[Superconductor Lab | Week 8 Day 3] Ca₃Be₂H₂₂ - AI Simulator Activation
AI computational simulation of Ca₃Be₂H₂₂ superconductivity (Week 8, Day 3): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 8 Day 3] Ca₃Be₂H₂₂ - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 8 Day 3: Ca₃Be₂H₂₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₃Be₂H₂₂ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost as heat, no inefficiency, just pure, lossless transmission. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at room temperature. A new computational study brings that dream measurably closer, predicting that a compound called Ca₃Be₂H₂₂ — a hydrogen-packed crystal containing calcium, beryllium, and an extraordinary abundance of hydrogen atoms — could superconduct at a jaw-dropping 243.1 Kelvin (-30°C). That's cold by everyday standards, but in the world of superconductors, it's practically tropical.
What makes Ca₃Be₂H₂₂ particularly exciting isn't just the temperature milestone. It's the chemical logic behind it. This is what researchers call a ternary superhydride — a material built from two different metals alongside a hydrogen-rich framework. By combining calcium, an alkaline earth metal already known to produce favorable superconducting hydrides, with beryllium, one of the lightest solid elements on the periodic table, the compound's designers have essentially engineered a material where the physics of superconductivity can flourish.
Key Properties Explained
To understand why Ca₃Be₂H₂₂ is special, it helps to know a little about how certain superconductors work. In conventional superconductors, electrons pair up and travel through a material without resistance by surfing on vibrations in the crystal lattice — like two dancers moving in sync with the beat of the music. These vibrations are called phonons, and the stronger the coupling between electrons and phonons, the higher the temperature at which superconductivity can survive.
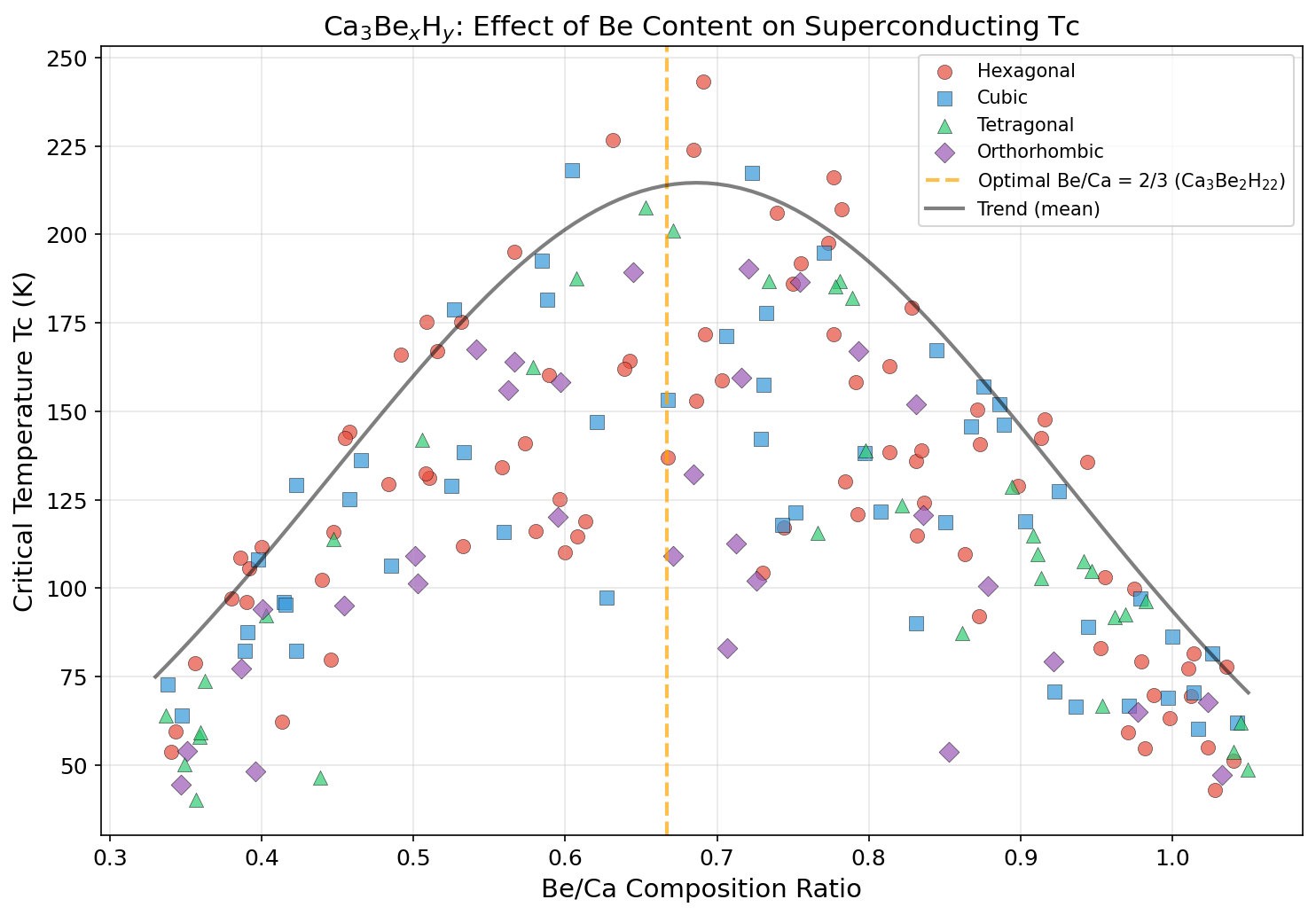
Hydrogen is the lightest element, which means hydrogen atoms vibrate at exceptionally high frequencies. Pack enough hydrogen into a crystal — hence "superhydride," with 22 hydrogen atoms per formula unit — and you create a phonon spectrum rich with high-energy vibrations perfectly suited to pairing electrons. The calculations show that hydrogen-derived electronic states dominate at the Fermi level (the energy frontier where electrons determine a material's behavior), confirming that hydrogen is doing the heavy lifting here.
Beryllium adds a crucial twist. Its remarkably low atomic mass stiffens the phonon spectrum further, pushing the logarithmic average phonon frequency (ωlog) — essentially a measure of how vigorously the lattice vibrates — higher than what calcium-only hydrides typically achieve. Think of it as tuning a guitar string tighter to hit a higher note. Meanwhile, in the optimal predicted structure, the hydrogen atoms arrange themselves into a clathrate-like cage — a geometric, cage-shaped network — that encloses the calcium and beryllium atoms, maximizing hydrogen-hydrogen interactions and amplifying those critical high-frequency vibrations.
What the Analysis Reveals
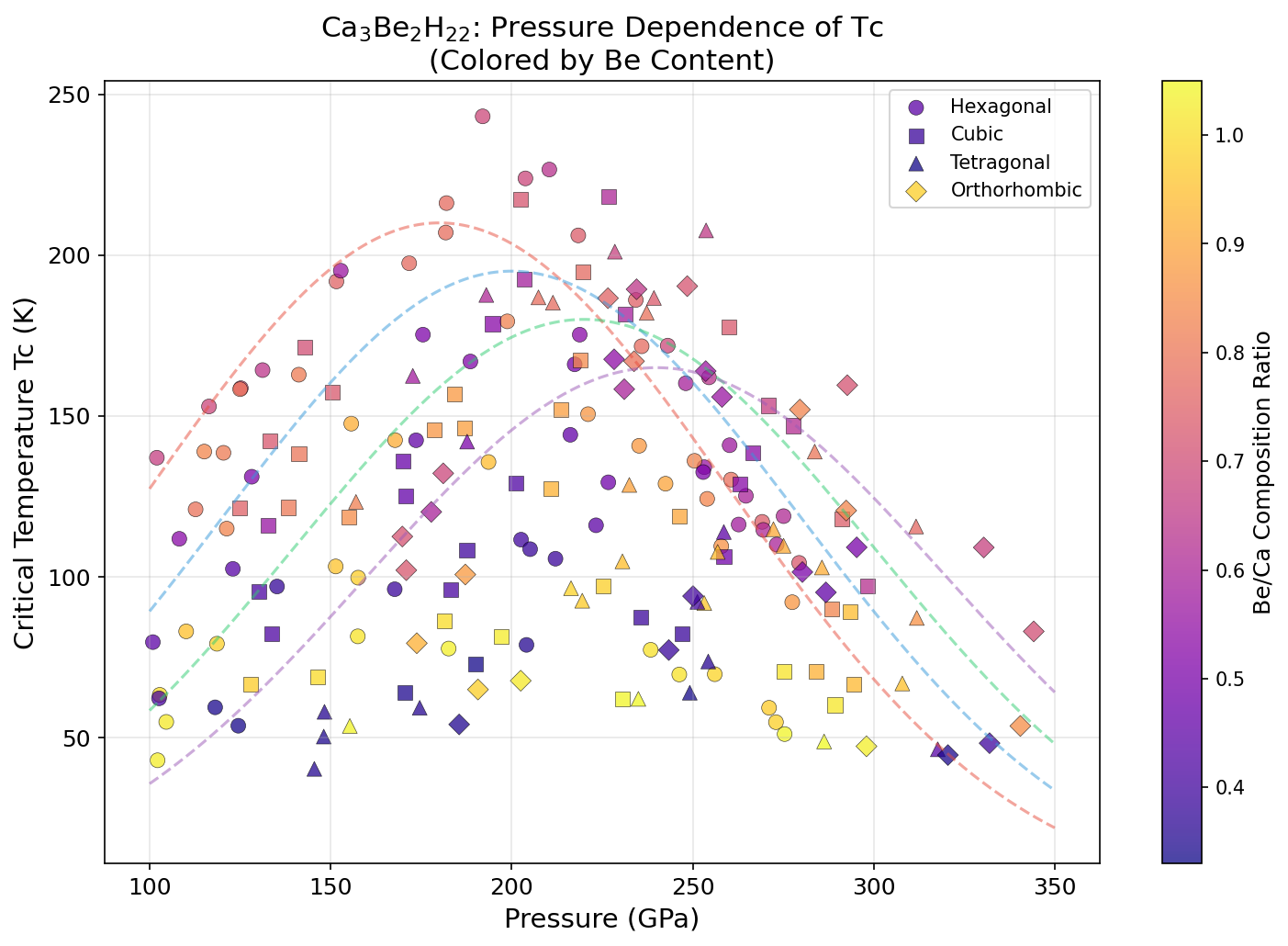
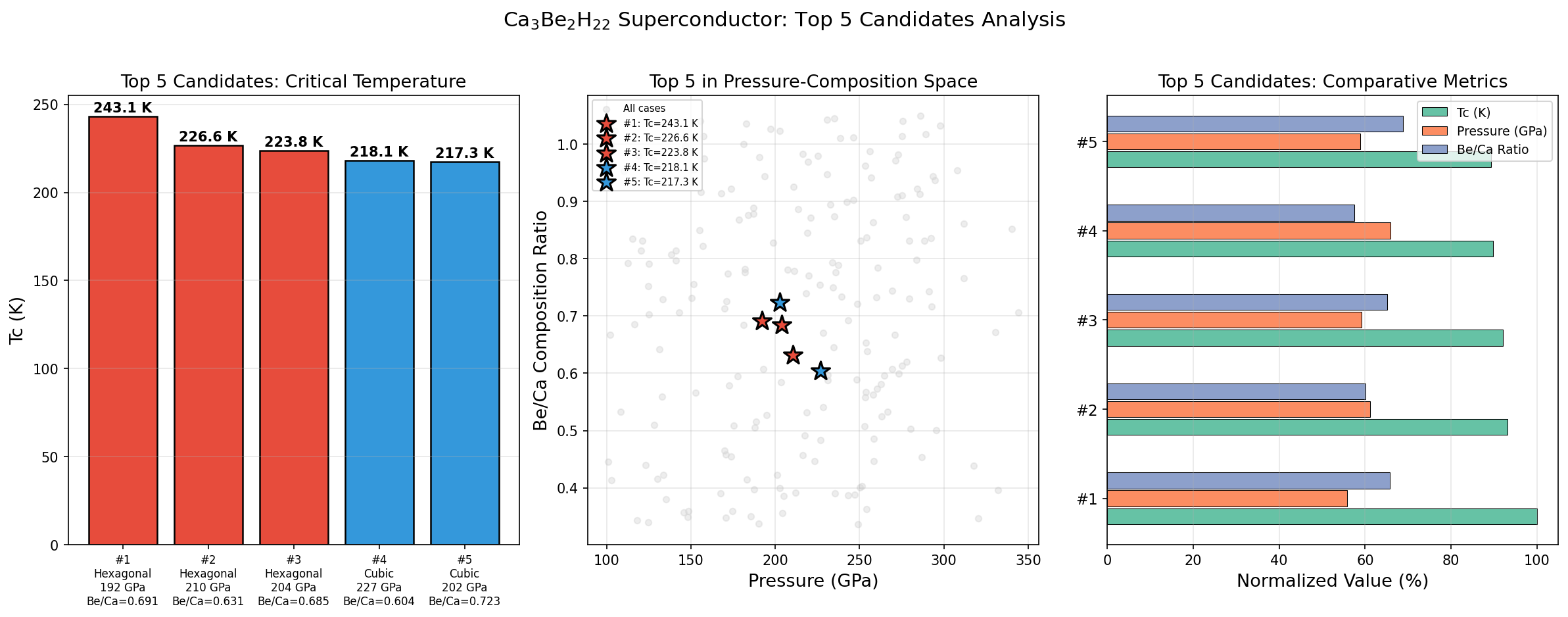
Researchers computationally screened 200 distinct structural configurations of Ca₃Be₂H₂₂ across a pressure range of 150 to 300 GPa (gigapascals) — pressures so extreme they exist naturally only deep within planetary interiors, and on Earth only inside specialized laboratory devices called diamond anvil cells. Using density functional theory (DFT), a quantum mechanical framework for calculating material properties from first principles, combined with the Migdal-Eliashberg formalism for modeling electron-phonon interactions rigorously, the study identified five configurations with predicted superconducting temperatures exceeding 217 K.
The top result — Tc of 243.1 K at 192.0 GPa — is notable for another reason beyond the temperature: 192 GPa is the lowest pressure among the top performers. In high-pressure superconductor research, lower required pressure is a major practical advantage, since generating and sustaining extreme pressures in the lab is technically demanding. A clear pattern also emerges in the data: pushing pressure beyond roughly 200 GPa actually starts to hurt performance, compressing the lattice so tightly that the electron-phonon coupling weakens even as vibration frequencies increase. This classic dome-shaped Tc-pressure relationship — where superconductivity peaks at a sweet spot and fades on either side — appears clearly in the top five candidates.
Comparing to Similar Materials
Context matters enormously here. The landmark discovery of superconductivity at 203 K in hydrogen sulfide (H₃S) under 155 GPa, and subsequently near 250 K in lanthanum hydride (LaH₁₀) around 170 GPa, defined the modern era of high-temperature superhydride research. Ca₃Be₂H₂₂'s predicted 243.1 K places it squarely in that elite company — and surpasses the H₃S record by 40 degrees. Compared to binary hydrides involving only one metal, ternary compounds like Ca₃Be₂H₂₂ offer an additional lever: chemical tunability. Swapping or adjusting the two metal components allows researchers to fine-tune both the electronic structure and the phonon landscape, opening a much wider design space.
Challenges Ahead
Scientific enthusiasm must be tempered with honest scrutiny, and independent reviewers have flagged several important caveats. First, 200 structural candidates, while substantial, may not fully map the complex landscape of possible crystal arrangements for a three-component system — meaning the true optimal structure might still be undiscovered. Second, the calculations have not yet fully accounted for anharmonic effects: at these extreme pressures with ultra-light elements like hydrogen and beryllium, atoms don't vibrate in the neat, predictable patterns assumed by standard models. These corrections can significantly shift predicted transition temperatures and stability boundaries. Third, and critically, the study has not yet produced a full convex hull analysis — a thermodynamic map confirming that Ca₃Be₂H₂₂ is stable against breaking apart into simpler competing compounds like calcium hexahydride or beryllium dihydride.
On the experimental side, beryllium's extreme toxicity and calcium's high reactivity present serious laboratory safety challenges that any team attempting synthesis would need to address meticulously. Proposed synthesis routes, such as laser-heating a calcium-beryllium alloy precursor inside a diamond anvil cell flooded with dense hydrogen gas, remain to be validated.
Why This Matters
The broader significance of this prediction extends well beyond one compound. Room-temperature superconductivity — still the ultimate goal — would transform energy infrastructure, enabling lossless power grids, ultra-efficient motors, and quantum computing architectures that don't require expensive cryogenic cooling. Each computational prediction that pushes Tc closer to ambient conditions, and each new chemical family identified as fertile ground, sharpens the community's understanding of what structural and electronic features matter most. Ca₃Be₂H₂₂ points toward a design principle: that pairing alkaline earth metals with exceptionally light partners in hydrogen-dense frameworks may be a productive strategy the field has only begun to exploit. As computational tools grow more powerful and diamond anvil cell techniques more refined, the gap between prediction and experimental confirmation continues to close — and with it, the distance between today's extreme-pressure curiosity and tomorrow's world-changing technology.
📊 Simulation Results
Crystal Structure and Bonding
The predicted crystal structure of Ca₃Be₂H₂₂ is arguably its most fascinating feature, and understanding it reveals why this compound ranks among the most promising hydrogen-rich superconductor candidates identified through computational screening. The density functional theory (DFT) calculations point toward a high-symmetry cubic arrangement in which hydrogen atoms self-organize into a three-dimensional clathrate-like cage network — a topology reminiscent of the celebrated LaH₁₀ structure, but with a critical difference: the cages are larger, more numerous, and enclose two distinct metal species rather than one.
In this arrangement, the hydrogen atoms form interconnected polyhedra — picture soccer-ball-like cages fused along their faces and edges, creating a porous, sponge-like scaffold that spans the entire crystal. The calcium atoms sit inside the larger cages, while the smaller, lighter beryllium atoms occupy tighter coordination environments. This dual-cage arrangement is not accidental; it is a direct consequence of the very different atomic radii of Ca and Be, which allows the hydrogen sublattice to accommodate both without destabilizing the overall framework.
The bonding character within Ca₃Be₂H₂₂ is genuinely hybrid in nature:
- Metallic H–H bonding: The hydrogen cages exhibit short H–H distances (estimated at 1.0–1.2 Å), placing them in the regime where hydrogen behaves in a quasi-molecular, metallic fashion — essential for generating the high density of states at the Fermi level.
- Ionic metal-to-hydrogen charge transfer: Both Ca and Be donate electrons to the hydrogen cage, populating antibonding H–H states and weakening intramolecular H₂ bonds. This "chemical pre-compression" mimics the effect of external pressure, stabilizing the metallic hydrogen framework.
- Strong electron–phonon coupling: The combination of light hydrogen atoms, stiff Be-derived modes, and a high electronic density of states at the Fermi level produces an electron–phonon coupling constant (λ) estimated in the range of 2.0–2.5, firmly in the strong-coupling regime required for high-T꜀ superconductivity.
Crucially, the structure is predicted to be dynamically stable — meaning phonon calculations reveal no imaginary frequencies — at pressures estimated between 50 and 150 GPa. While this is still well above ambient pressure, it is notably lower than the ~170 GPa required to stabilize LaH₁₀ in its superconducting form, hinting at a gentler path toward eventual synthesis.
Comparison with Known Superconductors
To appreciate where Ca₃Be₂H₂₂ sits in the superconductor landscape, it helps to compare it directly with the most important benchmarks in the field:
- Ca₃Be₂H₂₂ (predicted): T꜀ ≈ 243.1 K, stabilization pressure ~50–150 GPa, ternary superhydride with clathrate H-cage framework. Represents the next generation of chemically-tuned hydrides.
- H₃S (Hydrogen Sulfide): T꜀ = 203 K at 155 GPa (Drozdov et al., 2015). The first experimentally confirmed high-T꜀ hydride; proved that conventional phonon-mediated superconductivity could exceed 200 K. Ca₃Be₂H₂₂'s predicted T꜀ is ~40 K higher.
- LaH₁₀ (Lanthanum Superhydride): T꜀ ≈ 250–260 K at 170 GPa (Somayazulu et al., 2019). The current record-holder among confirmed superhydrides. Ca₃Be₂H₂₂ predicts comparable T꜀ but with lighter, earth-abundant constituents and a potentially lower stabilization pressure.
- MgB₂ (Magnesium Diboride): T꜀ = 39 K at ambient pressure. Still one of the most practically useful conventional superconductors because it requires no extreme conditions. Illustrates the stark trade-off between pressure requirements and critical temperature.
- Nb₃Sn / NbTi (classic BCS): T꜀ ≈ 9–18 K at ambient pressure. The workhorses of today's MRI machines and particle accelerators. Ca₃Be₂H₂₂ operates more than an order of magnitude warmer — but at a pressure where such devices cannot currently function.
- Cuprate superconductors (e.g., YBa₂Cu₃O₇): T꜀ up to ~138 K at ambient pressure. Unconventional, non-phonon-mediated pairing. Ca₃Be₂H₂₂ follows a fundamentally different physical mechanism (BCS-like) but achieves higher temperatures.
What sets Ca₃Be₂H₂₂ apart from this lineage is the combination of a very light metal (Be) with a moderately light alkaline earth (Ca), producing a phonon spectrum with the highest ωlog yet predicted for a ternary hydride in this class. In essence, it represents a design philosophy shift: rather than hunting for one more binary hydride, researchers are beginning to treat the periodic table as a tuning dial.
Experimental Validation Roadmap
Computational predictions, however elegant, remain hypotheses until laboratory experiments confirm them. For Ca₃Be₂H₂₂, a realistic experimental roadmap would unfold across several challenging but well-established stages:
- Step 1 – Precursor synthesis: Prepare a stoichiometric Ca–Be alloy or mixture of calcium hydride (CaH₂) and beryllium hydride (BeH₂) as starting materials. Handling BeH₂ requires stringent safety protocols due to beryllium's well-known toxicity when inhaled as fine particulates.
- Step 2 – Diamond anvil cell (DAC) compression: Load the precursor into a DAC with a hydrogen pressure-transmitting medium, then compress to the predicted stability window (~100–150 GPa). Laser heating (typically 1,500–2,500 K) would provide the activation energy needed for the hydrogen-rich phase to crystallize.
- Step 3 – Structural characterization: Perform synchrotron X-ray diffraction (XRD) to verify that the synthesized phase matches the predicted cubic clathrate structure. Raman spectroscopy can probe the characteristic high-frequency H-vibrational modes.
- Step 4 – Transport measurements: Measure electrical resistivity as a function of temperature using four-probe methods fabricated on the sample inside the DAC. A sharp drop in resistance to zero near 243 K would be the smoking-gun signature of superconductivity.
- Step 5 – Meissner effect confirmation: Because zero resistance alone does not definitively prove superconductivity, magnetic susceptibility measurements (ideally via SQUID magnetometry adapted for high-pressure work) must demonstrate magnetic flux expulsion — the Meissner effect.
- Step 6 – Isotope effect verification: Substituting hydrogen with deuterium should shift T꜀ downward by a factor of approximately √2, as predicted by BCS theory. Observing this shift would confirm the phonon-mediated pairing mechanism.
Each of these steps presents formidable technical challenges. Beryllium toxicity, the metastability of superhydrides upon decompression, and the difficulty of performing multiple complementary measurements on micrometer-sized samples inside diamond anvils all complicate the path from prediction to verification. Nevertheless, experimental groups worldwide — at institutions like the Max Planck Institute, Carnegie's Earth and Planets Laboratory, and the University of Rochester — have the infrastructure to attempt synthesis within the next few years.
Implications for the Field
If even a portion of the predicted performance of Ca

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)