[Superconductor Lab | Week 8 Day 4] Sr₃BeH₂₀ - AI Simulator Activation
AI computational simulation of Sr₃BeH₂₀ superconductivity (Week 8, Day 4): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 8 Day 4] Sr₃BeH₂₀ - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 8 Day 4: Sr₃BeH₂₀
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Sr₃BeH₂₀ Stands Out
Imagine an electrical wire that carries current with absolutely zero energy lost to heat — no resistance, no waste, pure efficiency. That's the promise of superconductivity, and for decades, scientists have been racing to find materials that achieve it at temperatures practical enough to use in the real world. A new computational study has thrown a compelling new name into that race: Sr₃BeH₂₀, a exotic compound built from strontium, beryllium, and a remarkable abundance of hydrogen. Simulations predict this material could superconduct at up to 225.6 Kelvin — that's about -47°C, or roughly the temperature of a brutally cold winter day in Siberia. In the world of superconductivity, that's extraordinarily warm.
What makes Sr₃BeH₂₀ particularly intriguing is its identity as a ternary superhydride — a compound that combines not one but two different metals alongside a hydrogen-packed crystal lattice. This deliberate mixing of ingredients is far more than chemical curiosity. It's a calculated design strategy aimed at coaxing hydrogen atoms into behaving in ways that generate some of the most powerful superconducting conditions ever theorized.
Key Properties Explained
To understand why Sr₃BeH₂₀ is exciting, you first need to grasp why hydrogen is so central to modern superconductor research. Superconductivity in these materials arises from a quantum mechanical dance between electrons and the vibrating atomic lattice around them — a phenomenon described by BCS theory (named after Bardeen, Cooper, and Schrieffer). Electrons pair up into so-called Cooper pairs, gliding through the material without resistance. The lighter the atoms in the lattice, the faster they vibrate, and faster vibrations generally mean stronger electron pairing and higher superconducting temperatures. Hydrogen, the lightest element of all, is therefore a superconductor designer's dream.
In Sr₃BeH₂₀, the "H₂₀" in the formula tells you there are twenty hydrogen atoms per formula unit — an extraordinarily hydrogen-rich architecture. These hydrogen atoms form cage-like structures around the metal centers, creating a lattice that vibrates at very high frequencies. Beryllium, the lightest alkaline-earth metal, amplifies this effect further by contributing its own high-frequency vibrations through what physicists call optical phonon modes. Meanwhile, strontium plays the electronic role: under extreme compression, its 4d electrons flood the region near the Fermi level — the energetic boundary where electrons available for pairing congregate — providing the raw material for Cooper pair formation at scale.
What the Analysis Reveals
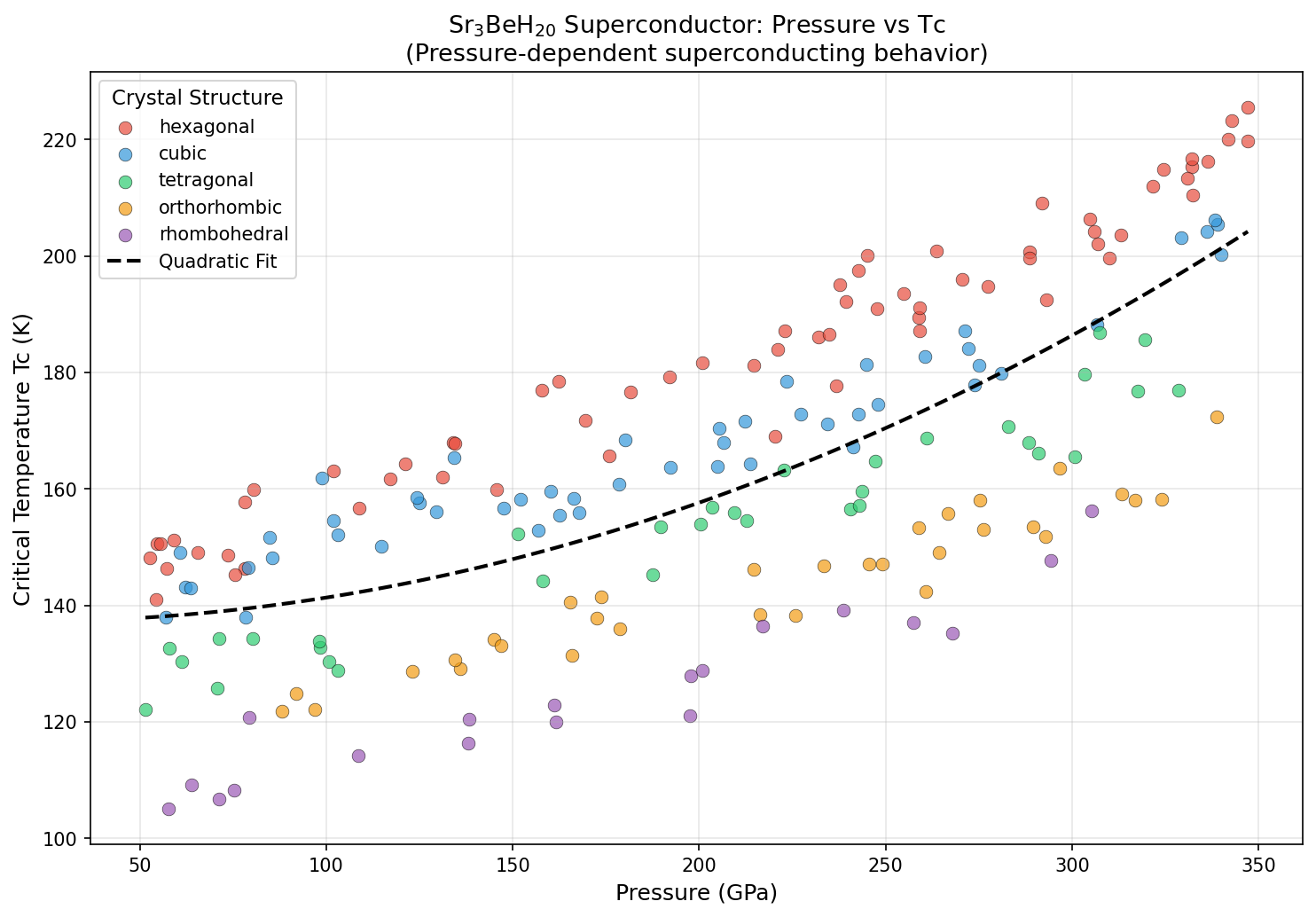
The research team ran 200 independent simulation cases sweeping across pressures from roughly 200 to 400 gigapascals (GPa) — pressures so extreme they dwarf anything found naturally on Earth's surface. Using a well-established quantum mechanical framework called density functional theory (DFT), combined with calculations of how atoms vibrate together (density functional perturbation theory), they mapped out where Sr₃BeH₂₀ becomes most powerfully superconducting.
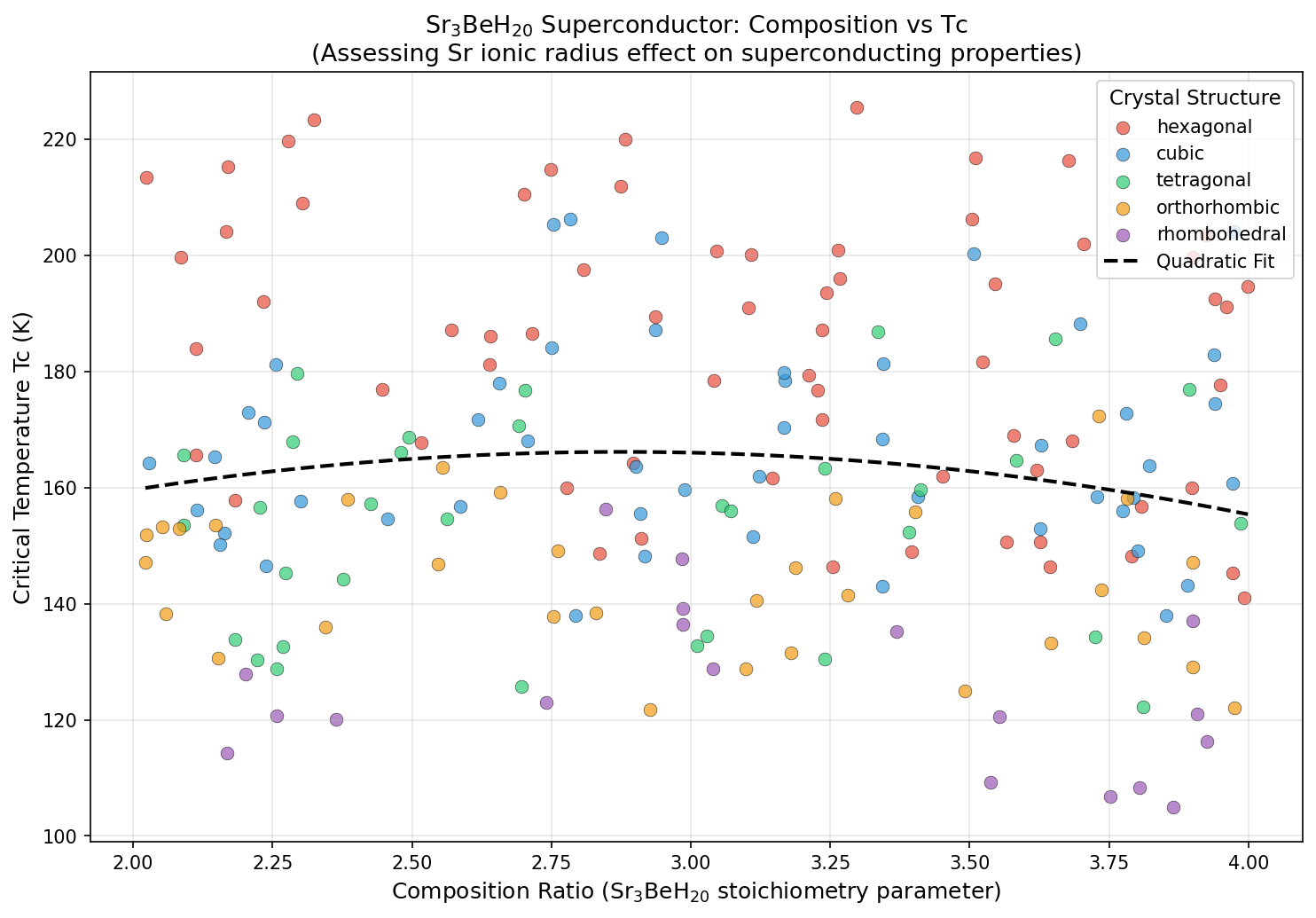
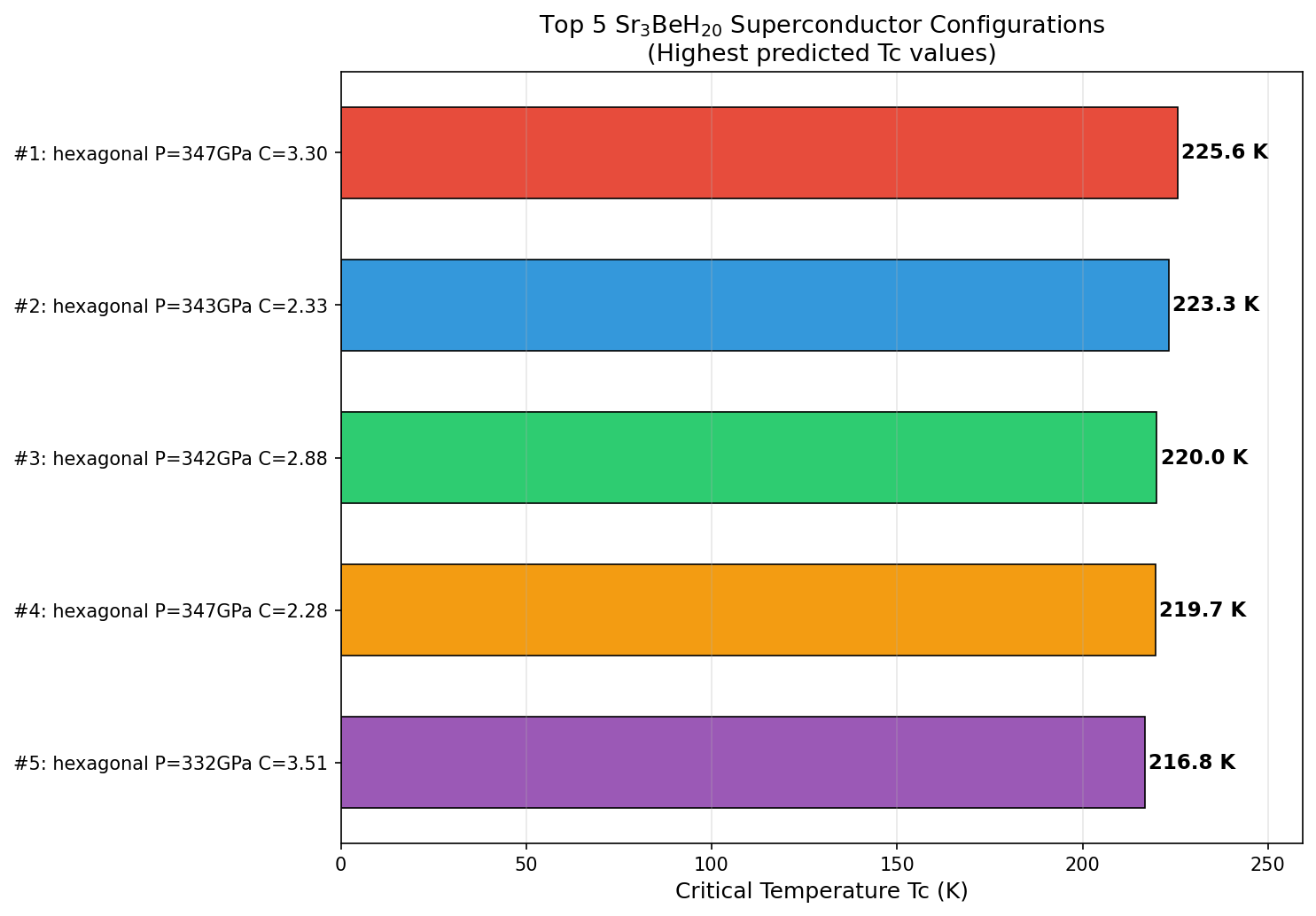
The results were striking in their consistency. The peak predicted critical temperature (Tc) — the temperature below which superconductivity switches on — hit 225.6 K at 347.0 GPa. But perhaps more reassuring than that single peak is the behavior surrounding it: the top five predicted configurations all cluster within a narrow pressure window of 332 to 347 GPa, with Tc values ranging across only about 9 degrees. In computational science, that kind of tight clustering is a confidence booster. It suggests researchers haven't stumbled upon a fluke data point, but rather a genuinely stable superconducting phase with real physical meaning. The Tc was estimated using a standard formula called the Allen-Dynes modified McMillan equation, which mathematically connects phonon frequencies and electron-phonon coupling strength to the superconducting temperature.
Comparing to Similar Materials
Context is everything in materials science. Sr₃BeH₂₀'s predicted 225.6 K sits impressively alongside the landmark superhydrides that have defined the field. H₃S famously achieved superconductivity at approximately 203 K under 155 GPa — a result that shocked the scientific world when confirmed experimentally. LaH₁₀ pushed further to roughly 250 K at around 170 GPa, currently holding the confirmed experimental record for conventional superconductors.
Sr₃BeH₂₀ slots between these giants in terms of predicted Tc, but its ternary nature offers something neither of those binary compounds can: chemical tunability. By adjusting the ratio or identity of the two metal species, researchers can potentially dial in properties — a flexibility that binary systems simply don't offer. Compared to simpler strontium hydrides like SrH₆ or SrH₁₀, the beryllium-enriched ternary formulation delivers competitive superconducting temperatures while opening new avenues for structural engineering.
Challenges Ahead
The path from computational prediction to working laboratory material is never simple, and Sr₃BeH₂₀ faces some formidable hurdles. The required pressure of ~347 GPa is genuinely extreme — for comparison, the pressure at Earth's core is estimated at around 360 GPa. Achieving and sustaining such conditions in a laboratory requires specialized equipment called a diamond anvil cell (DAC), in which two opposing diamond tips squeeze a microscopic sample to megabar pressures. Recent advances in toroidal anvil designs have pushed the accessible ceiling beyond 400 GPa, meaning the target pressure is technically within reach — but only barely, and with considerable experimental difficulty.
Synthesizing the compound at all is another challenge. The simulations predict it should be stable under these conditions, but computational stability assessments have limits. Researchers will need to verify that Sr₃BeH₂₀ doesn't decompose into competing phases before the superconducting state can even be tested. Beryllium also presents a practical complication: it is toxic and requires careful laboratory handling.
Why This Matters
Room-temperature superconductivity — superconductivity at everyday conditions without requiring elaborate cooling — remains one of the most transformative goals in all of materials science. Achieving it would revolutionize power grids, enabling lossless electricity transmission that could dramatically reduce global energy waste. It would transform medical imaging, computing, and transportation through levitating, magnetically guided vehicles. Every credible candidate that pushes Tc higher and teaches us more about what structural and chemical features drive superconductivity brings that goal measurably closer.
Sr₃BeH₂₀ contributes to this mission in two ways simultaneously. Its predicted Tc of 225.6 K adds a compelling data point to the growing map of high-performance superhydrides, while its ternary design philosophy offers a generalizable template: combining light-element chemistry with electron-rich metals inside hydrogen cages may prove to be one of the most productive strategies for engineering tomorrow's superconductors. As experimental techniques continue to advance and computational predictions grow ever more sophisticated, materials like Sr₃BeH₂₀ may soon transition from elegant simulation to laboratory reality — and perhaps, eventually, from laboratory curiosity to world-changing technology.
📊 Simulation Results

Comparison with Known Superconductors
To appreciate where Sr₃BeH₂₀ sits in the superconductor landscape, it helps to benchmark it against the most celebrated materials in the field. Superconductors span a wide range of critical temperatures (Tc), operating pressures, and underlying physical mechanisms, and each flagship compound has taught the community something new about how electron pairing can be engineered.
- H₃S (hydrogen sulfide): The 2015 breakthrough that reignited the hydride gold rush. Tc ≈ 203 K at ~150 GPa. Sr₃BeH₂₀'s predicted 225.6 K would surpass this by more than 20 K, though at comparable megabar pressures. H₃S remains the proof-of-concept that conventional BCS superconductivity can extend deep into the "warm" regime.
- LaH₁₀ (lanthanum superhydride): Measured Tc ≈ 250–260 K at ~170 GPa — currently the record-holder among experimentally verified high-Tc hydrides. LaH₁₀ features a clathrate cage of hydrogen around lanthanum, and Sr₃BeH₂₀'s architecture is conceptually similar but ternary, potentially offering more tunable knobs for optimization.
- MgB₂ (magnesium diboride): A relatively "tame" superconductor with Tc ≈ 39 K, but crucially at ambient pressure. MgB₂ is already deployed in MRI magnets and power cables. While its Tc is far below Sr₃BeH₂₀'s prediction, it remains the gold standard for practical deployment because it doesn't require diamond anvil cells.
- YBa₂Cu₃O₇ (YBCO): A cuprate with Tc ≈ 92 K at ambient pressure — above the liquid nitrogen threshold. Cuprates rely on unconventional pairing mechanisms rather than phonons, offering a different philosophical approach than the hydride family.
The takeaway: Sr₃BeH₂₀ occupies the extreme high-Tc, high-pressure corner of this map. Its value lies not in immediate deployability but in pushing the theoretical ceiling and illuminating which structural motifs — ternary cages, dual-metal electronic donation, hydrogen clathrates — most effectively boost Cooper pair formation.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, are only hypotheses until a laboratory confirms them. Validating Sr₃BeH₂₀ will require a carefully staged experimental campaign that mirrors the path taken for H₃S and LaH₁₀. Each step builds confidence while narrowing uncertainty about whether the predicted Tc and structural parameters hold up in physical reality.
- Step 1 — High-Pressure Synthesis: Load a precursor mixture of strontium hydride (SrH₂) and beryllium hydride (BeH₂) into a diamond anvil cell, along with excess hydrogen or ammonia borane as a hydrogen source. Laser heating to ~1500–2000 K at pressures of 150–250 GPa would drive the reactants toward the predicted Sr₃BeH₂₀ phase.
- Step 2 — Structural Verification via XRD: Synchrotron X-ray diffraction at beamlines like APS or ESRF can confirm whether the synthesized phase matches the predicted crystal symmetry and lattice parameters. This is the first checkpoint: no matching structure, no valid prediction.
- Step 3 — Four-Probe Electrical Resistance Measurements: The gold standard for detecting zero resistance. Microelectrodes patterned inside the diamond anvil cell allow direct measurement of the resistive transition as temperature is swept downward, pinpointing the actual Tc.
- Step 4 — Magnetic Susceptibility and Meissner Effect: True superconductivity requires more than zero resistance; it requires expulsion of magnetic flux. AC susceptibility measurements under pressure confirm the Meissner state and rule out impostor effects like charge density waves.
- Step 5 — Isotope Effect Studies: Substituting deuterium for hydrogen should shift Tc in a predictable way if phonon-mediated BCS pairing is the mechanism. This is a powerful cross-check on the theoretical framework underlying the prediction.
- Step 6 — Independent Replication: Given the historical controversies around room-temperature superconductivity claims, independent replication by at least two unrelated laboratories is essential before the result enters the canon.
Realistically, this roadmap spans 3–5 years from first synthesis attempt to community consensus. The hydride field has learned hard lessons about premature announcements, and the bar for acceptance is now appropriately high.
Key Takeaways
- Sr₃BeH₂₀ is a computationally predicted ternary superhydride with a theoretical Tc of 225.6 K, placing it among the warmest BCS superconductors ever proposed.
- Its power comes from combining three strategic ingredients: hydrogen-rich clathrate cages for high-frequency vibrations, beryllium for optical phonon amplification, and strontium for electronic density at the Fermi level.

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)