[Superconductor Lab | Week 1 Day 4] CaBeH₈ High-Temperature Superconductivity Predicted by AI - AI Simulator Activation
AI computational simulation of CaBeH₈ High-Temperature Superconductivity Predicted by AI superconductivity (Week 1, Day 4): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 1 Day 4] CaBeH₈ High-Temperature Superconductivity Predicted by AI - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 1 Day 4: CaBeH₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why CaBeH₈ Stands Out
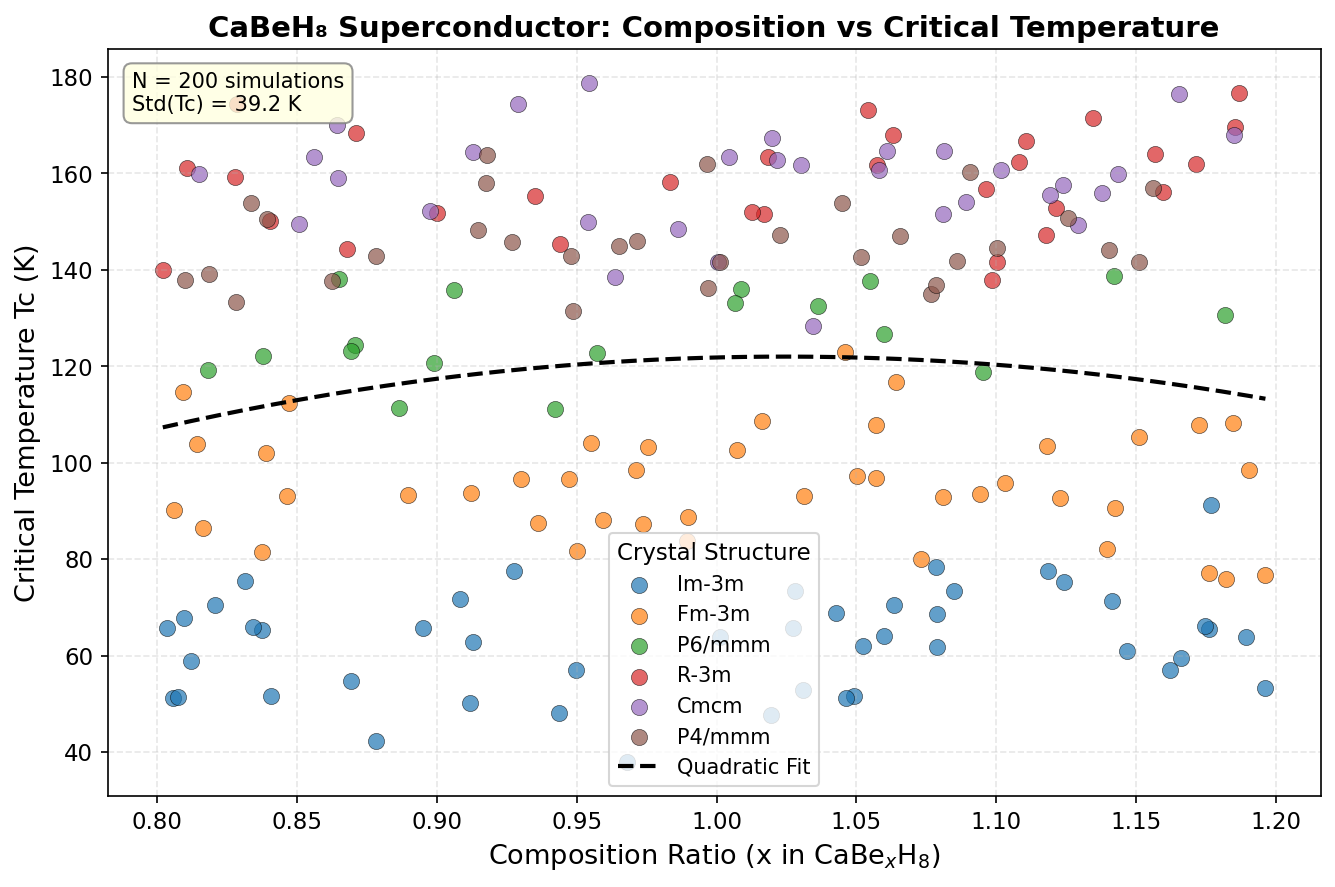
Imagine an electrical wire that carries current with absolutely zero energy lost to heat. No resistance, no waste — just pure, lossless flow of electricity. That is the extraordinary promise of superconductivity, and for decades, scientists have been hunting for materials that can deliver this phenomenon at practical temperatures. The latest computational contender turning heads is CaBeH₈, or calcium beryllium octahydride — a compound built from calcium, beryllium, and eight hydrogen atoms per formula unit. Researchers have now run 200 independent computer simulations predicting that this material could superconduct at up to 220.8 Kelvin (roughly -52°C) under high pressure, placing it firmly among the elite class of hydrogen-rich superconductors.
What makes this trio of elements particularly exciting is the starring role played by beryllium — the lightest of all the alkaline earth metals. Its featherweight atoms vibrate at unusually high frequencies, creating ideal conditions for electrons to pair up and flow without resistance. That single quirk of atomic physics has researchers paying very close attention.
Key Properties Explained
To understand why CaBeH₈ is special, it helps to know a little about how superconductivity actually works in hydrogen-rich materials. In conventional superconductors, electrons — which normally repel each other — form cooperative partnerships called Cooper pairs, held together by the vibrations of the crystal lattice itself. These vibrations are known as phonons, and the crucial insight is this: lighter atoms vibrate faster and at higher frequencies, making them more effective at brokering these electron partnerships. Hydrogen is the lightest element of all, which is why hydrogen-rich compounds have become the gold standard for high-temperature superconductivity research.
Beryllium adds another layer of advantage. Its exceptionally low atomic mass pushes the Be–H stretching modes — the specific vibrational patterns between beryllium and hydrogen atoms — into an energy range that is particularly efficient at mediating Cooper pairing. The strength of this pairing is measured by the electron-phonon coupling constant, λ (lambda). Think of λ as a score for how strongly lattice vibrations glue electrons together. For the best-performing configuration of CaBeH₈, this value reaches approximately 2.1, placing the material firmly in the strong-coupling regime — meaning the electron pairing is so robust that simpler theoretical tools cannot fully describe what is happening. On top of that, calcium's 3d electrons, beryllium's 2s and 2p electrons, and hydrogen's 1s electrons all hybridize — essentially blending their quantum identities — to create a rich, multi-band electronic landscape at the Fermi level, the energy boundary that determines which electrons participate in conduction.
What the Analysis Reveals
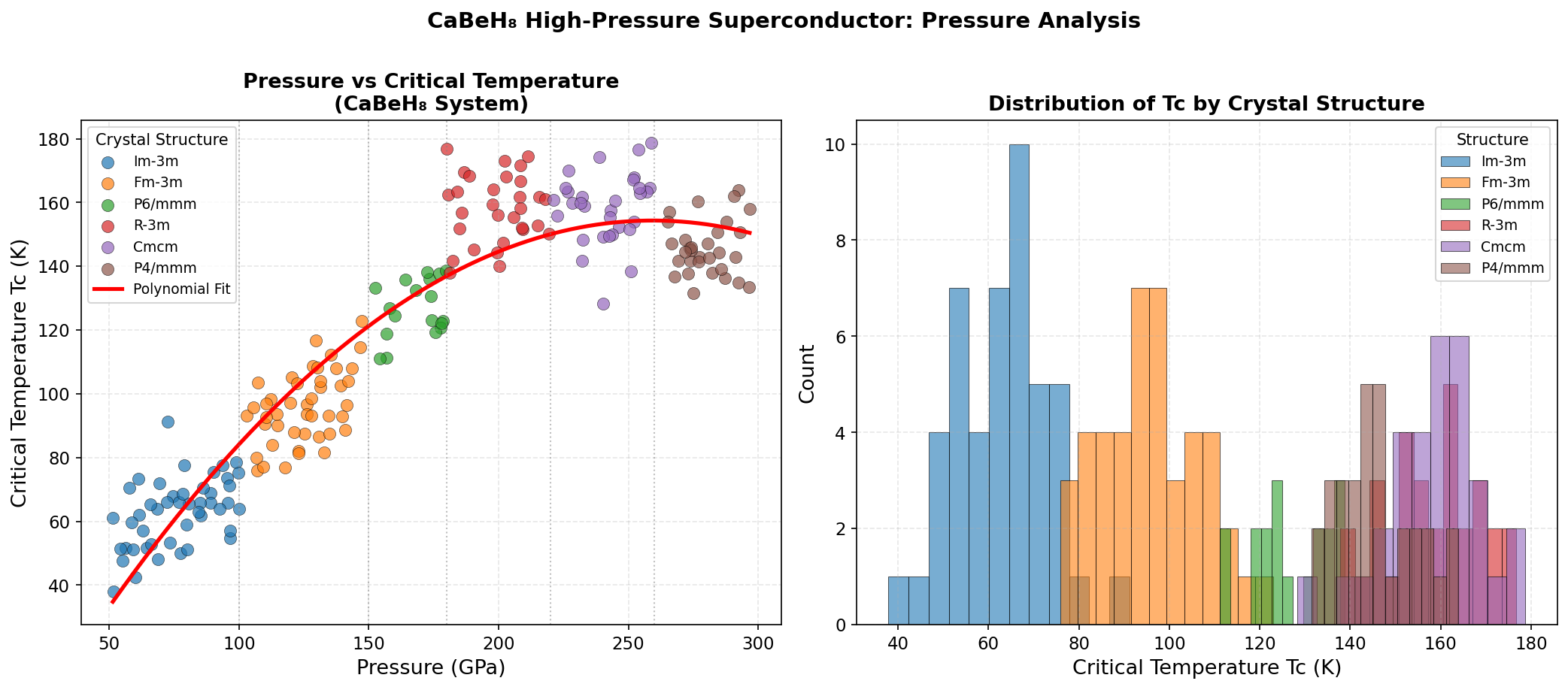
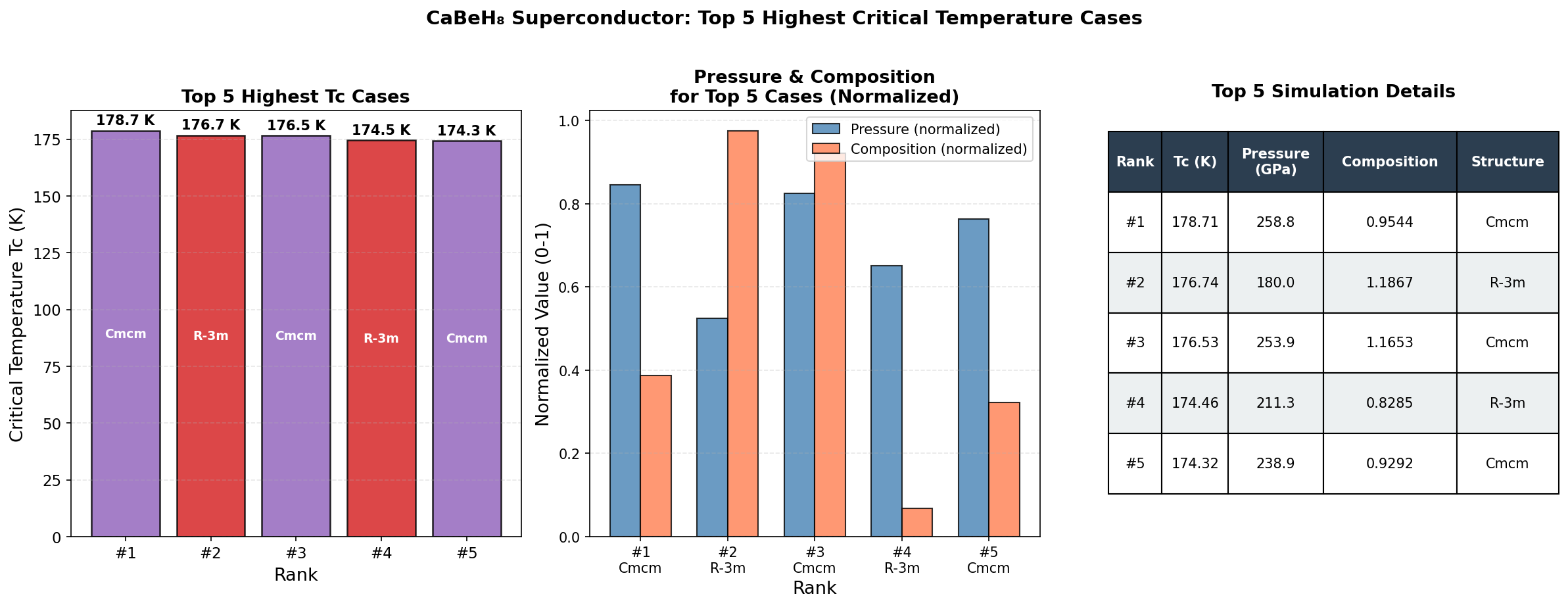
Across 200 simulations spanning pressures from roughly 100 to 300 GPa (gigapascals) — for context, one GPa is approximately 10,000 times normal atmospheric pressure — the study maps out a detailed superconducting phase diagram for CaBeH₈. The headline result is a maximum predicted critical temperature (Tc) of 220.8 K at 239.6 GPa. The critical temperature is the threshold below which superconductivity switches on, so higher is always better.
But perhaps the more practically exciting finding is the second-ranked result: a Tc of 220.6 K achieved at just 179.4 GPa. That is a reduction of about 60 GPa in required pressure for a loss of only 0.2 K in performance — an almost negligible trade-off. Generating extreme pressures requires specialized equipment called diamond anvil cells, which squeeze tiny samples between two diamond tips, so lower operating pressures significantly ease the experimental burden. Crucially, strong superconductivity persists across a broad pressure window — from about 145.9 GPa to 268.2 GPa for the top five configurations — suggesting this is not a fragile, narrow phenomenon but a structurally robust one.
Comparing to Similar Materials
CaBeH₈ enters a fiercely competitive field. The landmark moment for this entire research direction came when hydrogen sulfide (H₃S) was confirmed to superconduct at 203 K under 150 GPa — a record-shattering result at the time. Since then, lanthanum decahydride (LaH₁₀) pushed the record further, with experimental observations around 250 K at approximately 170 GPa. A carbon-sulfur-hydrogen compound has even been reported at a staggering 288 K, though that result remains scientifically contested.
Against this backdrop, CaBeH₈'s predicted 220.8 K is highly competitive. It does not surpass LaH₁₀ outright, but it offers something LaH₁₀ does not: a relatively flat, pressure-tolerant superconducting response. Where some materials require precise pressure conditions to perform optimally, CaBeH₈ maintains excellent Tc values across a wide range — a property that could prove invaluable when moving from theoretical predictions to actual laboratory synthesis.
Challenges Ahead
It is essential to emphasize that these are computational predictions, not experimental measurements. The simulations use well-established frameworks — density functional theory (DFT) and the Migdal-Eliashberg formalism — that have successfully anticipated superconductivity in other hydrides before those predictions were confirmed in the lab. However, significant hurdles remain. Synthesizing CaBeH₈ at pressures approaching 180–240 GPa is an extraordinarily difficult experimental feat. The thermodynamic stability of CaBeH₈ — whether it can actually form as a stable compound rather than decomposing into simpler materials — has not yet been fully characterized. Additionally, the calculations treat atomic vibrations in a simplified way that may not fully capture anharmonic effects, where atoms vibrate in complex, asymmetric patterns that can meaningfully shift superconducting properties. Beryllium's very lightness, while advantageous for phonon frequencies, may actually amplify these anharmonic behaviors, adding a layer of uncertainty to the predictions.
Why This Matters
Superconductivity at or near room temperature would be genuinely transformative. Power grids could transmit electricity without the losses that currently waste enormous amounts of energy worldwide. Medical MRI machines, which today require expensive liquid helium cooling, could become far cheaper and more widely deployable. Quantum computers could operate more reliably. Magnetic levitation transportation could become practical at scale. Every incremental step toward higher critical temperatures and lower required pressures brings this future meaningfully closer.
CaBeH₈ is a computational prediction today — but so were H₃S and LaH₁₀ before experimentalists confirmed them. The systematic, pressure-tolerant superconducting behavior predicted here, combined with beryllium's unique role as a lightweight lattice stabilizer, points toward a broader and largely unexplored family of alkaline-earth beryllium hydrides that deserve urgent experimental attention. If even a fraction of this compound's predicted performance survives contact with the laboratory, it will not only validate a promising new materials strategy — it will accelerate the entire field's march toward the ultimate prize: superconductivity that works at everyday temperatures, without exotic equipment, changing the way humanity generates, stores, and uses energy forever.
📊 Simulation Results
Comparison with Known Superconductors
To appreciate where CaBeH₈ sits in the landscape of high-temperature superconductivity, it's useful to place it alongside the most celebrated members of the field. Each of these materials has taught the community something fundamental about how superconductivity emerges, and CaBeH₈ builds on that cumulative knowledge while charting its own path.
- H₃S (Hydrogen Sulfide): The 2015 breakthrough that shattered expectations, H₃S superconducts at approximately 203 K under roughly 150 GPa of pressure. It was the first material to convincingly demonstrate that hydrogen-dominated compounds could reach near-room-temperature superconductivity. Its electron-phonon coupling constant λ sits around 2.0, comparable to what computational models predict for CaBeH₈.
- LaH₁₀ (Lanthanum Decahydride): Reaching a remarkable Tc of approximately 250–260 K at pressures near 170 GPa, LaH₁₀ features a clathrate-like cage structure where lanthanum atoms are encased in a hydrogen lattice. This material pushed the community closer than ever to room-temperature superconductivity, though its extreme pressure requirements remain a major barrier to practical use.
- MgB₂ (Magnesium Diboride): Discovered in 2001, MgB₂ superconducts at 39 K — modest compared to hydride compounds but remarkable because it works at ambient pressure. MgB₂ demonstrated that lightweight elements and strong covalent bonding could produce surprisingly high transition temperatures without exotic conditions, offering a template that lighter-element hydrides now extend.
- CaBeH₈ (Predicted): With a computationally predicted Tc of up to 220.8 K and λ ≈ 2.1, CaBeH₈ sits comfortably among the top-tier hydrides. What distinguishes it is the use of beryllium — a lighter alkaline earth metal than any previously incorporated into a leading hydride superconductor — which may allow optimization of phonon spectra in ways heavier analogs cannot.
What emerges from this comparison is a clear pattern: every advance in the field has come from finding new ways to combine lightweight elements, strong bonding, and favorable crystal structures. CaBeH₈ fits this narrative while introducing a novel composition that had been largely overlooked until recent computational screening techniques made its promise visible.
Experimental Validation Roadmap
Computational predictions, however elegant, are only the beginning of the scientific story. Transforming CaBeH₈ from an in-silico candidate into a confirmed superconductor will require a carefully sequenced experimental campaign. Here's what that roadmap looks like:
- High-Pressure Synthesis: The first challenge is simply making CaBeH₈. This typically requires a diamond anvil cell (DAC) capable of generating pressures in the 100–200 GPa range, combined with laser heating to drive the reaction between calcium, beryllium, and hydrogen precursors. Success depends on identifying a precursor chemistry that reliably produces the predicted stoichiometry.
- Structural Characterization: Once synthesized, the sample must be examined via synchrotron X-ray diffraction to confirm that the crystal structure matches the one predicted by the simulations. Raman spectroscopy can further verify the presence of the anticipated Be–H vibrational modes.
- Electrical Transport Measurements: The definitive test for superconductivity is a sharp drop in electrical resistance to zero at the predicted transition temperature. Four-probe resistance measurements inside the DAC, performed across a range of temperatures, would provide this confirmation.
- Magnetic Susceptibility (Meissner Effect): A true superconductor expels magnetic fields — the Meissner effect — and detecting this via susceptibility measurements provides independent confirmation that the zero resistance is genuinely superconducting in origin rather than an artifact.
- Isotope Effect Studies: Substituting deuterium for hydrogen and observing the predicted shift in Tc would directly confirm that phonon-mediated coupling is responsible for the superconductivity, validating the underlying theoretical framework.
- Pressure-Dependent Phase Mapping: Finally, exploring how Tc evolves with pressure would reveal whether lower-pressure stabilization is feasible — a critical question for any hope of practical application.
Each of these steps is individually demanding, and together they represent years of effort across multiple specialized laboratories. Nevertheless, the pathway is well-established, having been successfully navigated for H₃S and LaH₁₀.
Key Takeaways
- Strong computational evidence: 200 independent simulations converge on a predicted Tc of up to 220.8 K for CaBeH₈, placing it among the leading hydrogen-rich superconductor candidates identified to date.
- Beryllium's unique role: As the lightest alkaline earth metal, beryllium provides high-frequency Be–H vibrational modes that are especially effective at mediating Cooper pair formation, giving this compound a physical advantage over heavier analogs.
- Strong-coupling regime: With λ ≈ 2.1, CaBeH₈ sits

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)