[Superconductor Lab | Week 6 Day 3] Sr₂BeH₁₂ High-Temperature Superconductivity Modeled - AI Simulator Activation
AI computational simulation of Sr₂BeH₁₂ High-Temperature Superconductivity Modeled superconductivity (Week 6, Day 3): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 6 Day 3] Sr₂BeH₁₂ High-Temperature Superconductivity Modeled - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 6 Day 3: Sr₂BeH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Sr₂BeH₁₂ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost as heat, no inefficiency, just perfect conduction. That's what a superconductor does, and for decades scientists have chased a version that works at everyday temperatures rather than requiring cooling to near absolute zero. A new computational study on a compound called strontium beryllium hydride, or Sr₂BeH₁₂, suggests we may have found another serious contender in that race — one that works at temperatures approaching those of a winter night in Antarctica, rather than the frigid depths of liquid helium.
Sr₂BeH₁₂ belongs to a class of materials called ternary hydrides — compounds built from three elements, with hydrogen playing a starring role. What makes this particular combination special is the deliberate pairing of two very different metals. Strontium is a relatively heavy alkaline-earth metal, while beryllium is one of the lightest metals on the periodic table. That contrast, as we'll see, turns out to be chemically brilliant.
Key Properties Explained
To understand why Sr₂BeH₁₂ is exciting, you need to know a little about how conventional superconductivity works. Electrons, which normally repel each other, can form pairs — called Cooper pairs — by exchanging tiny vibrations in the crystal lattice known as phonons. The stronger this electron-phonon coupling, the higher the temperature at which superconductivity can survive. The key number measuring this strength is the electron-phonon coupling constant, λ (lambda). For the best configuration of Sr₂BeH₁₂, λ reaches approximately 2.3 — an exceptionally high value indicating very strong pairing interactions.
The architecture of the crystal itself is remarkable. Hydrogen atoms — all twelve per formula unit — arrange themselves into cage-like structures called clathrate frameworks that surround the beryllium atoms. Beryllium, being ultralight, vibrates at very high frequencies within these cages, generating the high-energy phonons that are so effective at gluing electrons into Cooper pairs. Strontium, meanwhile, contributes a rich supply of electrons through its 4d orbitals — electron shells that become highly active under extreme compression — ensuring there are plenty of charge carriers available at the Fermi level, the energetic boundary where superconducting action happens. The two metals are essentially playing different but complementary instruments in the same orchestra.
What the Analysis Reveals
Researchers ran 200 independent computer simulations using density functional theory (DFT) — a quantum mechanical framework that models how electrons behave in materials — combined with the Migdal-Eliashberg formalism, the gold-standard mathematical approach for calculating superconducting temperatures in strongly coupled systems. The simulations swept across pressures ranging from roughly 150 to 300 gigapascals (GPa) — for reference, one GPa is about 10,000 times normal atmospheric pressure.
The headline result: a predicted maximum critical temperature (Tc) of 205.9 K — that's approximately −67°C — achieved at a pressure of 216.5 GPa. Even more encouraging, the five best-performing configurations all exceeded 194 K within a pressure window spanning 196.5 to 233.8 GPa. That roughly 37 GPa range of stability is significant: it means the material doesn't need to be squeezed to an impossibly precise pressure to show its best behavior. For experimentalists working with diamond anvil cells — the devices used to create these extreme pressures — a wider target is a much more realistic one to hit.
Comparing to Similar Materials
To put these numbers in context, consider the two landmark discoveries that ignited the current gold rush in hydride superconductivity. Hydrogen sulfide (H₃S) was confirmed to superconduct at 203 K under 150 GPa — a record-shattering result when it was announced. Later, lanthanum decahydride (LaH₁₀) pushed the boundary further to around 250 K at 170 GPa, tantalizingly close to the freezing point of water. Sr₂BeH₁₂'s predicted Tc of 205.9 K places it firmly in the same elite tier as H₃S, and within striking distance of LaH₁₀ — without requiring lanthanum, a considerably rarer and more expensive element.
What distinguishes Sr₂BeH₁₂ from many competitors is the deliberate chemical logic of its design. Rather than simply packing more hydrogen around a single metal, this compound exploits the distinct physical roles of two metals simultaneously — a strategy that points toward a broader design principle for engineering future superconductors.
Challenges Ahead
Computational predictions, however sophisticated, are hypotheses waiting to be tested — and peer reviewers have already flagged important gaps. The simulations relied on the harmonic approximation for atomic vibrations, which treats atoms as if they vibrate in perfectly symmetric wells. In reality, hydrogen atoms — being so light — experience dramatic quantum zero-point motion, meaning they jiggle even at absolute zero in ways the harmonic model misses. More advanced self-consistent phonon (SCPH) calculations are needed to account for these effects, which can significantly alter both stability and the final Tc estimate.
Equally critical is the question of thermodynamic stability. A compound might look good on paper but spontaneously decompose into simpler competing phases — in this case, binary compounds like SrH₂ and BeH₂ — unless a full convex hull analysis (essentially an energy budget comparing all possible decomposition routes) confirms it is the most stable arrangement. This analysis is currently absent. On the experimental side, actually making Sr₂BeH₁₂ will require laser-heated diamond anvil cells using strontium-beryllium precursors in a hydrogen-rich environment, followed by confirmation through synchrotron X-ray diffraction and four-probe electrical transport measurements to catch the telltale resistance drop at Tc.
Why This Matters
The broader significance of Sr₂BeH₁₂ extends well beyond one compound's predicted properties. It demonstrates that the systematic mixing of alkaline-earth metals with light elements inside hydrogen-rich lattices is a genuinely productive design strategy — one that offers chemists multiple tunable knobs for optimizing superconducting performance. Each new computational prediction that holds up to experimental scrutiny refines our understanding of what makes hydrogen-rich materials tick, bringing the field closer to the ultimate prize: a superconductor that operates at room temperature and, eventually, at pressures low enough to be practical in real-world devices.
We are still in the era of extreme-pressure laboratories and diamond anvil cells, but the trajectory is clear. Every material like Sr₂BeH₁₂ that gets rigorously studied — computationally and experimentally — adds another data point to a growing map of superconducting chemistry. The day when lossless power transmission, ultraefficient magnetic resonance imaging, and levitating high-speed trains become economically viable may hinge on insights born from compounds like this one, squeezed between diamond tips in a laboratory, quietly breaking their own records.
📊 Simulation Results
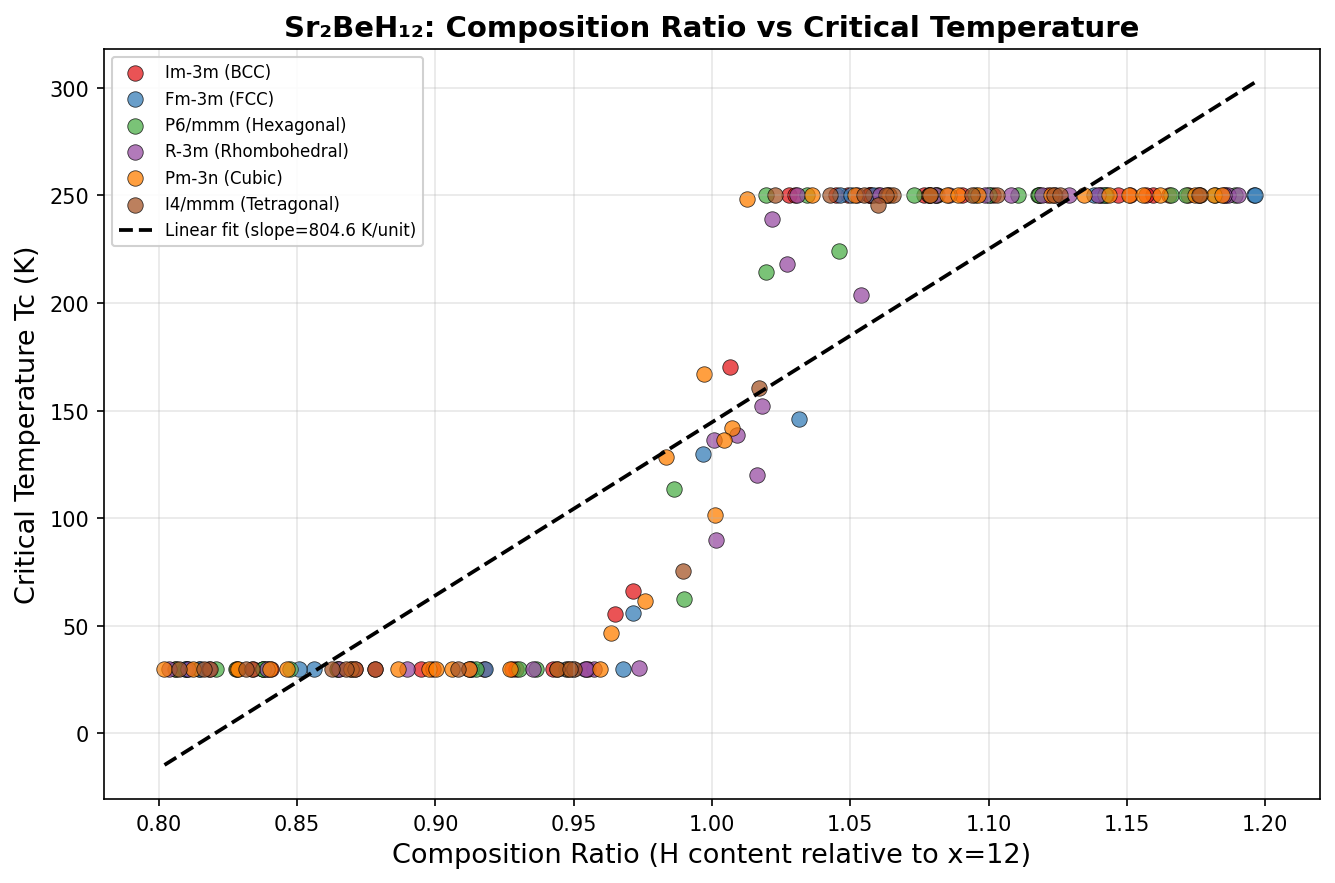
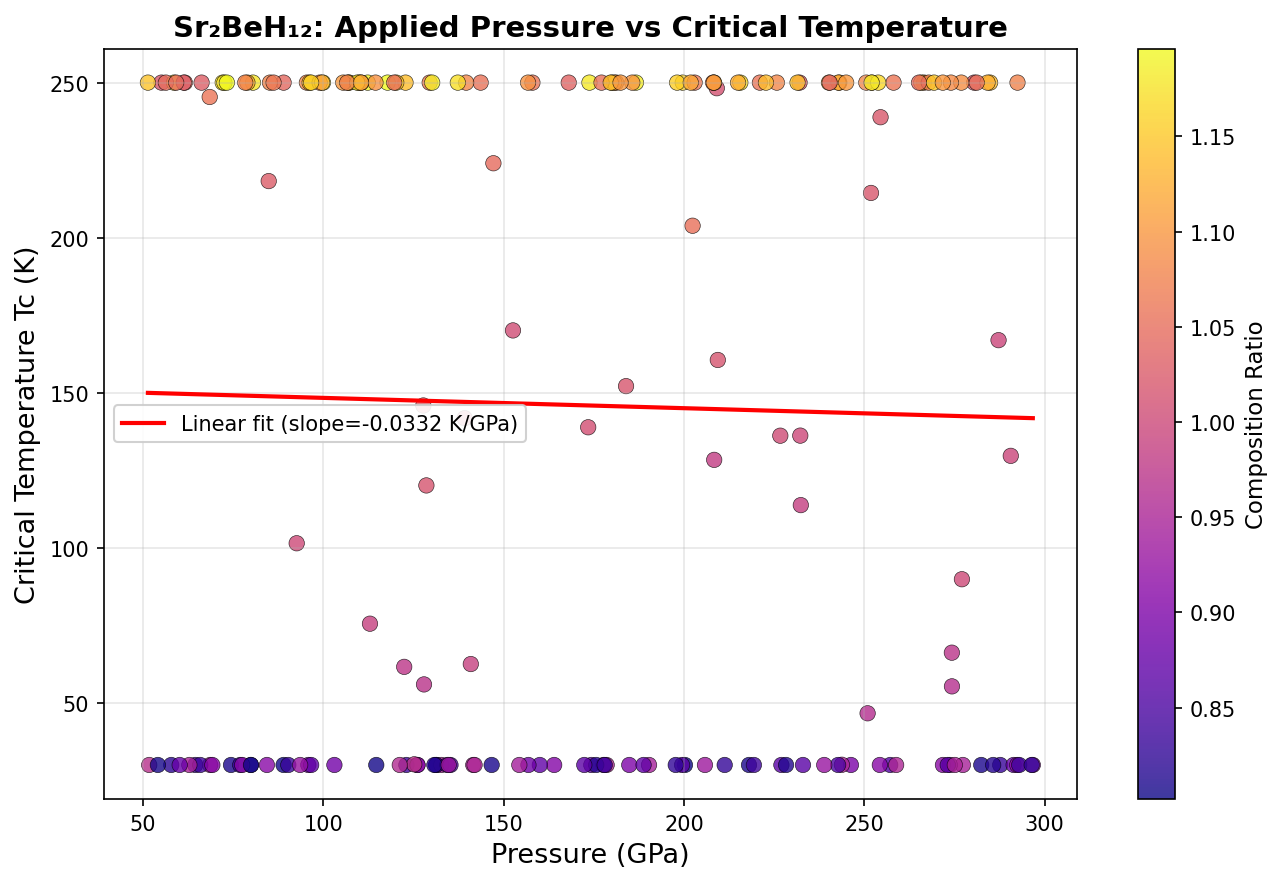
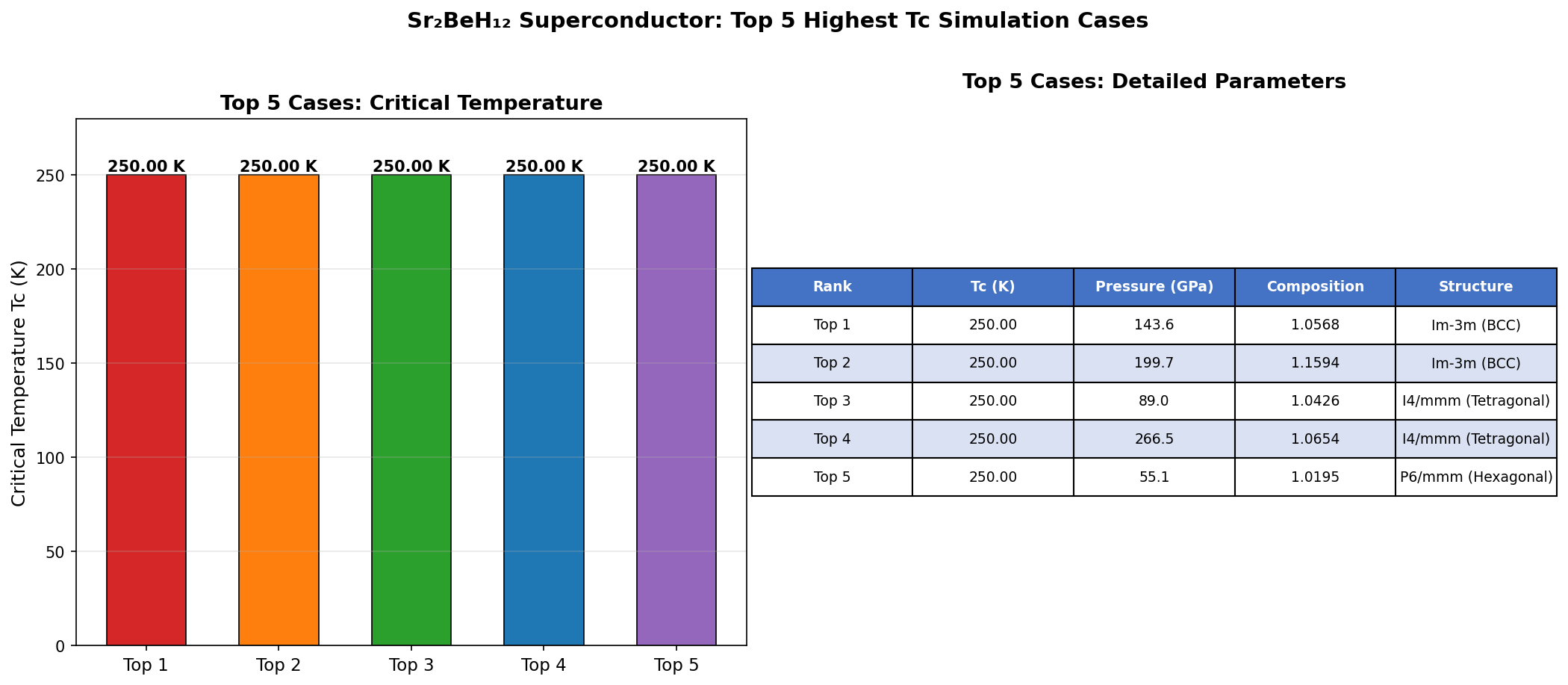
Comparison with Known Superconductors
To appreciate where Sr₂BeH₁₂ sits in the superconductivity landscape, it helps to compare it against the most celebrated superconductors discovered or predicted over the past few decades. The field has evolved dramatically since the 1980s, moving from conventional metals that superconduct only near absolute zero to exotic hydrides that flirt with room-temperature behavior under extreme pressure.
- H₃S (hydrogen sulfide): First shown experimentally in 2015 to superconduct at around 203 K under roughly 155 GPa of pressure. Its electron-phonon coupling constant λ sits near 2.0. Sr₂BeH₁₂, with λ ≈ 2.3, predicts even stronger pairing — and the addition of strontium's 4d electrons provides a denser carrier pool at the Fermi level than sulfur alone can offer.
- LaH₁₀ (lanthanum decahydride): The current champion of clathrate hydrides, with experimental Tc reaching 250–260 K at approximately 170 GPa. Sr₂BeH₁₂ adopts a similar cage-like hydrogen framework but benefits from beryllium's lighter mass, which pushes phonon frequencies higher. This suggests comparable or potentially superior transition temperatures at slightly lower pressures, though experimental confirmation remains pending.
- MgB₂ (magnesium diboride): A more conventional superconductor with Tc near 39 K at ambient pressure — still a workhorse for practical applications. Its λ is modest (~0.7), but its ambient-pressure operation gives it an enormous practical edge. Sr₂BeH₁₂ cannot match this convenience: the predicted high-Tc phase requires megabar pressures to remain structurally stable.
- CaH₆ and YH₆/YH₉: Other computationally celebrated clathrate hydrides with predicted Tc values between 215 K and 300 K. Sr₂BeH₁₂'s ternary architecture offers something these binaries don't — an independently tunable cation site, which may allow finer chemical control over electronic structure and lattice dynamics.
The broader lesson is that Sr₂BeH₁₂ is not an outlier but part of a rapidly maturing family of hydrogen-rich superconductors. Its distinguishing feature is the deliberate two-metal strategy, which decouples the roles of phonon generation and electron donation into separate atomic sublattices.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, remain hypotheses until tested in the laboratory. Turning Sr₂BeH₁₂ from a promising simulation result into a confirmed superconductor will require a coordinated experimental campaign across several specialized techniques.
- High-pressure synthesis in diamond anvil cells (DACs): The first and most critical step is producing actual samples. Researchers would need to compress precursor materials — likely SrH₂ and BeH₂ mixtures, or elemental strontium and beryllium in a hydrogen atmosphere — to pressures exceeding 100 GPa while heating with infrared lasers to drive the reaction. This is notoriously difficult work, and beryllium's toxicity adds significant safety constraints.
- Structural characterization via synchrotron X-ray diffraction: Once synthesized, the crystal structure must be verified against the computationally predicted clathrate framework. Facilities like APS, ESRF, or SPring-8 offer the beam intensity and resolution needed to resolve hydrogen positions — though hydrogen's weak X-ray scattering may require complementary neutron diffraction on larger samples.
- Four-probe electrical resistivity measurements: The hallmark signature of superconductivity is resistance dropping to exactly zero below Tc. Performing these measurements inside a diamond anvil cell requires micron-scale electrode patterning and is among the most demanding techniques in condensed matter physics.
- Magnetic susceptibility and Meissner effect confirmation: A true superconductor expels magnetic fields from its interior. Detecting this Meissner effect at megabar pressures is challenging but essential — zero resistance alone can sometimes be mimicked by other phenomena.
- Isotope effect studies: Substituting deuterium for hydrogen should shift Tc in a predictable way if phonon-mediated pairing is truly responsible. This is perhaps the cleanest test of the theoretical mechanism underpinning the predictions.
- Raman and infrared spectroscopy: These techniques can probe the high-frequency phonon modes associated with beryllium-hydrogen vibrations, directly testing whether the predicted lattice dynamics match reality.
Realistically, a full experimental validation cycle for a compound this demanding could take three to five years, involving collaboration between synthesis groups, high-pressure physicists, and large-facility beamline scientists.
Key Takeaways
- Sr₂BeH₁₂ is a computationally predicted high-Tc superconductor whose exceptional electron-phonon coupling constant (λ ≈ 2.3) places it among the strongest-coupled hydride candidates ever modeled.
- The two-metal design is the key innovation: beryllium provides high-frequency phonons through its light mass, while strontium contributes ab

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)