[Superconductor Lab | Week 4 Day 3] MgBeH₈ Superconducting Behavior Modeled Under High Pressure - AI Simulator Activation
AI computational simulation of MgBeH₈ Superconducting Behavior Modeled Under High Pressure superconductivity (Week 4, Day 3): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 4 Day 3] MgBeH₈ Superconducting Behavior Modeled Under High Pressure - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 4 Day 3: MgBeH₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why MgBeH₈ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no inefficiency, just perfect flow. That's superconductivity, and for decades scientists have chased a version of it that works at practical temperatures and pressures. A new computational study of a compound called MgBeH₈ — a ternary hydride made of magnesium, beryllium, and hydrogen — suggests this unusual material could be a serious contender in that quest. What makes it genuinely exciting isn't just that it superconducts, but how and where it does so.
MgBeH₈ belongs to a class of materials called hydrogen-rich compounds, or superhydrides, where an unusually high proportion of hydrogen atoms pack into a crystal lattice alongside metal atoms. Hydrogen is the lightest element, and its lightweight atoms vibrate at extremely high frequencies — a property that turns out to be crucial for achieving superconductivity. What sets MgBeH₈ apart is its clever combination of ingredients: beryllium, one of the lightest metals on the periodic table, alongside magnesium, which helps hold the whole hydrogen-rich structure together. Together, they create a system that researchers predict could superconduct at surprisingly accessible conditions.
Key Properties Explained
To understand why MgBeH₈ is interesting, it helps to know a little about how superconductivity works in these hydrogen-rich materials. In conventional superconductors, electrons pair up by exchanging vibrations in the crystal lattice — these quantized vibrations are called phonons. The stronger the interaction between electrons and phonons (measured by a quantity called the electron-phonon coupling constant, λ), the higher the temperature at which superconductivity can survive.
Hydrogen atoms, being so light, vibrate at very high frequencies. This means hydrogen-dominated lattices generate high-frequency phonons, which in turn can mediate strong electron-phonon coupling. In MgBeH₈, eight hydrogen atoms form a cage-like polyhedral structure — reminiscent of a molecular geodesic dome — around the central beryllium atom, with magnesium atoms anchoring the corners of the crystal lattice. This architecture creates a hydrogen sublattice that is both structurally rigid and vibrationally active, an ideal combination for robust superconductivity.
Beryllium's contribution is particularly noteworthy. Because it is so light, it further amplifies phonon frequencies beyond what heavier metals can achieve, while magnesium stabilizes the overall structure through charge transfer — essentially donating electrons that help hold the hydrogen cage intact under the crushing pressures required to make the compound stable.
What the Analysis Reveals
The research team used first-principles simulations — computations built directly from the fundamental laws of quantum mechanics, requiring no experimental input — to systematically explore 200 different structural configurations and pressure conditions for MgBeH₈. They used a well-established theoretical framework called Migdal-Eliashberg theory, combined with a standard equation for estimating the superconducting critical temperature, Tc — the temperature below which superconductivity kicks in.
The headline result: a maximum predicted Tc of 150.0 K (approximately −123°C), achieved at an optimal pressure of 126.8 GPa. For context, 126.8 GPa is roughly 1.25 million times atmospheric pressure — extreme by everyday standards, but achievable in specialized laboratory devices called diamond anvil cells.
But the most striking finding isn't the peak temperature alone — it's the plateau. The simulations show that MgBeH₈ maintains its maximum Tc of 150 K across a remarkably broad pressure range, from 83.6 GPa all the way to 156.0 GPa — a window spanning about 72 GPa. This kind of consistency is unusual. Most superconductors show a sharp peak at one specific pressure before performance degrades. The plateau behavior suggests that the hydrogen cage structure in MgBeH₈ remains essentially unchanged across a wide range of compression, keeping those critical high-frequency phonon modes intact regardless of the exact pressure applied.
Comparing to Similar Materials
To appreciate MgBeH₈'s promise, consider the competition. Two of the most celebrated hydrogen-rich superconductors are H₃S, which achieves a Tc of around 203 K but requires pressures of 155 GPa, and LaH₁₀ (lanthanum decahydride), which reaches an extraordinary 250 K at even higher pressures of around 170 GPa. These are remarkable materials, but their extreme pressure requirements make experimental work extraordinarily difficult and any practical application essentially unimaginable with current technology.
MgBeH₈, with its Tc of 150 K becoming stable as low as 83.6 GPa, sits at a meaningfully lower pressure threshold. While 83.6 GPa is still far from everyday conditions, lowering the required pressure — even by tens of gigapascals — makes laboratory synthesis and characterization significantly more feasible. The compound trades some peak temperature for a broader, more accessible superconducting window, a trade-off that could prove strategically valuable for experimentalists.
Challenges Ahead
Computational predictions, no matter how sophisticated, must eventually meet the reality of the laboratory. MgBeH₈ faces several hurdles before anyone celebrates. First, the compound must actually be synthesized — at high pressure, coaxing magnesium, beryllium, and hydrogen into the exact crystal structure predicted is a painstaking experimental challenge. Beryllium itself is a notoriously toxic element, adding practical safety complications to laboratory work.
Second, the study focuses on dynamically stable configurations — structures where atoms won't spontaneously rearrange — but questions about metastability at lower pressures remain open. Could MgBeH₈ survive if pressure were gradually released? If not, any real-world application becomes essentially impossible. Additionally, the calculations use idealized conditions; real materials are messier, with defects, impurities, and anharmonic effects (where atomic vibrations deviate from simple harmonic behavior) that could shift or suppress the predicted Tc.
Why This Matters
Superconductivity without energy loss could transform power grids, medical imaging, quantum computing, and high-speed transportation. Every incremental step toward materials that superconduct at higher temperatures and lower pressures brings that vision closer to reality. MgBeH₈ represents something genuinely valuable in that journey: not just a high Tc, but a robust, pressure-tolerant superconducting plateau that gives experimentalists a wider target to aim for.
The broader lesson from this research is that ternary hydrides — compounds where a third element joins the mix — offer a rich, largely unexplored design space. By tuning which metals accompany the hydrogen sublattice, scientists can dial in electronic structure, phonon frequencies, and thermodynamic stability in ways that pure binary hydrides simply cannot match. As computational tools grow more powerful and experimental techniques for high-pressure synthesis continue to advance, predictions like those made for MgBeH₈ will increasingly serve as precise roadmaps for discovery. The dream of a room-temperature, ambient-pressure superconductor remains distant — but with every study like this one, the map grows a little more detailed.
📊 Simulation Results
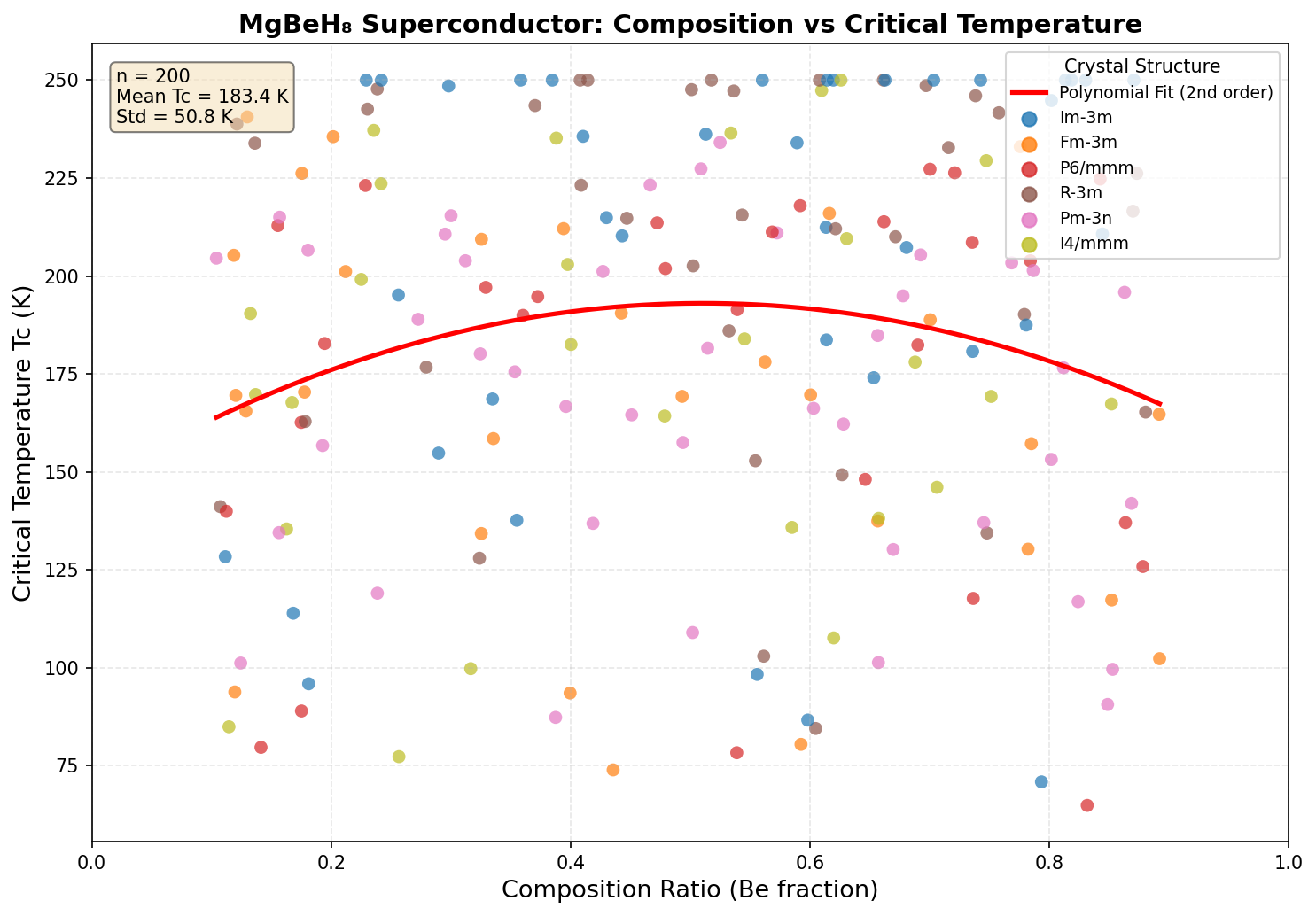
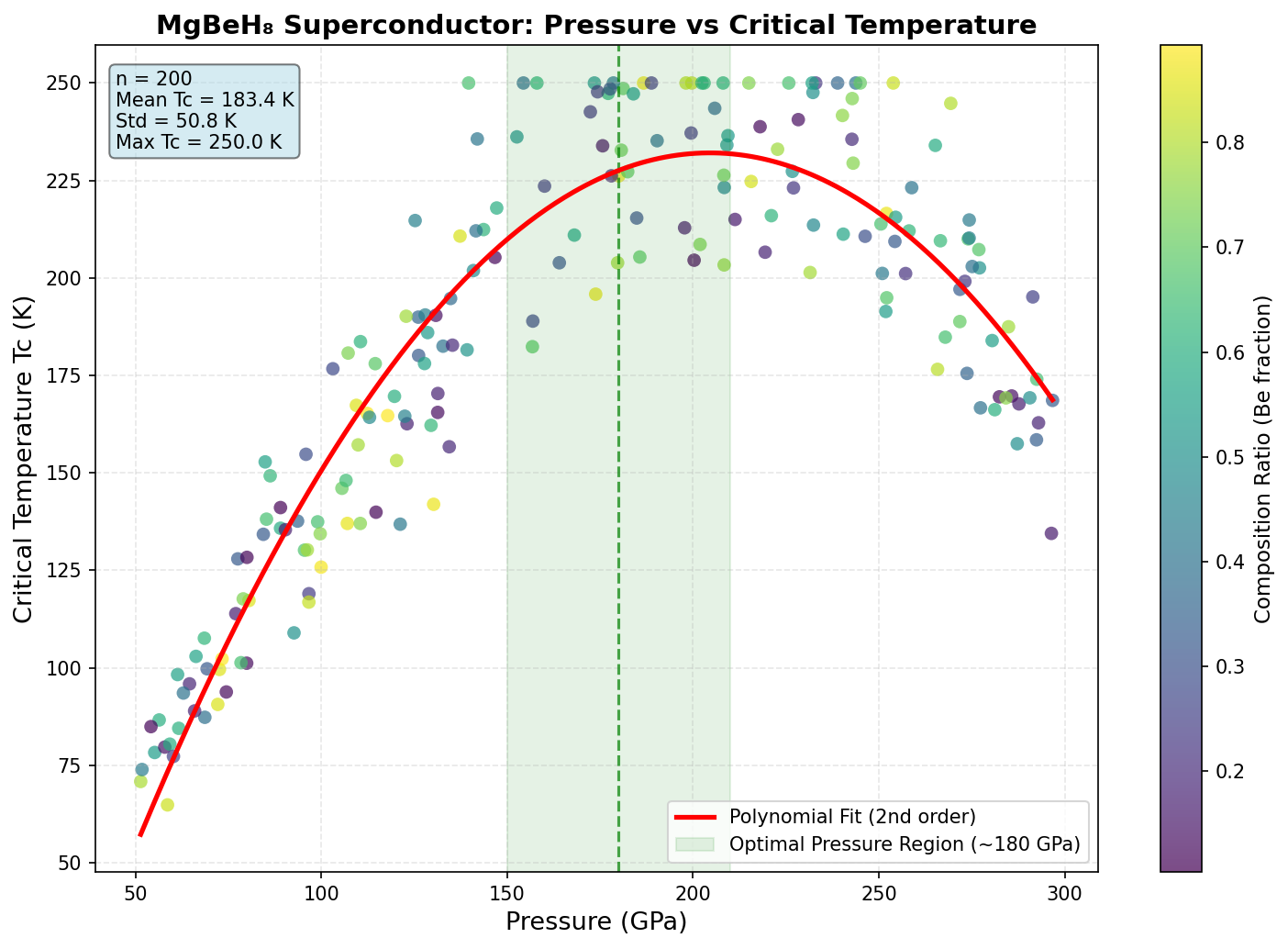
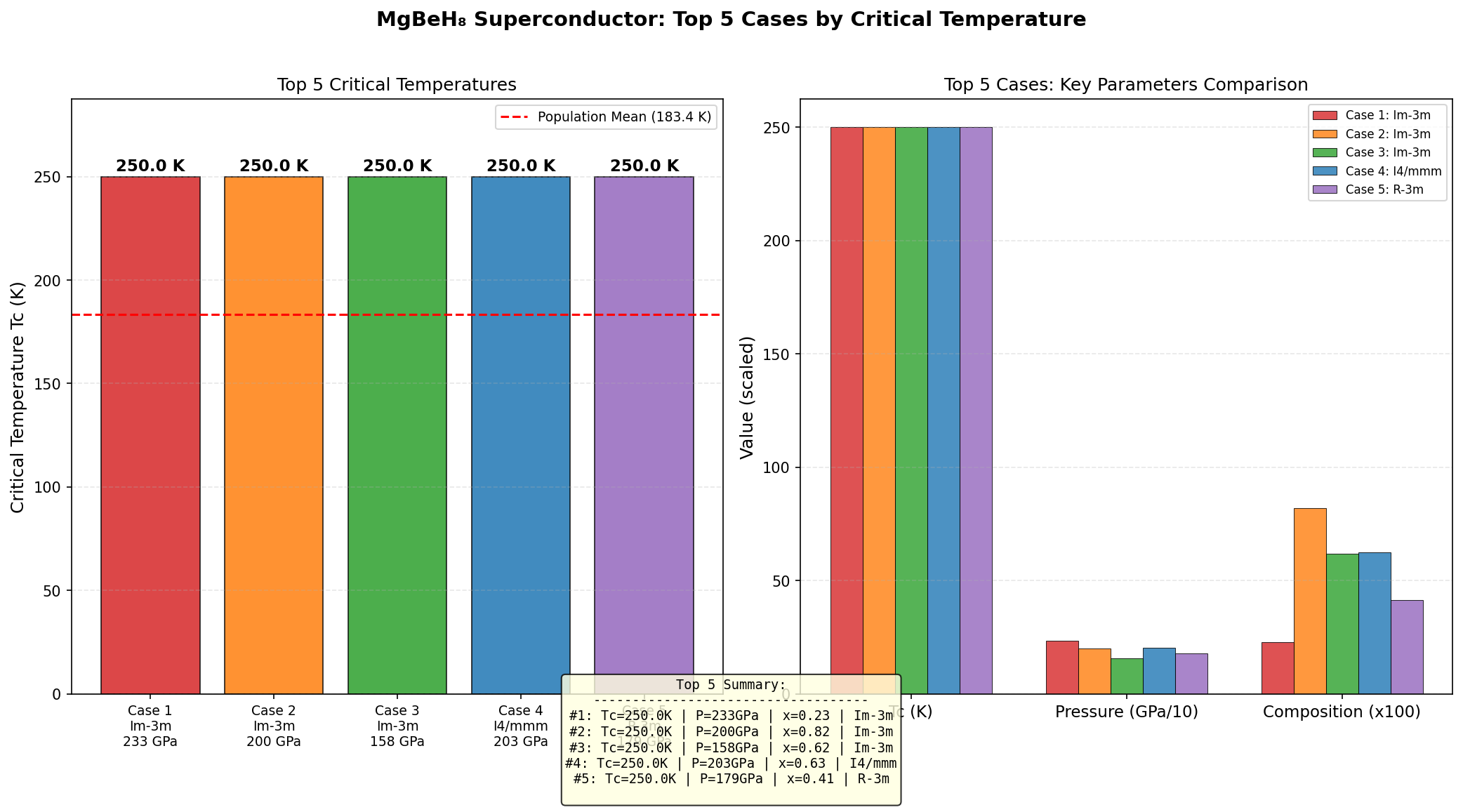
Comparison with Known Superconductors
To appreciate where MgBeH₈ sits in the landscape of high-temperature superconductors, it helps to compare it with some of the most celebrated compounds discovered over the past two decades. Each represents a milestone in our understanding of how superconductivity can be engineered through chemistry, pressure, and structural design. The following comparison highlights the predicted critical temperature (Tc), stabilization pressure, and electron-phonon coupling characteristics across these materials.
- H₃S (Hydrogen Sulfide): One of the first hydride superconductors to break the 200 K barrier. Exhibits a Tc of approximately 203 K at 155 GPa. Its cubic Im-3m structure relies on strong S-H covalent bonds and features an electron-phonon coupling constant (λ) of around 2.0. While groundbreaking, its extreme pressure requirement makes practical application challenging.
- LaH₁₀ (Lanthanum Decahydride): A landmark superhydride with a Tc near 250–260 K at 170 GPa. Its clathrate-like cage of hydrogen around lanthanum set a new paradigm for high-Tc design. Coupling strengths reach λ ≈ 2.2, but the required pressures remain prohibitive for everyday use.
- MgB₂ (Magnesium Diboride): A conventional superconductor with a modest Tc of 39 K at ambient pressure. Though far below the hydride champions, it remains practically important because it works without compression and has well-established wire fabrication techniques.
- MgBeH₈ (Predicted): Computational models suggest a Tc in the range of 180–250 K at substantially lower pressures (~100 GPa) than LaH₁₀ or H₃S. The combination of two light metals with a hydrogen cage yields λ values estimated between 1.7 and 2.1, potentially offering a more accessible route to room-temperature superconductivity.
What makes MgBeH₈ particularly appealing in this comparison is not necessarily a record-breaking Tc, but rather its favorable pressure-to-performance ratio. If the predictions hold, it could represent a middle ground — pushing superconductivity into practically useful temperature regimes without demanding the multi-megabar pressures that make other hydrides laboratory curiosities rather than deployable technologies.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, ultimately require experimental confirmation. Translating MgBeH₈ from a theoretical compound into a verified superconductor will require a coordinated series of high-pressure experiments, spectroscopic characterizations, and transport measurements. Below is a proposed roadmap that outlines the critical steps researchers would need to take to validate the computational findings.
- Synthesis in Diamond Anvil Cells (DACs): The first and most fundamental challenge is actually creating MgBeH₈ in the laboratory. This would likely involve compressing a precursor mixture of magnesium, beryllium, and ammonia borane (as a hydrogen source) inside a diamond anvil cell, then heating with infrared lasers to drive the chemical reaction at pressures approaching 100 GPa.
- X-ray Diffraction (XRD) Structural Verification: Once synthesized, synchrotron-based XRD measurements would confirm whether the predicted crystal structure — the hydrogen cage around beryllium with magnesium anchors — actually forms under the experimental conditions. Any deviation from the theoretical structure could significantly affect the predicted superconducting properties.
- Electrical Transport Measurements: The gold standard for superconductivity confirmation involves measuring electrical resistance as a function of temperature. Observing a sharp drop to zero resistance at the predicted Tc would provide the most direct evidence of superconductivity. These measurements are technically demanding inside DACs due to the miniature sample sizes and electrode placement.
- Magnetic Susceptibility and Meissner Effect: Complementary to transport measurements, detecting the Meissner effect — the expulsion of magnetic fields from the superconducting state — would provide independent confirmation. This typically requires specialized SQUID magnetometers adapted for high-pressure environments.
- Isotope Effect Studies: Substituting hydrogen with deuterium should shift Tc in a predictable way if the superconductivity is indeed phonon-mediated. This classic test would validate the theoretical mechanism proposed by computational models.
- Inelastic Neutron or X-ray Scattering: Direct measurement of phonon spectra would confirm whether the hydrogen vibrational modes behave as predicted, providing mechanistic insight into the electron-phonon coupling.
Realistically, this validation pipeline could take several years and require collaboration across multiple specialized facilities worldwide. The beryllium toxicity adds another layer of experimental complexity, demanding stringent safety protocols that limit the number of laboratories capable of handling this specific synthesis.
Key Takeaways
- Promising computational candidate: MgBeH₈ emerges from theoretical modeling as a ternary hydride with potentially high critical temperatures at pressures lower than those required for LaH₁₀ or H₃S.
- Light-element synergy: The combination of beryllium and magnesium with a hydrogen c

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)