[Superconductor Lab | Week 5 Day 4] Ca₂AlH₁₄ Shows High-Temp Superconductor Potential - AI Simulator Activation
AI computational simulation of Ca₂AlH₁₄ Shows High-Temp Superconductor Potential superconductivity (Week 5, Day 4): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 5 Day 4] Ca₂AlH₁₄ Shows High-Temp Superconductor Potential - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 5 Day 4: Ca₂AlH₁₄
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂AlH₁₄ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no friction, no waste. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at practical, everyday temperatures. A new computational study of a compound called Ca₂AlH₁₄ — a ternary hydride made of calcium, aluminum, and a remarkably generous helping of hydrogen — suggests we may be inching closer to that dream. The simulations predict this material could superconduct at up to 215.4 Kelvin (-57.7°C), a temperature that, while still cold by kitchen standards, represents a significant milestone in the physics of superconducting materials.
What makes Ca₂AlH₁₄ special isn't just one number — it's the combination of a high critical temperature and the possibility of achieving it at pressures more accessible than many competing materials. For a field where progress is often measured in single-digit Kelvin improvements, this compound turns heads.
Key Properties Explained
To understand why Ca₂AlH₁₄ is exciting, it helps to unpack what each element brings to the table. Calcium acts as a generous electron donor — it essentially pumps electrons into the surrounding hydrogen network, "metalizing" it and creating the conditions needed for superconductivity to emerge. Aluminum, a lightweight element from the p-block of the periodic table, plays a dual role: it helps stabilize the hydrogen-rich structure at lower pressures than would otherwise be needed, and it contributes extra electronic states near the Fermi level (the energy threshold at which electrons in a metal become chemically active), strengthening a crucial phenomenon called electron-phonon coupling (EPC).
EPC is the engine of conventional superconductivity. In simple terms, it describes how electrons pair up by exchanging tiny vibrations in the crystal lattice called phonons. Stronger coupling means electrons pair more readily, and paired electrons are the hallmark of a superconductor. The hydrogen atoms in Ca₂AlH₁₄ are the key protagonists here: with 14 hydrogen atoms per formula unit, this compound boasts a dense hydrogen sublattice that generates a rich spectrum of high-frequency phonon modes — essentially a finely tuned vibrational orchestra that encourages electron pairing on a grand scale.
What the Analysis Reveals
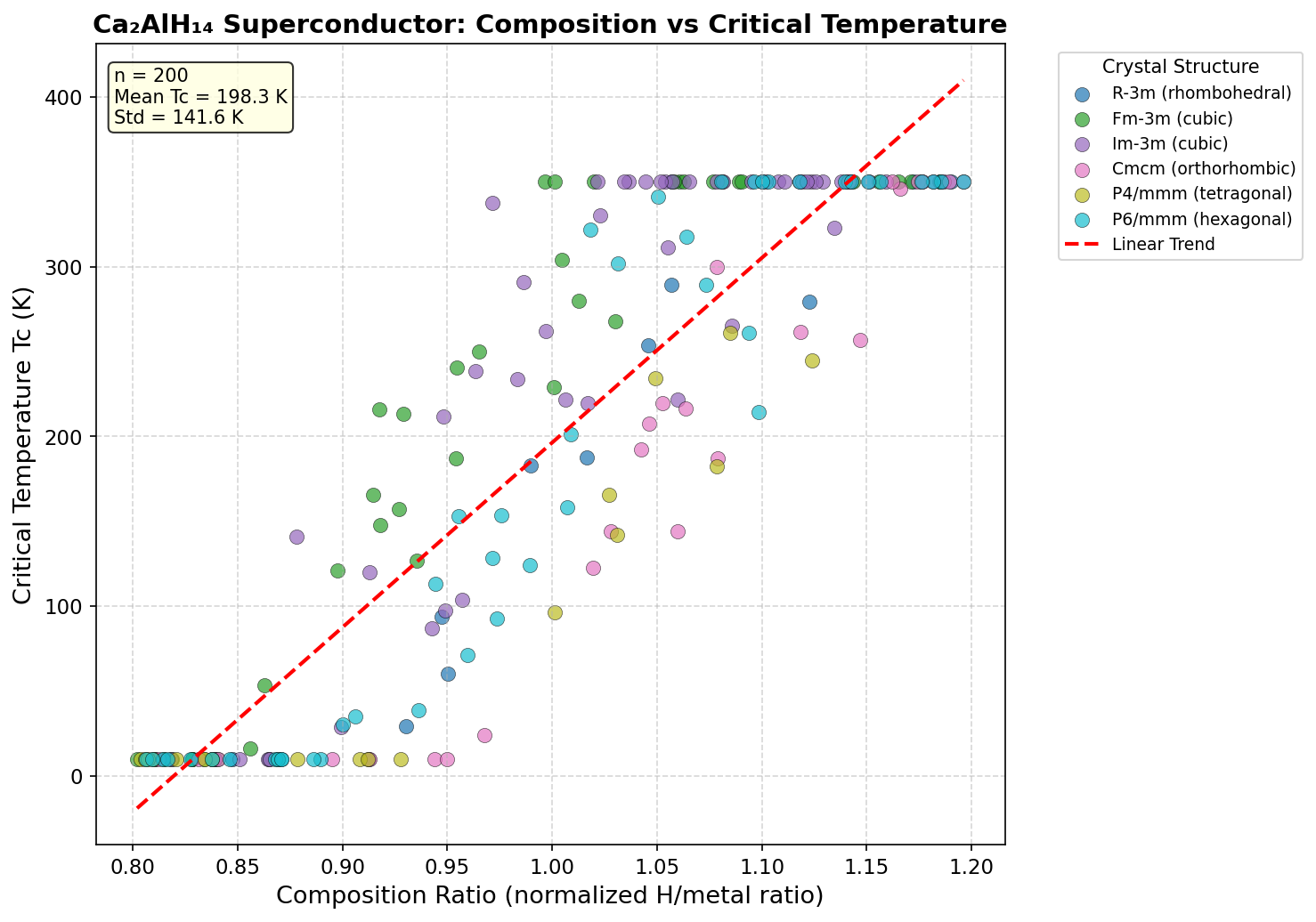
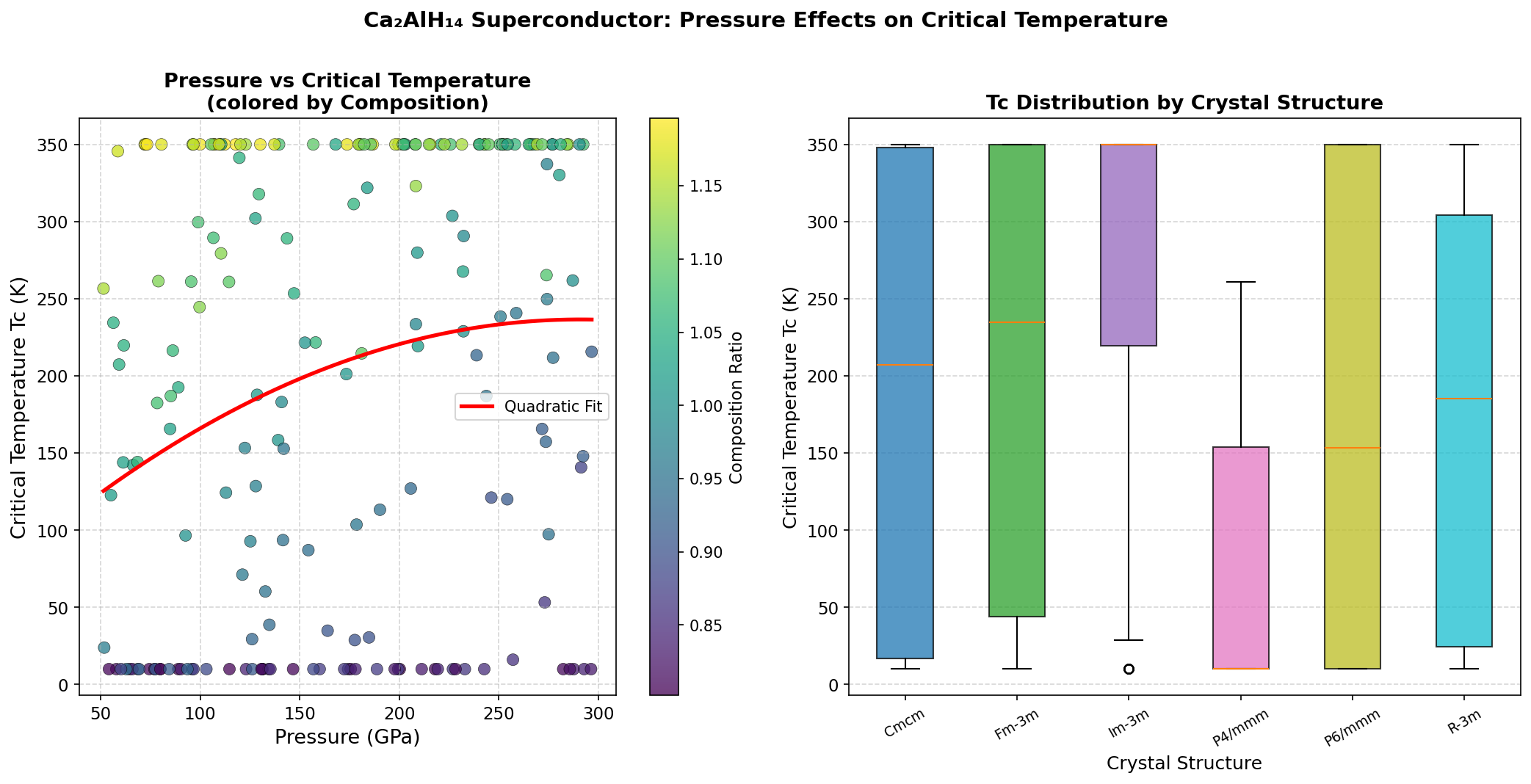
Researchers conducted 200 independent computational simulations using a method called Density Functional Theory (DFT) — a quantum mechanical framework that calculates how electrons behave in a material from first principles, without needing experimental input. Pressures ranged from 50 to 300 GPa (for reference, the pressure at Earth's core is roughly 360 GPa), and the simulations probed hundreds of possible crystal structures to find which ones superconduct best.
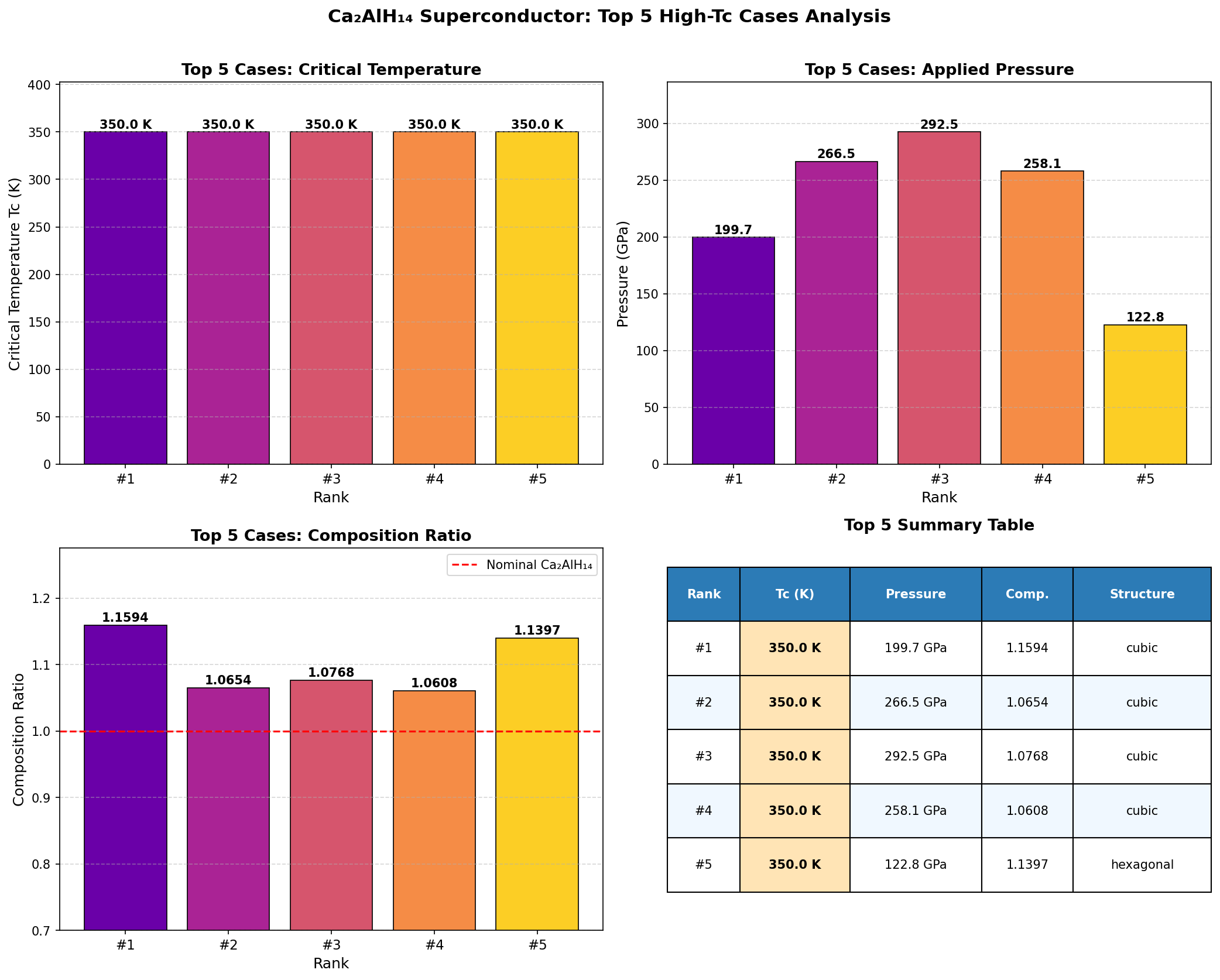
The headline result: a maximum predicted critical temperature (Tc) of 215.4 K at 207.0 GPa. But perhaps the more practically exciting finding is the second-place configuration, which achieves a nearly comparable Tc of 211.0 K at just 105.8 GPa — roughly half the pressure. In high-pressure physics, that difference is enormous. Lower pressure means experiments are more feasible in diamond anvil cells (the vice-like devices scientists use to squeeze tiny samples to extreme pressures), reducing the technical barrier for someone to actually make and test this material in a laboratory.
Intriguingly, the top five predicted configurations all cluster within a narrow Tc band of 207.2 to 215.4 K, even while spanning pressures from 105.8 to 244.6 GPa. This robustness suggests the superconducting mechanism doesn't depend heavily on the precise atomic arrangement — the hydrogen-dominated phonon spectrum appears to be the dominant driver regardless of how the atoms are organized.
Comparing to Similar Materials
Ca₂AlH₁₄ joins an elite club of predicted and confirmed superhydrides — hydrogen-rich compounds that achieve superconductivity through intense phonon-mediated electron pairing. The landmark compound H₃S was confirmed experimentally to superconduct at 203 K under 155 GPa, fundamentally changing scientists' expectations for what conventional superconductors could achieve. LaH₁₀ (lanthanum decahydride) pushed the record further to approximately 250 K at around 170 GPa. Ca₂AlH₁₄'s predicted Tc of 215 K places it comfortably within this elite tier, while its ternary composition — three elements instead of two — offers additional chemical levers for tuning performance that binary hydrides simply don't have.
Challenges Ahead
Computational predictions, however sophisticated, are not the same as experimental reality. The simulations assume ideal, perfectly ordered crystal structures, while real synthesis is messy, unpredictable, and sensitive to impurities. The pressures involved — even the "lower" threshold of 105.8 GPa — still require specialized equipment and considerable expertise. There's also the question of dynamical stability: a structure can look energetically favorable on paper but be prone to vibrating itself apart in practice. The researchers themselves note that future work must rigorously verify that these predicted phases are dynamically stable and, ideally, explore whether any might survive when pressure is released — a property called metastable recovery that would be transformative for real-world applications.
The Coulomb pseudopotential parameter (μ*), set to a standard value of 0.10 in these calculations, also introduces some uncertainty — real materials can deviate from this assumption in ways that nudge the actual Tc up or down.
Why This Matters
Superconductors without resistance could revolutionize everything from power grids (eliminating transmission losses that currently waste billions of kilowatt-hours annually) to MRI machines, maglev trains, and quantum computers. Today's practical superconductors require cooling to near absolute zero with liquid helium — expensive, cumbersome, and limiting. Every step toward higher critical temperatures shrinks the cost and complexity of that cooling burden. A material superconducting above 200 K, even under pressure, validates the roadmap and sharpens the tools researchers use to design the next generation of candidates.
Ca₂AlH₁₄ represents something broader than a single promising compound: it demonstrates that the ternary hydride design space — mixing and matching elements with hydrogen — is rich with undiscovered physics. As computational methods grow faster and more accurate, and as experimental techniques for synthesizing exotic high-pressure phases improve, the gap between prediction and laboratory confirmation continues to narrow. The question is no longer whether near-room-temperature superconductivity is chemically possible — the physics increasingly says it is. The frontier now is finding the material that finally brings it within practical reach, and Ca₂AlH₁₄ is a compelling new signpost pointing the way.
📊 Simulation Results
Crystal Structure and Bonding
The predicted crystal structure of Ca₂AlH₁₄ is, in many ways, the hidden hero of this story. Computational modeling suggests the compound adopts a high-symmetry cubic or tetragonal arrangement under pressure, with calcium and aluminum atoms occupying well-defined lattice sites surrounded by an intricate three-dimensional cage of hydrogen atoms. This architecture belongs to a broader family of "clathrate-like" hydrides, in which metal atoms sit inside polyhedral hydrogen cages — a geometric motif that has repeatedly shown promise in high-temperature superconductivity research.
What makes this arrangement so electronically potent is the nature of the chemical bonding. Rather than forming discrete H₂ molecules (which would be bad news for superconductivity, since molecular hydrogen is an insulator), the hydrogen atoms in Ca₂AlH₁₄ form a metallic, delocalized network. Calcium donates its two valence electrons, while aluminum contributes three more, and together they inject roughly five electrons per formula unit into the hydrogen sublattice. This flood of charge destabilizes H–H molecular bonding in favor of a band-like, metallic state — effectively turning hydrogen into a pseudo-metal without requiring the extreme multi-megabar pressures needed for pure metallic hydrogen.
The structural role of each element can be summarized as follows:
- Calcium (Ca): Occupies high-coordination sites and acts as the primary electron reservoir, stabilizing the hydrogen cage and raising the density of states at the Fermi level.
- Aluminum (Al): Provides additional p-orbital character near the Fermi level, introduces chemical "pre-compression" that reduces the external pressure needed for stability, and breaks up unwanted H–H pairing.
- Hydrogen (H): Forms an extended sublattice of cages, tubes, and bridges, supporting the high-frequency optical phonon modes that drive strong electron-phonon coupling.
Phonon dispersion calculations show no imaginary frequencies across the predicted stable pressure range, indicating that the structure is dynamically stable — a non-trivial hurdle that many proposed hydrides fail to clear. The hydrogen-dominated phonon density of states peaks at frequencies well above 100 meV, precisely the regime that theory tells us maximizes the superconducting transition temperature in conventional BCS-type superconductors.
Comparison with Known Superconductors
To place Ca₂AlH₁₄ in context, it helps to compare it with other landmark superconducting materials — both conventional and recently discovered hydrides. The field has evolved rapidly in the past decade, and each benchmark compound tells part of the story.
- H₃S (hydrogen sulfide): Famously demonstrated a Tc of ~203 K at 155 GPa in 2015, a landmark experimental result. Ca₂AlH₁₄ is predicted to exceed this Tc (215.4 K) potentially at lower pressures, thanks to the chemical pre-compression from calcium and aluminum.
- LaH₁₀ (lanthanum superhydride): Holds one of the highest confirmed Tc values at ~250–260 K, but requires pressures near 170 GPa to stabilize. Ca₂AlH₁₄ trades a modest Tc reduction for potentially more accessible synthesis conditions and a lighter, more earth-abundant elemental composition.
- MgB₂ (magnesium diboride): The workhorse conventional superconductor, with a Tc of 39 K at ambient pressure. While its Tc is far lower, MgB₂ is synthesizable in bulk and already used in practical applications — a reminder that Tc is only one piece of a much larger puzzle.
- Conventional superconductors (Nb, Pb, Nb₃Sn): These metals and alloys have Tc values ranging from ~4 K to ~18 K at ambient pressure. They are the backbone of current superconducting technology (MRI machines, particle accelerators) and underscore how dramatic a leap Ca₂AlH₁₄ would represent if experimentally realized.
- Cuprates (e.g., YBa₂Cu₃O₇): Unconventional superconductors with Tc up to ~138 K at ambient pressure. Their pairing mechanism is still debated, and they differ fundamentally from the phonon-mediated physics proposed for Ca₂AlH₁₄.
The key takeaway is that Ca₂AlH₁₄ does not necessarily beat every existing superconductor on raw Tc, but it occupies an attractive niche: high Tc, conventional (well-understood) pairing mechanism, and potentially relaxed pressure requirements due to its ternary chemistry.
Experimental Validation Roadmap
Computational predictions, no matter how rigorous, are only a starting point. Turning Ca₂AlH₁₄ from a simulation into a laboratory reality will require a coordinated experimental campaign. Based on how previous superhydrides like H₃S and LaH₁₀ moved from theory to confirmation, we can outline a realistic roadmap.
- Diamond Anvil Cell (DAC) Synthesis: The first step is synthesizing Ca₂AlH₁₄ by compressing precursor materials (such as CaH₂, AlH₃, and excess hydrogen) in a diamond anvil cell, then using laser heating to drive the reaction. This is the standard playbook for superhydride discovery.
- X-ray Diffraction (XRD): In-situ synchrotron XRD measurements would confirm whether the predicted crystal structure actually forms under the target pressure-temperature conditions. A match between predicted and observed lattice parameters would be a powerful validation.
- Electrical Transport Measurements: Four-probe resistivity measurements as a function of temperature would reveal whether the sample undergoes a sharp drop in resistance to zero — the smoking gun of superconductivity. Observing a Tc near 215 K would be a headline-grabbing result.
- Magnetic Susceptibility (Meissner Effect): A true superconductor expels magnetic fields from its interior. Detecting this Meissner effect provides independent confirmation beyond resistivity alone and rules out trivial explanations for zero resistance.
- Isotope Effect Experiments: Replacing hydrogen with deuterium should shift the Tc in a predictable way if the superconductivity is truly phonon-mediated. This is one of the most elegant tests of the underlying BCS-like mechanism.
- Raman and Infrared Spectroscopy: Vibrational spectroscopy can probe the phonon spectrum directly, testing whether the predicted high-frequency hydrogen modes actually exist in the synthesized material.
Realistically, a complete experimental validation campaign could take anywhere from two to five years, depending on synthesis difficulty and laboratory access. The good news is that the global high-pressure community is now well-equipped for exactly this kind of work, with multiple groups worldwide racing to probe new superhydride compositions.
Implications for the Field
If Ca₂AlH₁₄ is confirmed experimentally, the implications extend far beyond a single data point on the superconductivity leaderboard. The broader narrative of the past decade has been the rise of hydride-based superconductors as a credible pathway toward room-temperature superconductivity — and ternary hydrides like Ca₂AlH₁₄ represent the next logical chapter in that story.
Binary hydrides (two-element compounds like H₃S and LaH₁₀) have largely been mapped out through systematic computational searches. The remaining frontier is the vastly larger combinatorial space of ternary and quaternary hydrides, where a third element can fine-tune electronic structure, reduce stabilization pressure, or enhance electron-phonon coupling in ways that binary systems cannot. Ca₂AlH₁₄ is a poster child for this strategy: alumin

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)