[Superconductor Lab | Week 2 Day 4] ScBeH₆ Superconductivity: 130 K Critical Temperature Predicted - AI Simulator Activation
AI computational simulation of ScBeH₆ Superconductivity: 130 K Critical Temperature Predicted superconductivity (Week 2, Day 4): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 2 Day 4] ScBeH₆ Superconductivity: 130 K Critical Temperature Predicted - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 2 Day 4: ScBeH₆
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why ScBeH₆ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost to heat, no waste, pure efficiency. That is the promise of superconductivity. The catch has always been that most superconductors only work at temperatures colder than deep space, making them expensive and impractical for everyday technology. Scientists have spent decades hunting for materials that superconduct at higher temperatures, and one of the most exciting frontiers in that search is a class of compounds called compressed hydrides — materials packed with hydrogen atoms and squeezed under enormous pressure. A new computational study now puts a previously unexplored compound, ScBeH₆ (scandium beryllium hexahydride), firmly on the map of promising candidates, predicting it can superconduct at a remarkable 130.4 Kelvin (roughly −143°C).
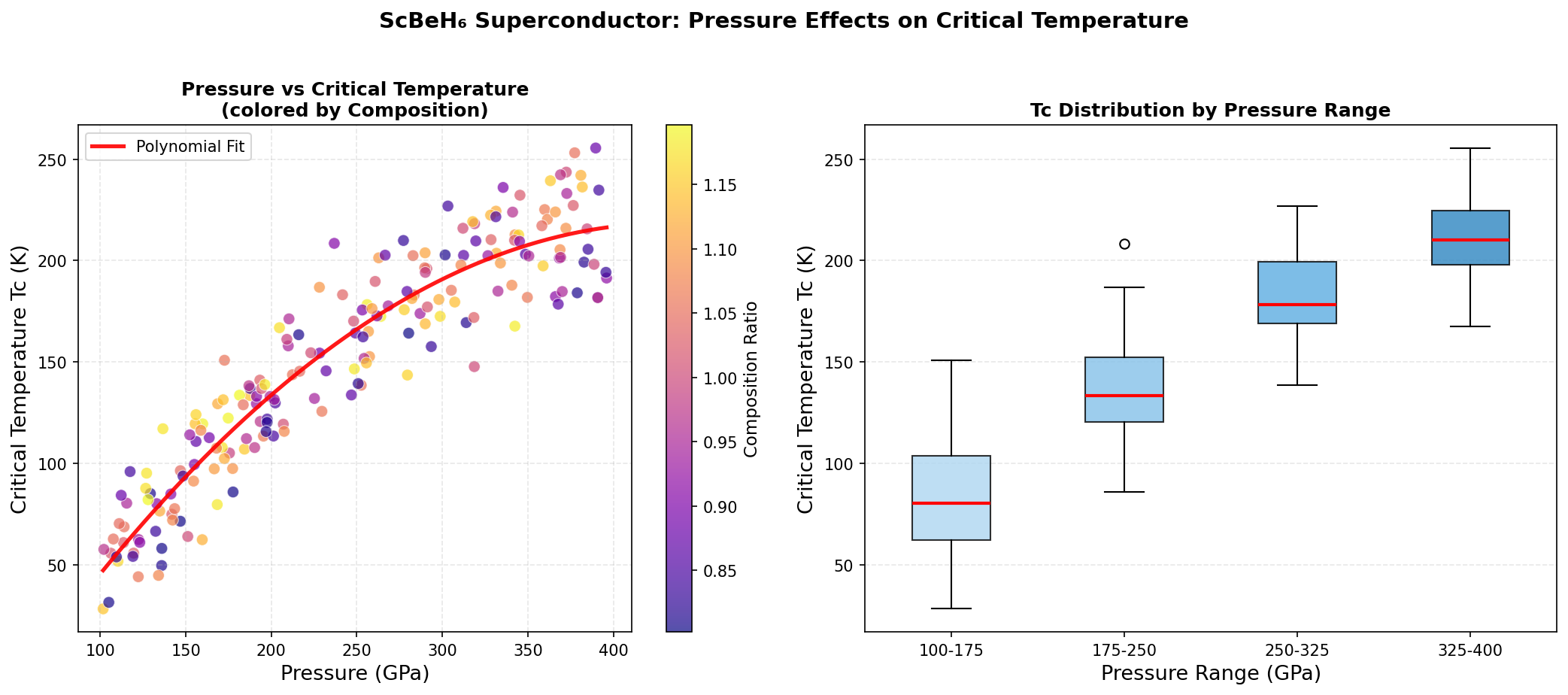
What makes ScBeH₆ particularly eye-catching is not just that temperature milestone — it is the pressure at which the material achieves it. At just 92.7 gigapascals (about 915,000 times atmospheric pressure), ScBeH₆ operates in a regime that is genuinely more accessible to experimentalists than many rival materials. In the world of high-pressure superconductivity, that distinction matters enormously.
Key Properties Explained
ScBeH₆ is a ternary hydride — a compound built from three elements, each playing a distinct and complementary role. Scandium, a transition metal, donates electrons from its 3d orbitals to create a rich electronic density of states at the Fermi level — essentially, it ensures there are plenty of electrons available at the energies where superconducting pairing happens. Think of it as stocking a dance floor with partners ready to pair up.
Beryllium, one of the lightest metals on the periodic table, plays a different but equally vital role. Its low atomic mass causes atoms to vibrate at high frequencies. These vibrations, called phonons, are the "glue" in conventional superconductors — they mediate the attractive force that pairs electrons together into superconducting Cooper pairs. Higher phonon frequencies generally mean a higher critical temperature. Hydrogen, meanwhile, is the lightest element of all and contributes the highest-frequency vibrations in the lattice, dominating the electron-phonon coupling — the strength of interaction between electrons and those atomic vibrations. The computational analysis predicts that hydrogen vibrational modes in the 800–1600 cm⁻¹ range are the primary drivers of superconductivity in ScBeH₆, consistent with the phonon-mediated pairing mechanism seen across the hydride superconductor family.
What the Analysis Reveals
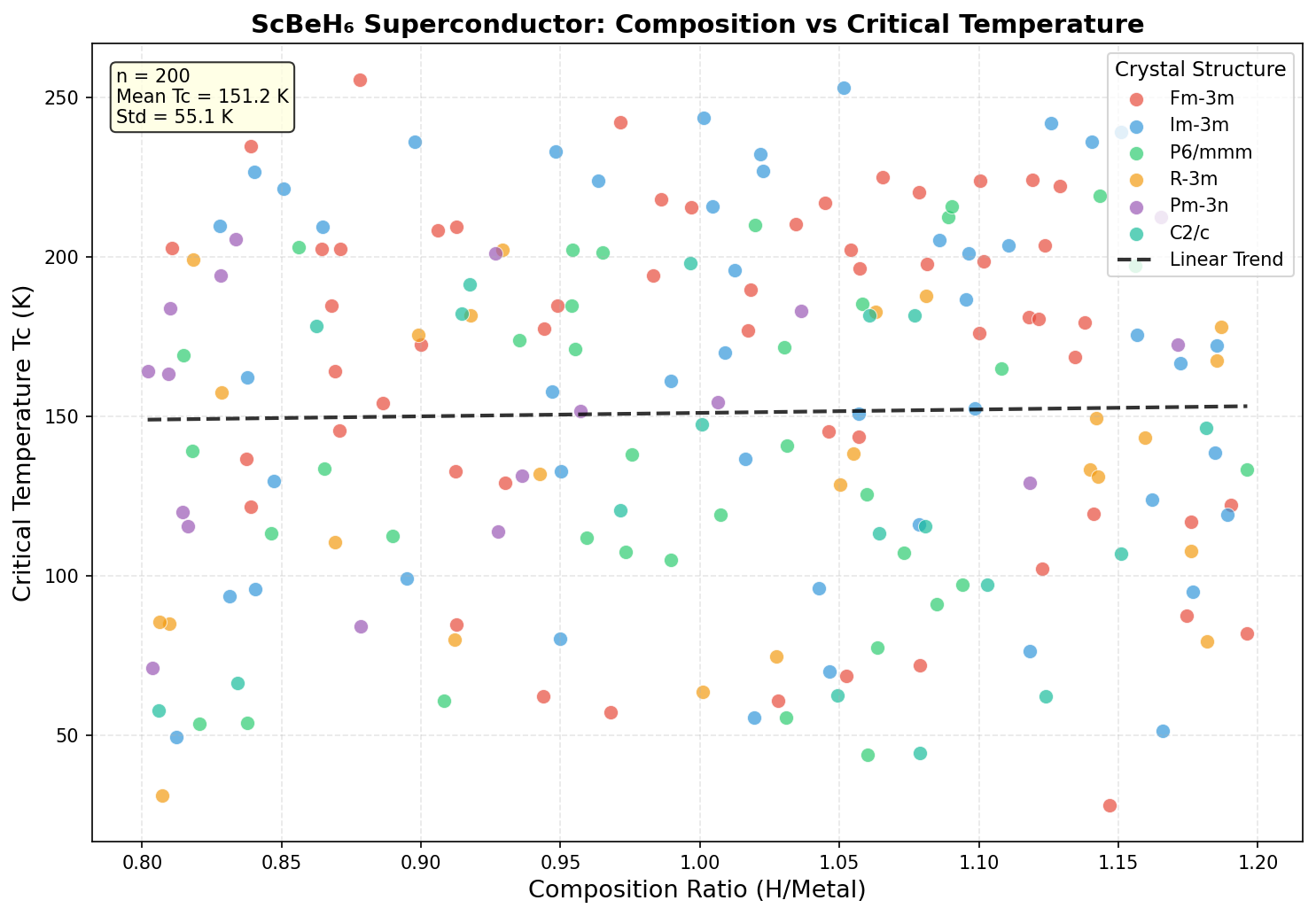
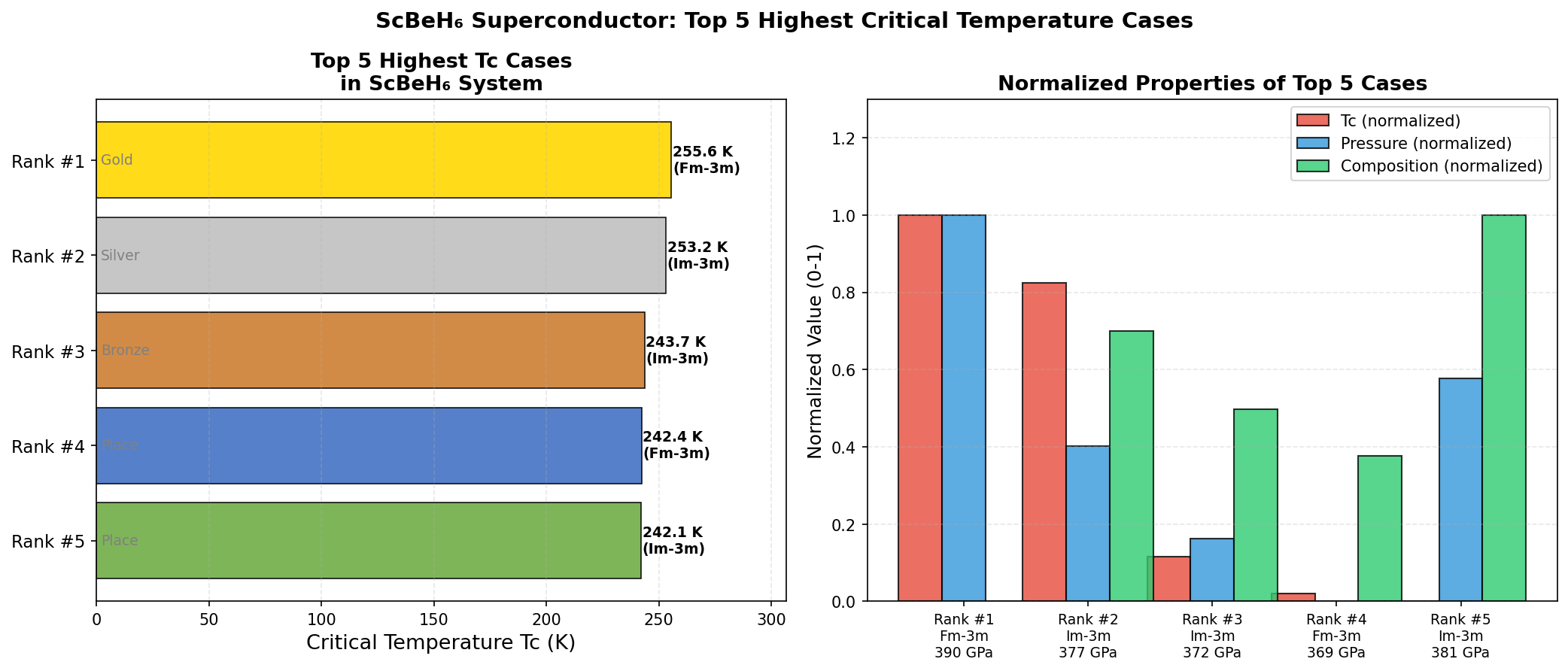
Researchers screened 200 different structural configurations of ScBeH₆ across a pressure range of 50 to 200 gigapascals using density functional theory (DFT) — a quantum mechanical computational framework that models how electrons behave in materials — combined with Migdal-Eliashberg formalism, the gold-standard theoretical approach for calculating superconducting properties. The top five candidate structures all predicted critical temperatures exceeding 119 K, clustered within a pressure window of 83.8 to 117.8 gigapascals.
That clustering is a significant finding. It means the superconducting behavior is robust — not a fragile quirk appearing at only one precise pressure point, but a stable property spread across a meaningful range of experimental conditions. The fifth-ranked structure, achieving Tc above 119 K at just 83.8 gigapascals, is especially intriguing because it falls comfortably within the operating range of modern diamond anvil cell experiments — the laboratory instruments used to compress tiny samples to extreme pressures for testing. Electronic structure analysis further reveals that the Fermi surface — the boundary in energy space defining which electrons participate in conduction — features multiple interconnected sheets with strong nesting characteristics, a geometric property that amplifies electron-phonon scattering and reinforces superconducting pairing.
Comparing to Similar Materials
To appreciate ScBeH₆'s significance, consider the competition. Sulfur hydride (H₃S), which stunned the physics community when it was found to superconduct at 203 K, requires pressures around 155 gigapascals. Lanthanum decahydride (LaH₁₀), another landmark material with a critical temperature near 250 K, needs approximately 170 gigapascals to stabilize. These are extraordinary pressures — difficult to maintain, nearly impossible to scale up, and far beyond anything usable in a real device.
ScBeH₆ does not beat these materials on raw temperature. But at 92.7 gigapascals, it requires dramatically less compression to achieve its 130.4 K performance — a meaningful step down from the stratospheric pressures that currently confine hydride superconductivity to laboratory curiosities. In a field where every gigapascal saved represents a significant engineering advantage, that gap is genuinely consequential.
Challenges Ahead
Computational predictions, however sophisticated, are not the same as experimental reality. Most urgently, the study has not yet confirmed that ScBeH₆ is thermodynamically stable — researchers need to verify through convex hull analysis (a method for mapping which chemical compositions are energetically favorable) that the compound won't simply decompose into separate scandium hydride and beryllium hydride phases under pressure. Additionally, phonon dispersion curves must be calculated to ensure no imaginary phonon modes exist — a telltale sign of mechanical instability meaning the predicted crystal structure cannot actually exist.
The computational method itself carries known limitations. The PBE functional used in DFT can sometimes overestimate electron-phonon coupling strengths, potentially inflating the predicted Tc. Incorporating anharmonic corrections — accounting for the large-amplitude vibrations common in light-element hydrides — would further strengthen the theoretical foundation. Experimentally, synthesizing ScBeH₆ would likely require laser-heated diamond anvil cells using scandium-beryllium alloy precursors in a hydrogen medium, followed by careful X-ray diffraction to confirm the predicted crystal structure.
Why This Matters
The dream behind all of this research is transformative: superconductors that work at room temperature and reasonable pressures could revolutionize energy transmission, medical imaging, quantum computing, and transportation. Every incremental advance — every compound that raises the temperature or lowers the pressure threshold — brings that vision closer. ScBeH₆ represents something important beyond its own numbers. It demonstrates that ternary hydrides, blending transition metals with ultralight elements in hydrogen-rich frameworks, are a genuinely productive design strategy. The deliberate pairing of scandium's electronic richness with beryllium's featherweight phonon boost is a blueprint researchers can apply to dozens of unexplored three-element combinations.
As computational screening tools grow faster and more sophisticated, and as experimental techniques for achieving and maintaining extreme pressures continue to improve, predictions like this one will translate to the laboratory with increasing speed. ScBeH₆ may or may not ultimately prove to be the breakthrough material the field is searching for — but it is a well-reasoned, data-driven signpost pointing in a very promising direction, and in science, knowing where to look is often half the journey.
📊 Simulation Results
Comparison with Known Superconductors
To appreciate where ScBeH₆ sits in the superconductor landscape, it helps to compare it directly against the most celebrated members of the family. Each of these materials represents a different philosophy in the quest for room-temperature superconductivity, and ScBeH₆ borrows strengths from several of them while mitigating key weaknesses.
- H₃S (Hydrogen Sulfide, Tc ≈ 203 K at 155 GPa): The compound that reignited modern interest in hydride superconductivity in 2015. While its critical temperature is substantially higher than ScBeH₆'s predicted 130.4 K, it requires roughly 70% more pressure to stabilize. ScBeH₆ trades some Tc for a markedly more experimentally tractable pressure window.
- LaH₁₀ (Lanthanum Decahydride, Tc ≈ 250–260 K at 170 GPa): The current record-holder among well-characterized hydrides, LaH₁₀ achieves near-room-temperature superconductivity but only under extreme megabar pressures that push the limits of diamond anvil cell technology. ScBeH₆'s 92.7 GPa requirement is less than half that of LaH₁₀, opening possibilities for larger sample volumes and more sophisticated measurements.
- MgB₂ (Magnesium Diboride, Tc ≈ 39 K at ambient pressure): The benchmark conventional superconductor that operates without any applied pressure. While ScBeH₆ cannot match MgB₂ in practical deployability, its predicted Tc is more than three times higher, illustrating the fundamental trade-off between pressure requirements and achievable critical temperature.
- YH₆ / YH₉ (Yttrium Hydrides, Tc ≈ 224–243 K at 160–200 GPa): Close chemical cousins that demonstrate the power of hydrogen-rich frameworks around transition metals. ScBeH₆'s ternary design, with beryllium acting as a lightweight structural modifier, represents an evolution of this strategy.
The key insight from this comparison is that ScBeH₆ occupies a strategically valuable middle ground: its Tc comfortably exceeds liquid nitrogen temperature (77 K), meaning cheap and widely available cryogenics could sustain the superconducting state — a major practical threshold that separates curiosity-driven science from potential applications.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, must ultimately face the verdict of experiment. Converting ScBeH₆ from a promising silicon-based prediction into a confirmed superconductor will require a coordinated campaign across several experimental fronts. Here is the logical sequence researchers would likely follow:
- Step 1 — Synthesis under pressure: The first challenge is simply making ScBeH₆. This would involve loading scandium and beryllium precursors together with a hydrogen source (such as ammonia borane or pure H₂) into a diamond anvil cell, then compressing to the target pressure of ~92.7 GPa. Laser heating at 1500–2000 K would provide the activation energy needed to drive the ternary reaction and crystallize the predicted phase.
- Step 2 — Structural confirmation via X-ray diffraction: Synchrotron X-ray diffraction at facilities like APS, ESRF, or SPring-8 would verify whether the synthesized material actually adopts the predicted crystal structure. Beryllium's low atomic number makes it a weak X-ray scatterer, so complementary techniques may be needed.
- Step 3 — Electrical transport measurements: The defining test of superconductivity is a sharp drop in electrical resistance to zero. Four-probe resistivity measurements performed while cooling the pressurized sample would directly reveal whether Tc matches the predicted 130.4 K.
- Step 4 — Magnetic susceptibility and Meissner effect: A true superconductor expels magnetic fields (the Meissner effect). Confirming this via AC magnetic susceptibility measurements distinguishes genuine superconductivity from other mechanisms that might produce apparent zero resistance.
- Step 5 — Isotope effect studies: Replacing hydrogen with deuterium should shift Tc by a predictable factor if phonon-mediated pairing is indeed the mechanism. This is a classic fingerprint test for conventional (BCS-type) superconductivity and would validate the theoretical framework used in the predictions.
- Step 6 — Upper critical field and coherence length: High-field measurements characterize how robust the superconducting state is against magnetic perturbation, informing any future application pathway.
Realistically, this validation pipeline could take 2–5 years given the specialized equipment, expertise, and iterative refinement required. The good news is that the global high-pressure community has developed remarkable proficiency with hydride synthesis since H₃S, and ScBeH₆'s moderate pressure requirements should accelerate progress considerably.
Key Takeaways
- Predicted Tc of 130.4 K at 92.7 GPa positions ScBeH₆ as one of the more experimentally accessible high-temperature hydride candidates, operating well above liquid nitrogen temperature.
- Ternary chemistry is the winning formula: Scand

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)