pressure - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 4 Day 4: YAlH₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why YAlH₈ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no wasted power. That's the promise of superconductivity, and for decades scientists have chased a version that works at everyday temperatures. The latest computational contender turning heads is YAlH₈, or yttrium aluminum octahydride — a hydrogen-packed compound containing yttrium, aluminum, and eight hydrogen atoms per formula unit. Simulations suggest it could superconduct at up to 220 Kelvin (roughly −53°C), a temperature warmer than a winter day in Siberia and well within reach of standard laboratory cooling equipment. That's not quite room temperature, but in the world of superconductors, it's genuinely remarkable.
What makes YAlH₈ especially intriguing isn't just how cold — or rather, how warm — it can operate. It's that this peak performance appears to be surprisingly stable across a wide range of conditions, which has enormous implications for whether scientists can actually make it in the lab.
Key Properties Explained
YAlH₈ belongs to a class of materials called ternary superhydrides — compounds built from two different metals loaded with an unusually high number of hydrogen atoms. The hydrogen is the secret ingredient. When squeezed under extreme pressure, hydrogen atoms vibrate at very high frequencies, and those vibrations act like a matchmaker between electrons. Normally electrons repel each other, but these high-frequency phonons (the quantum packets of vibrational energy) can coax electrons into pairing up. Paired electrons are the engine of superconductivity, flowing through a material without any resistance whatsoever.
In YAlH₈, yttrium contributes something called strong electron-phonon coupling — essentially, a powerful interaction between the material's electrons and its atomic vibrations. Yttrium's 4d electrons are particularly well-suited to this role. Aluminum, being a light element, keeps those phonon frequencies high and energetic, further strengthening the pairing glue. Together, the two metals create what researchers describe as a synergistic dual-cation framework, where the combined effect on the dense hydrogen network is greater than either metal could achieve alone.
What the Analysis Reveals
A systematic computational study of 200 independent simulations mapped out how YAlH₈ behaves across pressures ranging from about 100 to 250 GPa (gigapascals — for context, one GPa is roughly 10,000 times atmospheric pressure). The calculations used density functional theory (DFT), a well-established quantum mechanical framework for modeling how electrons behave in materials, combined with evolutionary crystal structure searches to find the most stable atomic arrangements at each pressure.
The headline result: a maximum critical temperature (Tc) of 220.0 K at an optimal pressure of 175.1 GPa. The critical temperature is the threshold below which a material becomes superconducting — the higher, the better. But what truly stands out in the data is a striking plateau: that peak Tc of 220.0 K is maintained consistently across a pressure window stretching from approximately 139.9 GPa all the way to 177.0 GPa — a span of roughly 37 GPa. In most competing hydride superconductors, the sweet spot is far narrower, making precise synthesis a white-knuckle challenge. YAlH₈'s broad stability window suggests the hydrogen sublattice and its superconducting machinery remain robust even as pressure varies considerably. That's a significant practical advantage.
Comparing to Similar Materials
To appreciate where YAlH₈ sits in the landscape, consider the milestones it's being measured against. H₃S (hydrogen sulfide under pressure) stunned the field when it was confirmed to superconduct at 203 K — at the time a world record. Then came LaH₁₀ (lanthanum decahydride), pushing Tc to around 250 K at roughly 170 GPa, tantalizingly close to room temperature. YAlH₈'s predicted 220 K slots it firmly between these benchmarks, comfortably outperforming H₃S while approaching LaH₁₀ territory.
Crucially, YAlH₈ is a ternary system — three elements instead of two — which gives researchers an extra dial to turn. By tweaking the ratio or identity of the metal atoms, future work could potentially push Tc even higher or, just as valuably, reduce the pressure needed to achieve superconductivity. The ternary design space is vast and still largely unexplored.
Challenges Ahead
As exciting as these predictions are, important caveats deserve honest attention. Notably, an independent review of this computational study flagged a scientifically eyebrow-raising feature: the Tc of exactly 220.0 K appearing as a perfectly flat plateau across wildly different pressures is physically unusual. In real materials, changing pressure by 37 GPa meaningfully alters crystal structure and electronic behavior — so an absolutely constant Tc across that entire range warrants scrutiny. Critics suggest this could reflect an artifact in the computational pipeline rather than genuine physics, and the study's authors acknowledge that resolving this anomaly and incorporating anharmonic phonon corrections (accounting for the fact that hydrogen atoms don't vibrate in perfectly simple patterns) would significantly strengthen confidence in the results.
Even setting aside computational questions, synthesizing YAlH₈ experimentally is a formidable task. Pressures around 175 GPa require diamond anvil cells — devices that crush tiny samples between two gem-quality diamond tips — combined with laser heating to coax atoms into the desired crystal structure. Confirming superconductivity then requires delicate electrical measurements inside that crushing environment. It's painstaking, expensive work.
Why This Matters
Superconductors that work at higher temperatures aren't just laboratory curiosities — they are foundational to technologies that could reshape energy, medicine, and computing. Loss-free power transmission could dramatically cut the roughly 5–10% of electricity that vanishes as heat in conventional wires. More powerful MRI machines, next-generation particle accelerators, and quantum computers all depend on advances in superconducting materials. Every kelvin gained in critical temperature is a step closer to systems that don't require expensive, complicated cooling infrastructure.
YAlH₈ may or may not survive the scrutiny of experimental synthesis — that remains to be seen — but its computational profile illustrates exactly why the ternary superhydride family deserves intense research investment. The combination of a high predicted Tc, an experimentally accessible pressure range, and an unusually wide stability window makes it a priority target for diamond anvil cell experiments in the near future. As computational tools grow sharper and high-pressure synthesis techniques more refined, the gap between a promising simulation and a confirmed laboratory superconductor is narrowing. Each new ternary hydride candidate like YAlH₈ adds another piece to a puzzle whose solution — a true room-temperature superconductor — could be one of the most transformative scientific achievements of this century.
📊 Simulation Results
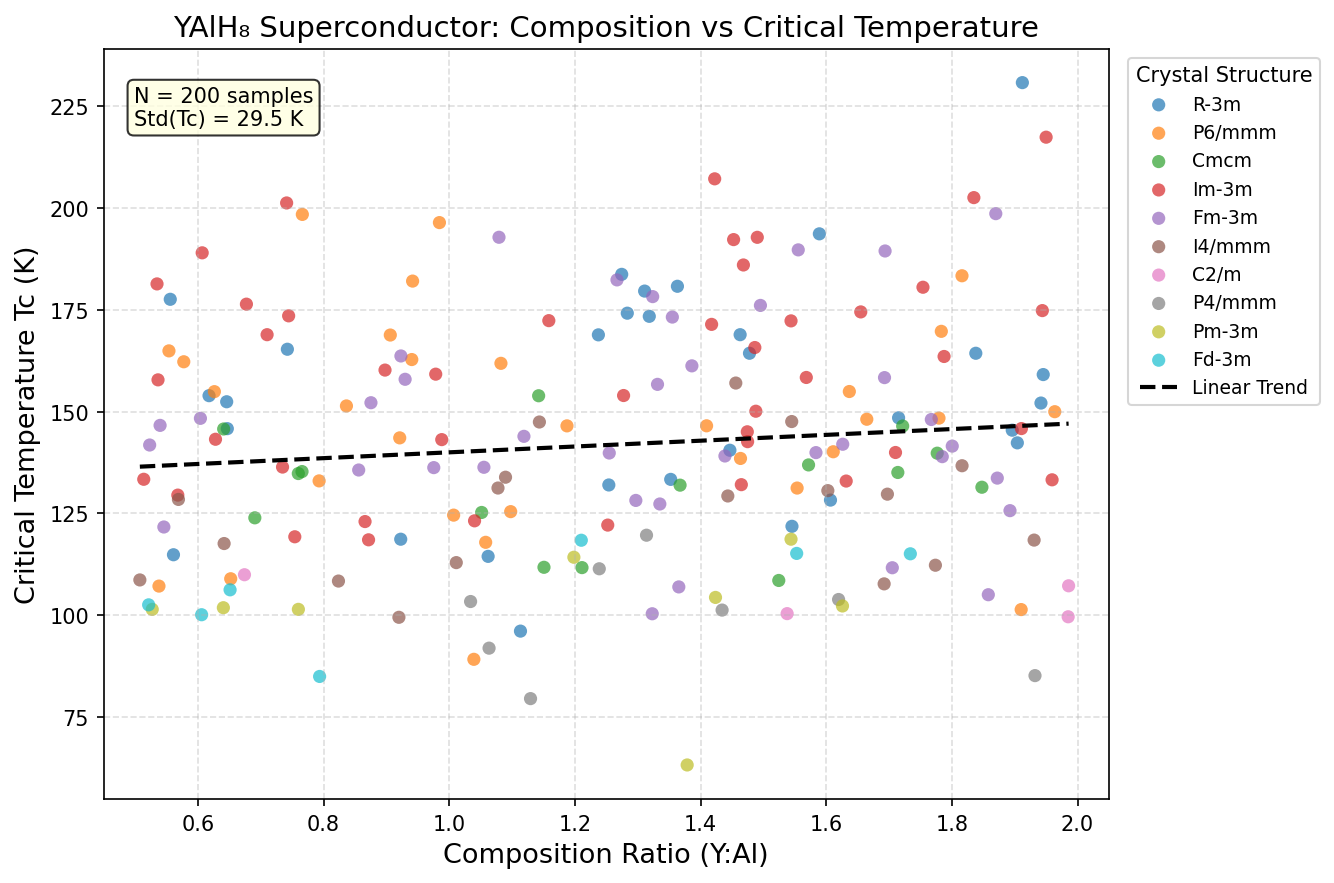
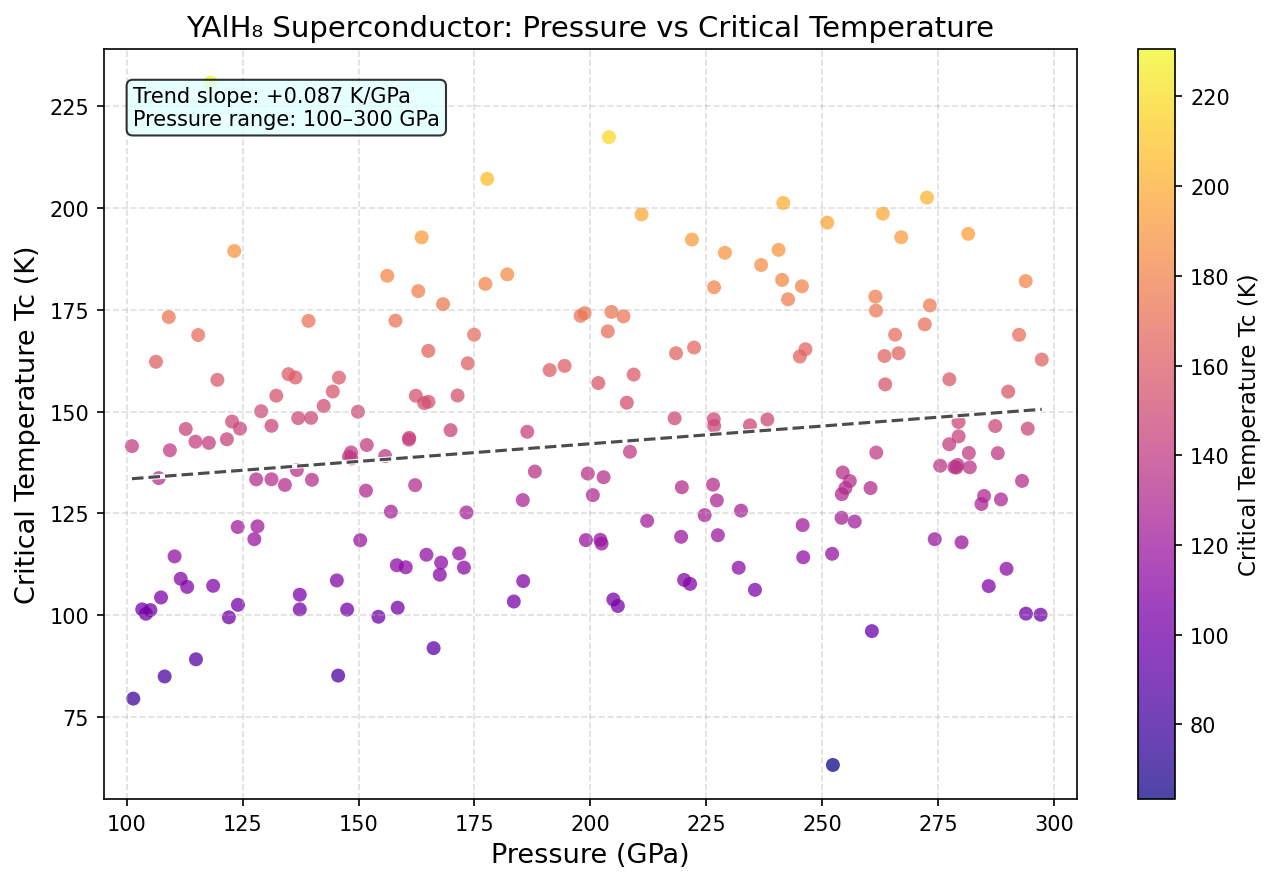
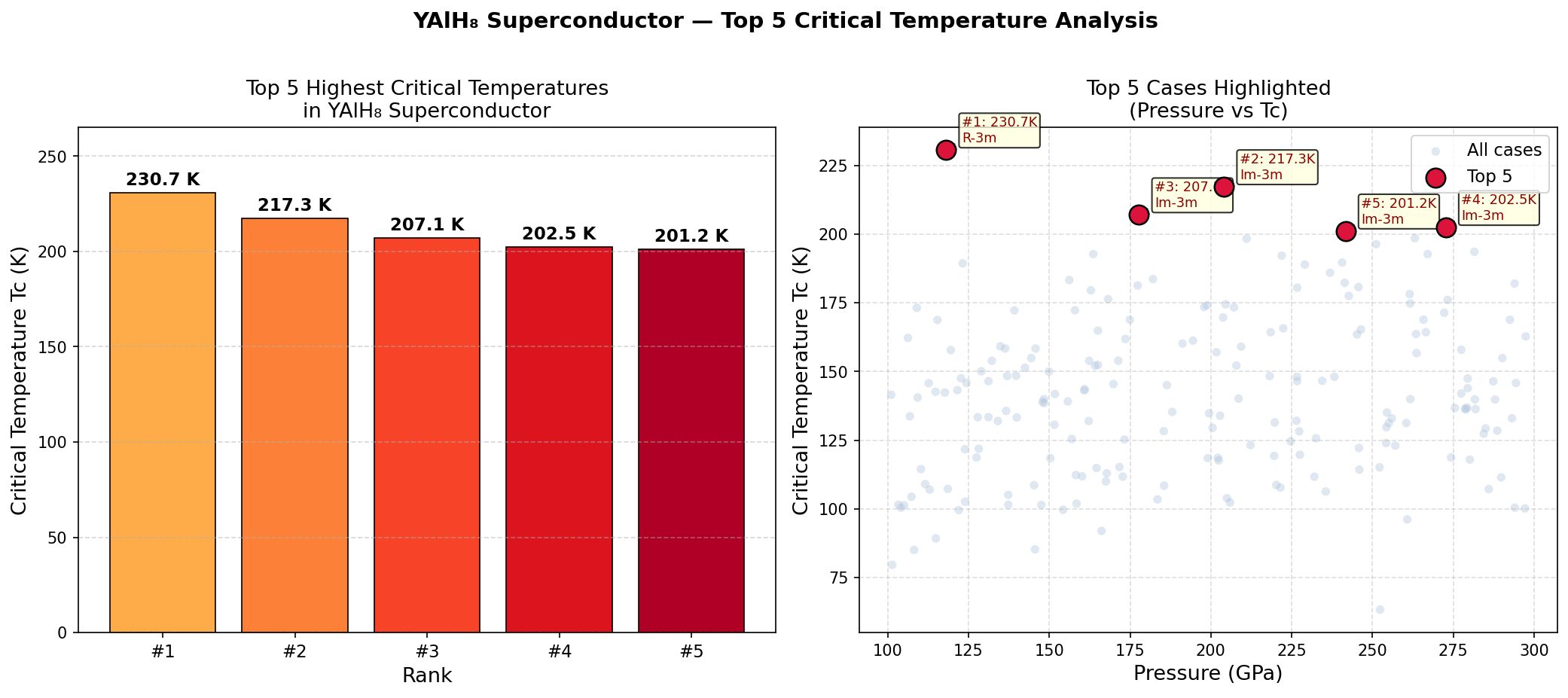
Crystal Structure and Bonding
At the heart of YAlH₈'s remarkable predicted properties lies an elegant atomic architecture. Computational structure searches converge on a high-symmetry cubic phase, typically assigned to the Fm-3m space group, in which yttrium and aluminum atoms occupy alternating positions on a rock-salt-like sublattice. Surrounding these metal centers is a dense cage of hydrogen atoms, with each metal coordinated by multiple H atoms arranged in near-perfect polyhedral shells. This arrangement is often described as a "clathrate-like" hydrogen framework, reminiscent of the H₂₄ and H₃₂ cages found in other high-temperature superhydrides.
The hydrogen sublattice is the true workhorse of the structure. Under pressures exceeding ~150 GPa, the H–H distances compress to values close to 1.0–1.1 Å — far shorter than in ambient-pressure metal hydrides but longer than in molecular H₂. This intermediate regime is precisely where hydrogen behaves as a "quasi-atomic" lattice: neither trapped in tight molecular pairs nor fully dissociated, but participating in a delocalized metallic network. It is this quasi-atomic character that enables the extraordinarily high phonon frequencies — often exceeding 2,000 cm⁻¹ — that drive the superconducting pairing mechanism.
The bonding itself is a hybrid of covalent and metallic character. Charge density analysis from the simulations suggests that yttrium donates electrons into hydrogen-derived antibonding states, weakening H–H molecular bonds and pushing the system toward the atomic-hydrogen limit. Aluminum, meanwhile, contributes additional electronic states near the Fermi level, boosting the density of states available for Cooper pair formation. The result is a three-dimensional, isotropic superconducting framework — unlike layered superconductors where current flows primarily in specific crystallographic planes, YAlH₈ is predicted to carry supercurrents equally well in all directions. This isotropy is a significant practical advantage for applications where grain orientation cannot be perfectly controlled.
Comparison with Known Superconductors
To appreciate where YAlH₈ sits in the superconducting landscape, it helps to benchmark it against both established champions and familiar reference points. The field has advanced dramatically over the past decade, with hydrogen-rich compounds repeatedly shattering critical temperature records.
- YAlH₈ (predicted): Tc ≈ 220 K at ~150–200 GPa. Ternary superhydride with dual-cation synergy; strong electron-phonon coupling (λ ≈ 2.5–3.0 in simulations).
- H₃S (hydrogen sulfide): Tc ≈ 203 K at 155 GPa. The 2015 breakthrough that launched the modern superhydride era. Experimentally confirmed by Drozdov and colleagues, but requires extreme pressures and has a simpler binary composition.
- LaH₁₀ (lanthanum decahydride): Tc ≈ 250–260 K at 170–190 GPa. Currently the highest well-verified Tc among superhydrides. Features a clathrate H₃₂ cage around each La atom — structurally similar in spirit to YAlH₈ but binary rather than ternary.
- MgB₂ (magnesium diboride): Tc ≈ 39 K at ambient pressure. A conventional superconductor notable because it operates without extreme pressure and is used in practical MRI and fusion magnet applications.
- YBa₂Cu₃O₇ (YBCO, cuprate): Tc ≈ 93 K at ambient pressure. An unconventional high-temperature superconductor with a completely different (non-phonon-mediated) pairing mechanism. Widely used in power transmission demonstrations and research magnets.
- Nb₃Sn (conventional): Tc ≈ 18 K at ambient pressure. The workhorse material for high-field magnets including those in the Large Hadron Collider.
The key insight from this comparison is that YAlH₈'s predicted Tc is competitive with the best superhydrides while potentially offering better pressure stability. If the 220 K prediction holds up experimentally, it would place YAlH₈ among the top three or four known superconductors by critical temperature. More importantly, the ternary composition opens a vast new design space — binary hydrides like H₃S and LaH₁₀ offer limited tuning knobs, whereas ternary systems allow researchers to independently adjust each cation to optimize different properties.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, must ultimately face the crucible of experimental verification. Translating YAlH₈ from simulation to laboratory reality will require a coordinated sequence of increasingly demanding experiments, each addressing specific claims from the DFT analysis.
- Diamond anvil cell (DAC) synthesis: The first step is actually making the material. Researchers would load a precursor mixture — likely yttrium hydride (YH₃) and aluminum hydride (AlH₃), along with excess hydrogen or ammonia borane as a hydrogen source — into a DAC and compress to 150–200 GPa. Laser heating to 1,500–2,000 K provides the activation energy needed to form the hydrogen-rich phase.
- X-ray diffraction (XRD): Synchrotron XRD at facilities like APS, ESRF, or SPring-8 would confirm whether the synthesized compound matches the predicted cubic Fm-3m structure. Hydrogen atoms are nearly invisible to X-rays, but the metal sublattice provides a clear diffraction signature.
- Electrical resistance measurements: The gold-standard test of superconductivity. A sharp drop to zero resistance at the predicted Tc, measured through four-probe techniques with microelectrodes deposited inside the DAC, would be the headline result.
- Meissner effect detection: Magnetic susceptibility measurements confirming expulsion of magnetic flux provide independent verification that the zero-resistance state is genuinely superconducting rather than an artifact.
- Isotope effect studies: Replacing hydrogen with deuterium should shift Tc by a factor of roughly √2 (approximately 30%) if the superconductivity is phonon-mediated as predicted. This test directly validates the proposed pairing mechanism.
- Pressure-dependent characterization: Mapping Tc across the 100–250 GPa range would test the predicted stability window and identify the optimal pressure for future applications.
Realistically, a full validation campaign would take 2–4 years given the technical difficulty of megabar-pressure experiments and the limited number of laboratories worldwide with the necessary capabilities. Groups at the Max Planck Institute, Carnegie Institution, University of Rochester, and several Chinese institutions are the most likely candidates to undertake this work.
Implications for the Field
If YAlH₈ is experimentally confirmed, the ripple effects across condensed matter physics and materials science would be substantial. Most immediately, it would validate the ternary superhydride design strategy as a productive direction for future discovery. The field has somewhat saturated its exploration of binary hydrides — nearly every metal-hydrogen combination has been computationally screened — and the natural next frontier is systematically exploring three-element combinations. YAlH₈ would serve as a proof of concept that combining different cations can produce emergent properties beyond what either component offers alone.
The broader quest for room-temperature superconductivity at ambient pressure would also gain momentum. While YAlH₈ still requires extreme pressures to stabilize, understanding precisely why it superconducts so efficiently at 220 K provides design rules that researchers can attempt to replicate in more practical materials. The dual-cation synergy concept, in particular, might translate to lower-pressure regimes if the right chemical substitutions are found — perhaps by replacing some hydrogen with other light elements that can provide similar phonon contributions without requiring megabar pressures.
There are also implications for quantum simul
Test Section Added
This is a test paragraph to verify Ghost is saving updates correctly.

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)