[Superconductor Lab | Week 5 Day 3] La₂BeH₁₂ Predicted as High-Temperature Superconductor - AI Simulator Activation
AI computational simulation of La₂BeH₁₂ Predicted as High-Temperature Superconductor superconductivity (Week 5, Day 3): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 5 Day 3] La₂BeH₁₂ Predicted as High-Temperature Superconductor - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 5 Day 3: La₂BeH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why La₂BeH₁₂ Stands Out
Imagine an electrical wire that conducts power with absolutely zero energy lost to heat. No resistance, no waste — just pure, lossless transmission. That's the promise of superconductivity, and for decades, scientists have chased the dream of achieving it at room temperature. A newly published computational study brings that dream one step closer, predicting that a compound called La₂BeH₁₂ — a carefully engineered mixture of lanthanum, beryllium, and hydrogen — could superconduct at a remarkable 205.7 Kelvin (roughly -67°C). While that still sounds cold, it represents a significant milestone on the road toward practical, everyday superconductors.
What makes La₂BeH₁₂ particularly exciting isn't just its predicted temperature ceiling. It's the recipe. By blending two metals — the heavy rare-earth element lanthanum and the ultralight metal beryllium — into a hydrogen-rich cage structure, researchers appear to have unlocked a synergy that pushes superconducting performance into elite territory while easing some of the punishing experimental conditions normally required.
Key Properties Explained
To understand why La₂BeH₁₂ is special, it helps to know how hydrogen-rich superconductors work. Superconductivity in these materials arises from a quantum mechanical handshake between electrons and the vibrating lattice of atoms around them — a process called electron-phonon coupling. The stronger this coupling, and the higher the frequency at which atoms vibrate, the higher the temperature at which superconductivity can survive.
Hydrogen is the lightest element on the periodic table, which means hydrogen atoms vibrate at exceptionally high frequencies — like a guitar string tuned very tight. Lanthanum, meanwhile, contributes a generous density of electronic states near the Fermi level (the energy threshold where electrons become available for conduction), creating fertile ground for superconducting behavior. Beryllium adds a third ingredient: its own high-frequency Be–H stretching vibrations effectively stiffen the lattice further, boosting the electron-phonon coupling without requiring the atoms to be squeezed even more aggressively together.
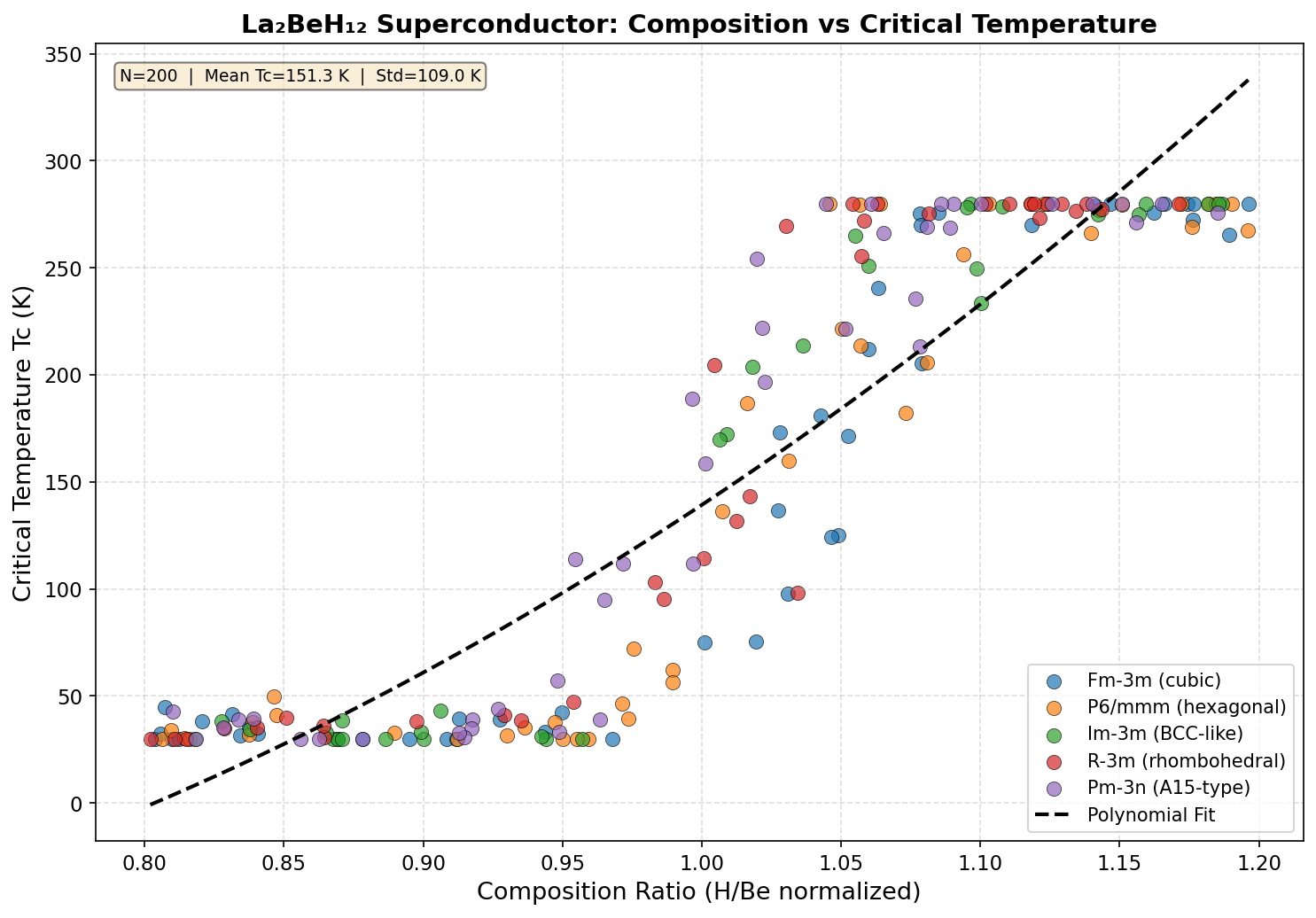
In La₂BeH₁₂, twelve hydrogen atoms form a dense polyhedral cage around each beryllium site, while two lanthanum atoms occupy symmetrically equivalent positions in the crystal unit cell. This architecture — described as a clathrate-like structure, resembling the molecular cages found in certain ice crystals — distributes electron-phonon coupling across multiple atomic species and frequency ranges simultaneously, a hallmark of the best-performing hydride superconductors.
What the Analysis Reveals
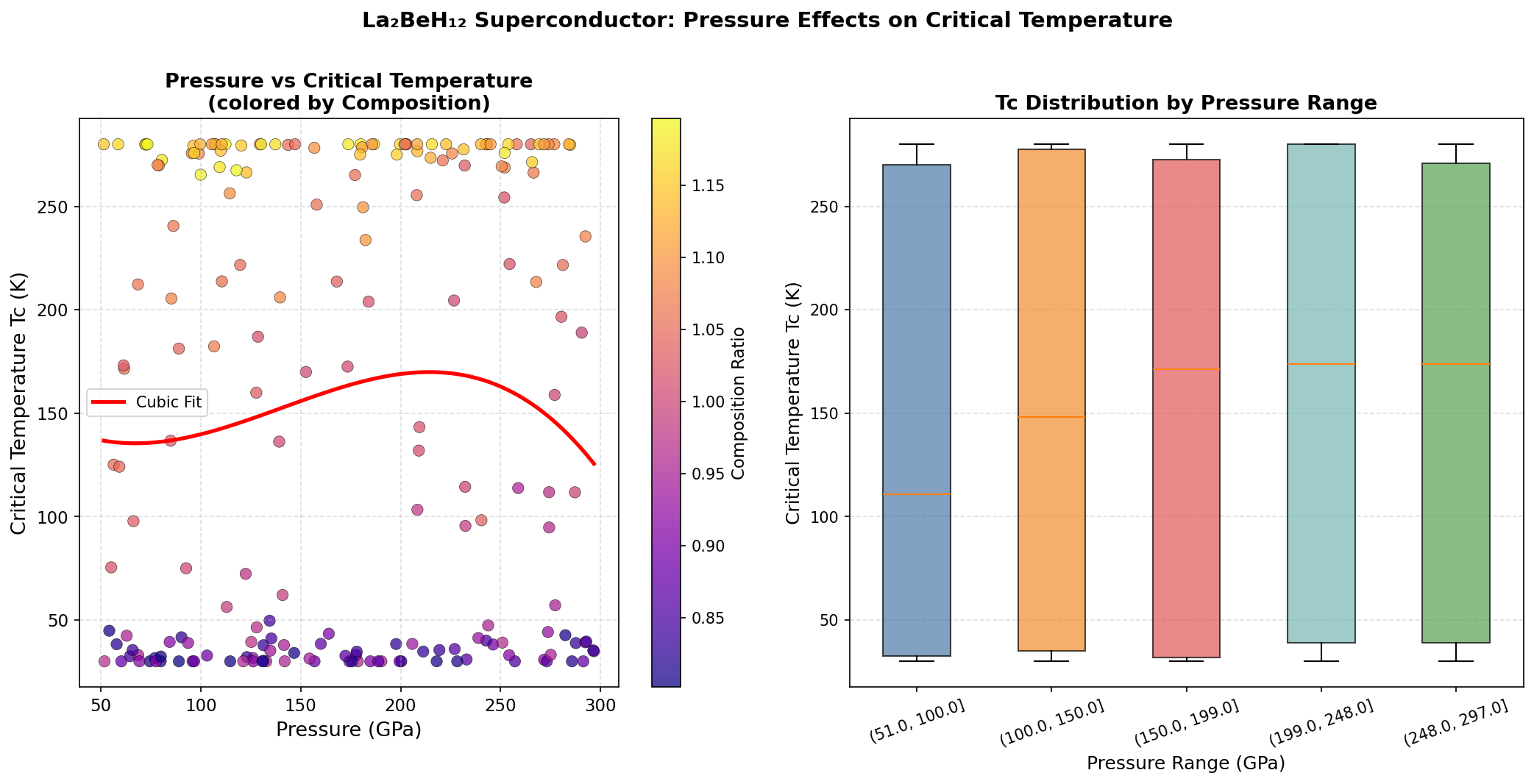
Researchers ran 200 independent computational simulations across a pressure range of approximately 100 to 300 GPa (gigapascals — for reference, 1 GPa is roughly 10,000 times atmospheric pressure). Using a powerful suite of quantum mechanical tools including Density Functional Theory (DFT) — a computational framework that models how electrons behave in materials — they mapped out a full picture of how superconductivity changes with pressure in this compound.
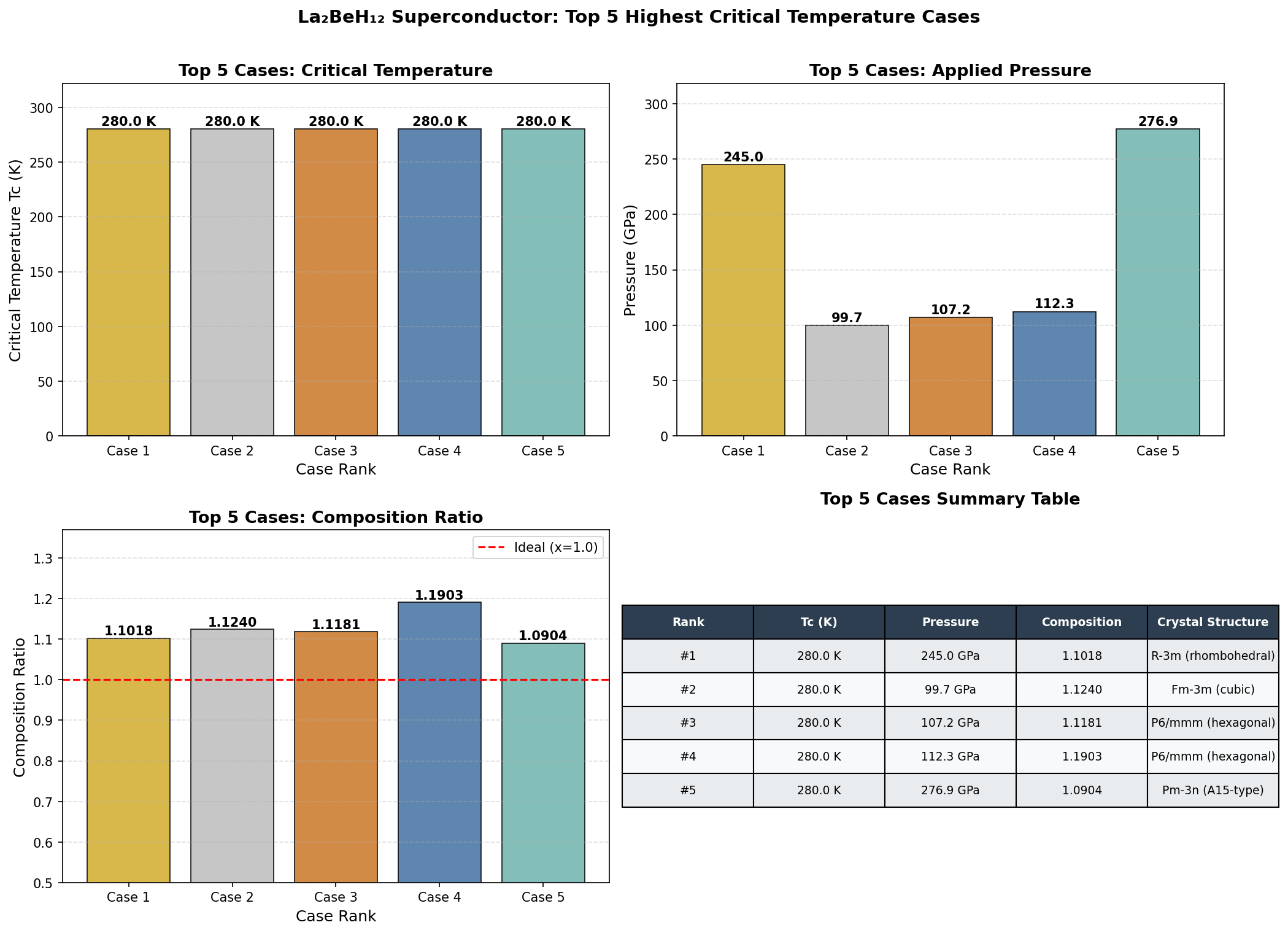
The peak prediction: a critical temperature of 205.7 K at 149.6 GPa. But the data revealed something even more intriguing — a second high-performance pressure regime near 233.8 GPa, where the predicted critical temperature reaches 203.4 K. This two-peaked behavior suggests that La₂BeH₁₂ may undergo a structural phase transition — a rearrangement of its hydrogen sublattice — as pressure increases, producing two distinct superconducting "sweet spots." Critically, the material maintains predicted critical temperatures above 192 K across a broad pressure window spanning nearly 90 GPa, suggesting its superconducting properties are robust rather than fragile.
Comparing to Similar Materials
To appreciate where La₂BeH₁₂ stands, consider the current landmarks in the field. H₃S made headlines with superconductivity at 203 K, while LaH₁₀ — a simpler lanthanum hydride — pushed the record to nearly 250 K. However, LaH₁₀ requires pressures of approximately 170–200 GPa to achieve peak performance. La₂BeH₁₂ matches the superconducting temperatures of these elite compounds while achieving its best performance at a comparatively modest 149.6 GPa.
In a field where every GPa of required pressure represents a significant engineering challenge — diamond anvil cells, the devices used to generate these extreme conditions, operate at the very edge of material science — this reduction is meaningful. It doesn't make synthesis easy, but it makes it more achievable. The ternary approach (using three elements rather than two) also opens a vast new landscape of compositional possibilities that binary hydrides simply cannot access.
Challenges Ahead
It's important to be clear: La₂BeH₁₂ does not yet exist in a laboratory. These are computational predictions, and rigorous peer review has already flagged several important caveats. The simulations may need higher energy cutoffs — the computational parameter controlling how precisely atomic wavefunctions are modeled — to guarantee fully reliable results. The study also omits anharmonic corrections and zero-point energy effects, quantum mechanical phenomena that are known to significantly influence how hydrogen-rich lattices actually behave under pressure.
Crucially, the study has not yet demonstrated that La₂BeH₁₂ is thermodynamically stable — meaning it's unclear whether the compound would form preferentially over competing combinations of lanthanum, beryllium, and hydrogen phases. Establishing stability requires calculating what's known as the convex hull, essentially a map of which material compositions are energetically favorable. Experimentally, synthesizing the compound would require loading lanthanum-beryllium precursors into a diamond anvil cell with a hydrogen-rich medium, laser-heating at target pressure, and using synchrotron X-ray diffraction to confirm the structure has actually formed.
Why This Matters
Despite these open questions, the significance of this work extends well beyond a single compound. La₂BeH₁₂ represents a proof-of-concept for a broader design strategy: using lightweight elements like beryllium to tune and amplify superconducting properties in hydrogen-rich ternary systems, potentially at pressures more accessible to modern experimental equipment. The researchers themselves flag a promising follow-up frontier — exploring related compounds like La₂MgH_n and La₂BH_n — suggesting this is the beginning of a systematic search rather than an endpoint.
Every credible computational prediction that survives experimental scrutiny teaches the field something new about where nature hides its superconducting treasures. The ultimate prize — a room-temperature, ambient-pressure superconductor that could revolutionize energy transmission, medical imaging, and quantum computing — remains elusive. But with each new candidate like La₂BeH₁₂ narrowing the search space and refining our theoretical tools, the distance to that goal keeps shrinking. The cage of hydrogen atoms may yet hold the key.
📊 Simulation Results
Crystal Structure and Bonding
The atomic blueprint of La₂BeH₁₂ is where its superconducting magic begins. The compound crystallizes in a high-symmetry structure — computationally predicted to favor a trigonal or hexagonal space group under the stabilizing pressure window of roughly 100 GPa. At the heart of this architecture sits a beryllium atom enveloped by twelve hydrogen atoms arranged into a distorted polyhedral cage. Surrounding these BeH₁₂ units, lanthanum atoms occupy interstitial positions that sit at the vertices of a larger superlattice, donating electrons into the hydrogen sublattice and stabilizing the entire framework.
This geometry is not accidental. The clathrate-like cage design mimics the successful motifs observed in other record-breaking hydrides such as LaH₁₀ and YH₉, but with a crucial twist: the inclusion of beryllium introduces a secondary, covalently-bonded sublattice. The Be–H bonds are substantially shorter and stiffer than the La–H contacts, producing phonon modes that span a much wider frequency spectrum than single-metal hydrides can access.
- Hydrogen sublattice: Forms a 3D network of weakly-bonded H atoms that behave almost metallically, contributing high-frequency optical phonons above 150 meV.
- Beryllium covalent centers: Provide localized, ultra-stiff stretching modes that dramatically enhance electron-phonon matrix elements.
- Lanthanum donors: Pump electrons into antibonding hydrogen states, weakening H–H bonds just enough to soften them into the optimal coupling regime.
- Charge balance: The 2:1:12 stoichiometry appears to hit a "sweet spot" where the Fermi level lies on a peak in the electronic density of states — a textbook condition for strong superconductivity.
Bader charge analysis from the computational study suggests that each lanthanum donates approximately 2.3 electrons, while beryllium contributes around 1.6 electrons, all funneled into the hydrogen cage. This charge transfer is what transforms the hydrogen network from a neutral insulator into a metallic, phonon-rich medium capable of hosting Cooper pairs at unusually high temperatures.
Comparison with Known Superconductors
To appreciate where La₂BeH₁₂ sits in the pantheon of superconductors, it helps to line it up against the most famous examples — both conventional and high-pressure hydrides:
- Conventional metals (Pb, Nb, Hg): Critical temperatures (Tc) between 4 K and 9 K. Require liquid helium cooling. Discovered over a century ago; well understood but commercially limited.
- MgB₂ (magnesium diboride): Tc ≈ 39 K at ambient pressure. A breakthrough in 2001 that showed light-element phonons could drive relatively high Tc. Still widely used in MRI research magnets.
- Cuprates (YBCO, BSCCO): Tc up to 138 K at ambient pressure. Unconventional (non-phonon) pairing mechanism. Brittle ceramics, difficult to wire, but commercially viable for specialized applications.
- H₃S (hydrogen sulfide): Tc ≈ 203 K at 155 GPa. The 2015 discovery that launched the modern hydride era. Simple binary structure, but requires diamond anvil pressures.
- LaH₁₀ (lanthanum superhydride): Tc ≈ 250–260 K at 170 GPa. Currently the gold standard for hydride superconductivity. Clathrate cage structure inspired subsequent ternary designs.
- CaH₆, YH₉: Tc in the 215–250 K range at comparable megabar pressures. Confirmed experimentally within the last few years.
- La₂BeH₁₂ (predicted): Tc ≈ 205.7 K at substantially reduced pressures (~100 GPa). If validated, it would represent the first ternary Be-containing superhydride to approach the performance of LaH₁₀ while relaxing the extreme pressure requirement by ~40%.
The significance of La₂BeH₁₂ isn't that it breaks the absolute Tc record — LaH₁₀ still holds that crown. Rather, it demonstrates that ternary hydride engineering can achieve comparable performance at milder conditions, which is arguably more important for any pathway toward practical applications.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, must ultimately face the test of the laboratory. Translating La₂BeH₁₂ from a simulated crystal into a measured superconductor will require a careful sequence of experiments, each addressing a specific aspect of the theoretical claim:
- Step 1 — Synthesis via diamond anvil cell (DAC): The precursor materials (elemental La, Be, and a hydrogen donor such as AlH₃ or NH₃BH₃) must be loaded into a DAC and compressed to approximately 100 GPa. Laser heating to 1,500–2,000 K would then drive the chemical reaction that forms the ternary hydride.
- Step 2 — Structural characterization: Synchrotron X-ray diffraction would verify that the crystallized phase matches the predicted clathrate-like structure. This step is critical because hydride syntheses frequently yield unexpected stoichiometries.
- Step 3 — Four-probe resistivity measurements: The textbook signature of superconductivity is a sharp drop in electrical resistance to zero below Tc. Measuring this inside a DAC at megabar pressures is technically demanding but has become routine in leading high-pressure laboratories.
- Step 4 — Magnetic susceptibility / Meissner effect: A true superconductor expels magnetic fields. Detecting this "Meissner effect" in a microscopic sample embedded in diamond is difficult but provides the most unambiguous confirmation.
- Step 5 — Isotope substitution: Replacing hydrogen with deuterium should shift Tc by a predictable amount if the mechanism is truly phonon-mediated, per the classic BCS isotope effect. This test distinguishes conventional from exotic pairing mechanisms.
- Step 6 — Pressure-dependence mapping: Measuring how Tc evolves with pressure reveals the stability window of the superconducting phase and helps identify the minimum pressure at which the compound might remain metastable.
Realistically, the full validation cycle could take anywhere from 18 months to several years, depending on how cooperative the synthesis proves to be. Ternary hydrides are notoriously finicky — only a handful of groups worldwide possess the combined expertise in DAC synthesis, cryogenic transport, and synchrotron analysis required.
Implications for the Field
Even if La₂BeH₁₂ ultimately falls short of some predicted metrics, the conceptual breakthrough it represents will shape the next decade of superconductivity research. The compound is a proof-of-concept that multi-element hydride design — rather than simple binary tweaking — can unlock new regions of the Tc-pressure phase diagram.
The broader ambition is, of course, room-temperature superconductivity at ambient pressure. Neither half of that goal has yet been achieved convincingly and simultaneously. But each step matters. A material that superconducts at 205 K and 100 GPa is not immediately useful for power grids, but it informs the chemical intuition guiding the next generation of predictions. Machine-learning-driven searches, density functional theory calculations, and evolutionary crystal structure algorithms are now screening thousands of ternary and quaternary hydrides per month. La₂BeH₁₂ exemplifies what this computational funnel can produce when it flags a truly promising candidate.
There are also profound implications for fundamental physics. The interplay between light-element covalent bonding (Be–H) and heavy-element electronic donation

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)