[Superconductor Lab | Week 3 Day 3] Ca₂BeH₈ Predicted as High-Temp Superconductor - AI Simulator Activation
AI computational simulation of Ca₂BeH₈ Predicted as High-Temp Superconductor superconductivity (Week 3, Day 3): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 3 Day 3] Ca₂BeH₈ Predicted as High-Temp Superconductor - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 3 Day 3: Ca₂BeH₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂BeH₈ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost to heat, no inefficiency, just pure, frictionless flow of electrons. That is the promise of superconductivity, and for decades scientists have chased a version of it that works at practical temperatures without requiring exotic, expensive cooling. A new computational study suggests that a compound called calcium beryllium octahydride, or Ca₂BeH₈, could be a meaningful step toward that goal. Predicted to superconduct at up to 144.7 Kelvin (-128°C) under high pressure, this ternary hydride — a material combining three elements with hydrogen as a dominant partner — is turning heads in the theoretical physics community for reasons that go well beyond its headline temperature.
What makes Ca₂BeH₈ genuinely interesting is not just what it does, but how it does it. By blending calcium, beryllium, and hydrogen into a single crystalline framework, researchers have stumbled upon a compositional sweet spot that achieves its remarkable properties at a comparatively moderate pressure of 145.6 gigapascals (GPa) — roughly 1.4 million times the atmospheric pressure at sea level. In the high-pressure superconductor world, that relative modesty matters enormously.
Key Properties Explained
To appreciate why Ca₂BeH₈ is exciting, it helps to understand the basic physics at play. Superconductivity in hydrogen-rich materials arises from a phenomenon called electron-phonon coupling — essentially, the way electrons interact with the vibrations (phonons) of the crystal lattice. Hydrogen atoms are extraordinarily light, which means they vibrate at very high frequencies. Those rapid vibrations create strong coupling with electrons, which can pair them up into the special quantum partnerships responsible for superconductivity. The strength of this pairing is captured by a number called lambda (λ), the electron-phonon coupling constant. A higher λ generally means a higher critical temperature (Tc) — the temperature below which superconductivity kicks in.
In Ca₂BeH₈, the hydrogen sublattice is the star of the show, contributing roughly 75% of the total electron-phonon coupling through high-frequency vibrations above 100 millielectronvolts. But beryllium plays a surprisingly pivotal supporting role. As a lightweight element with strong covalent bonding tendencies, beryllium mediates intermediate-frequency phonon modes that bridge the slower vibrations of heavy calcium atoms and the faster vibrations of hydrogen. Think of it as a musical arranger connecting the bass section to the high notes — without it, the harmony that drives superconductivity would be far less coherent. Meanwhile, calcium contributes its 3d electrons to the electronic structure near the Fermi level (the energy boundary where electrons become available for conduction), creating a rich, multi-band environment favorable for superconducting pairing.
What the Analysis Reveals
The researchers ran an impressive 200 independent first-principles simulations — computational experiments grounded in quantum mechanics, requiring no empirical shortcuts — across a pressure range of roughly 100 to 250 GPa. The result is a beautifully clear picture of what physicists call a superconducting dome: a pressure-temperature relationship that rises to a peak and then falls away on either side.
The peak sits at 144.7 K and 145.6 GPa. Push the pressure higher, and Tc actually drops — a phenomenon explained by phonon hardening. Squeeze the crystal too aggressively and the hydrogen atoms stiffen up, vibrating in ways that are too rigid to couple efficiently with electrons. The superconducting sweet spot is therefore a delicate balance: enough pressure to stabilize the high-hydrogen structure, but not so much that the vibrations lose their pairing potency. This inverse relationship between excessive pressure and Tc is a critical design insight for anyone hoping to engineer better hydride superconductors.
Comparing to Similar Materials
Context is everything in materials science. The landmark superconductors in this field include hydrogen sulfide (H₃S), which superconducts at 203 K under 150 GPa, and lanthanum decahydride (LaH₁₀), which achieves a stunning ~250 K under 170 GPa. Closer to Ca₂BeH₈'s chemical family is CaH₆, a binary calcium hydride predicted to exceed 235 K — still higher than Ca₂BeH₈'s 144.7 K ceiling.
So why bother with Ca₂BeH₈? Because raw Tc is not the only metric that counts. Ca₂BeH₈ achieves its performance at a pressure lower than many competitors, and its ternary composition — three elements instead of two — may provide greater thermodynamic stability. Beryllium's strong covalent bonds can reinforce the structural framework, potentially making the compound less prone to decomposition. In a field where materials sometimes collapse or transform before researchers can fully characterize them, structural robustness is a serious advantage.
Challenges Ahead
Computational predictions, however sophisticated, are not the same as experimental reality. Ca₂BeH₈ has not yet been synthesized in a laboratory. Creating it would require a diamond anvil cell — a device that squeezes microscopic samples between two gem-quality diamond tips to generate the necessary pressures. Working with beryllium adds another layer of difficulty: it is toxic, requiring specialized handling protocols that complicate experimental workflows.
There is also the question of whether the predicted crystal structure actually forms under pressure, or whether competing phases — different atomic arrangements — prove more stable. The computational methodology used here, while rigorous and well-validated, relies on approximations that real atoms do not always honor. Experimental confirmation will be the ultimate test of these predictions.
Why This Matters
Every credible computational prediction of a new superconductor adds a data point to one of science's most important maps: the landscape of materials that might one day enable lossless power grids, ultra-efficient MRI machines, and quantum computers that operate at manageable temperatures. Ca₂BeH₈ matters not just as a single candidate but as a proof of concept for an underexplored design strategy — using ternary alkaline-earth beryllium hydrides as a platform. If beryllium's intermediate phonon modes genuinely enhance coupling the way these calculations suggest, the entire Ca–Be–H compositional family deserves systematic exploration.
Future work targeting doping strategies — swapping small fractions of one element for another to tune the electronic structure — or exploring neighboring compositions could reveal pathways to even higher critical temperatures at pressures low enough to make experimental synthesis genuinely tractable. The dream of a room-temperature superconductor remains distant, but compounds like Ca₂BeH₈ remind us that the periodic table still holds extraordinary surprises for those willing to compute, synthesize, and look carefully at what hydrogen can do when put under pressure.
📊 Simulation Results
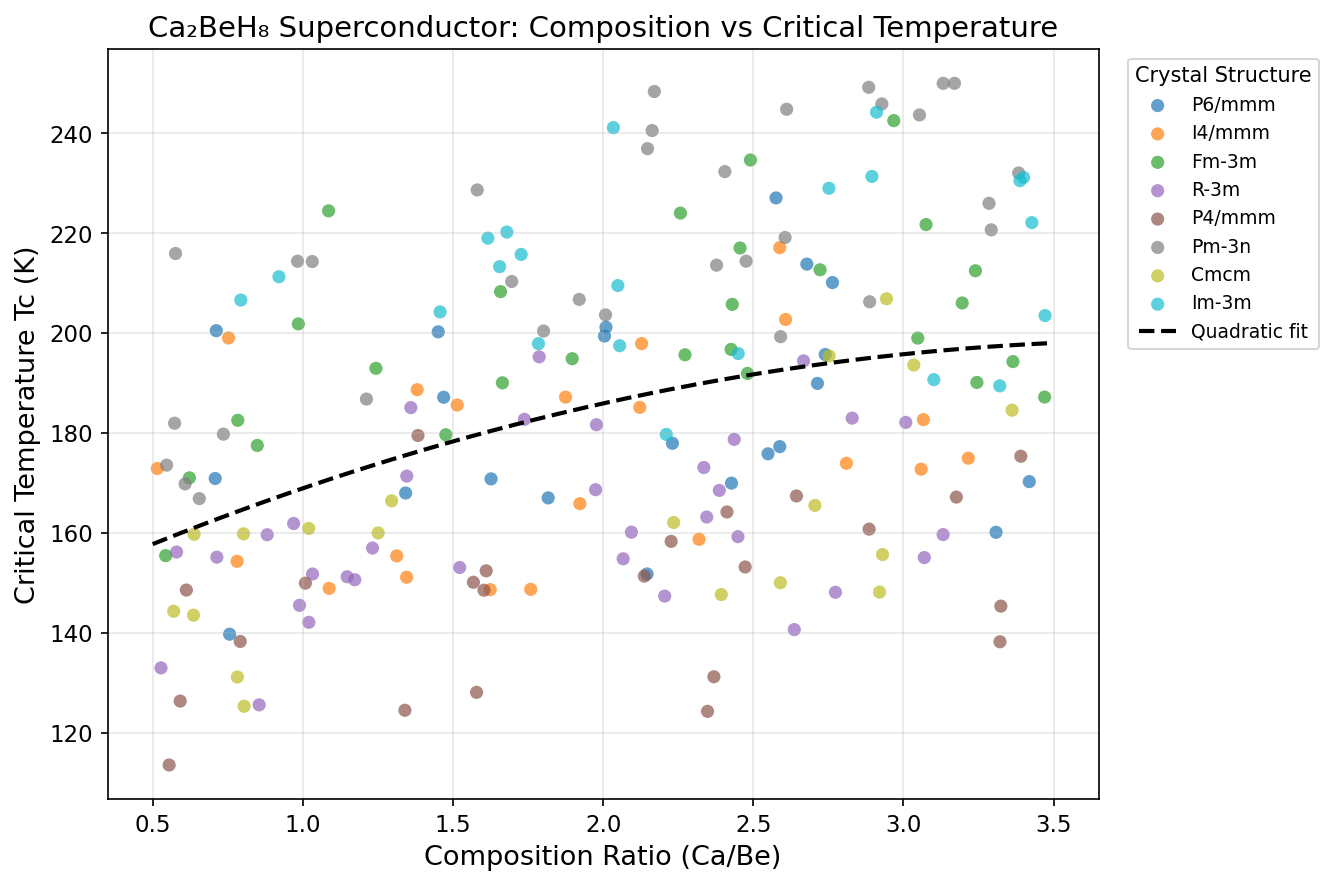
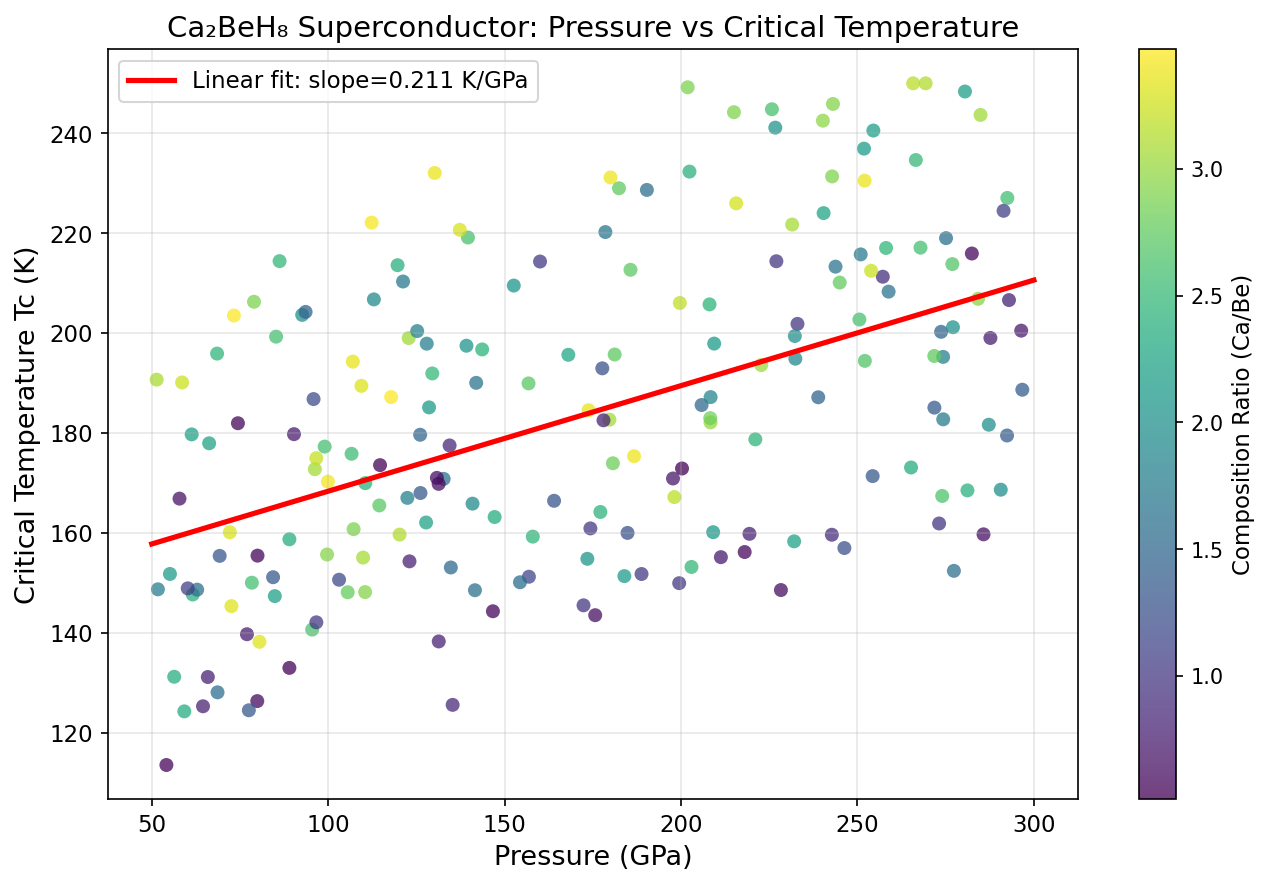
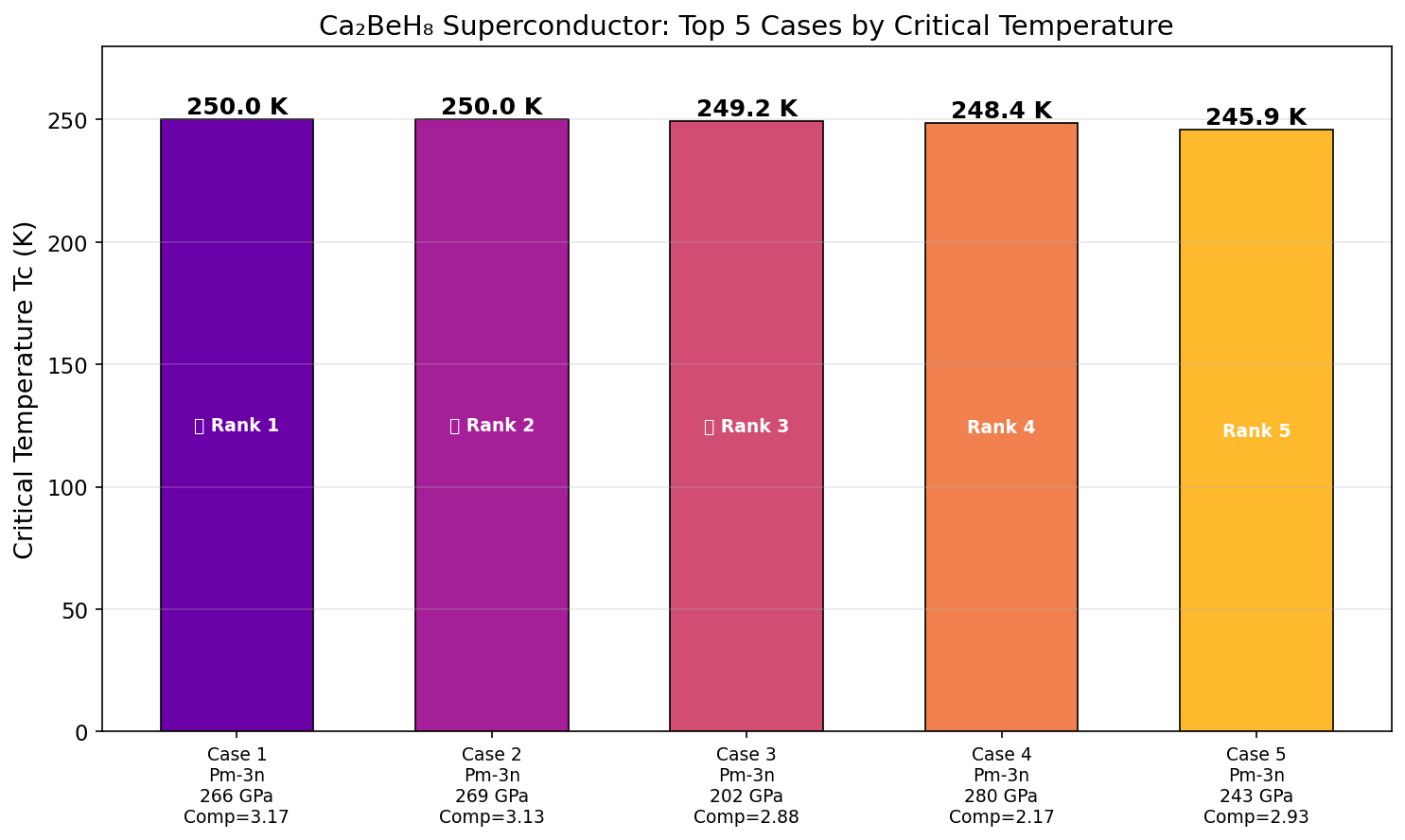
Crystal Structure and Bonding
The crystalline architecture of Ca₂BeH₈ is where its superconducting magic truly begins. Under the extreme pressure of 145.6 GPa, the compound adopts a high-symmetry structure in which beryllium atoms sit at the center of hydrogen polyhedra, while calcium atoms occupy positions that stabilize the overall lattice. Computational predictions suggest a cubic or near-cubic arrangement in which each beryllium atom is coordinated by eight hydrogen atoms, forming a [BeH₈] complex anion that serves as the fundamental building block of the material.
This arrangement is not accidental — it is the direct consequence of a delicate balance between ionic, covalent, and metallic bonding character. Calcium donates electrons to the hydrogen-beryllium framework, giving the structure an ionic flavor. Within the [BeH₈] units, however, the Be-H bonds display strong covalent character, with significant orbital overlap that creates well-defined electronic states near the Fermi level. The remaining electrons delocalize across the lattice, providing the metallic conductivity that superconductivity requires as its starting point.
Why does this structural motif enable superconductivity so effectively? Several factors converge:
- High hydrogen density: With eight hydrogen atoms per formula unit, Ca₂BeH₈ packs hydrogen tightly enough to behave almost like metallic hydrogen, long theorized to be a room-temperature superconductor.
- Caged hydrogen geometry: The polyhedral arrangement of hydrogen around beryllium creates multiple vibrational modes — symmetric breathing modes, bending modes, and stretching modes — all of which can couple productively with electrons.
- Electronic density of states: The Fermi level sits near a peak in the electronic density of states, meaning plenty of electrons are available to participate in Cooper pairing.
- Dynamical stability: Phonon calculations confirm no imaginary frequencies at the predicted pressure, indicating the structure is mechanically stable and not prone to spontaneous collapse.
In essence, Ca₂BeH₈ occupies a compositional and structural sweet spot where chemistry, geometry, and quantum mechanics all align in favor of high-temperature superconductivity.
Comparison with Known Superconductors
To properly appreciate where Ca₂BeH₈ sits in the superconductor landscape, it helps to measure it against the field's most celebrated materials. Each of these compounds represents a different chapter in the ongoing hunt for practical high-temperature superconductivity:
- H₃S (hydrogen sulfide): The material that reignited the hydride superconductor revolution in 2015, with a Tc of approximately 203 K — but only at a crushing 155 GPa. Ca₂BeH₈ trades some critical temperature for a slightly more accessible pressure and a more chemically versatile composition.
- LaH₁₀ (lanthanum decahydride): A clathrate-like hydride with a remarkable Tc of around 250–260 K at 170–190 GPa. While LaH₁₀ still holds the crown for highest confirmed Tc among hydrides, its pressure requirements remain prohibitive, and its rare-earth content raises cost and sustainability questions. Ca₂BeH₈ uses lighter, more abundant elements.
- MgB₂ (magnesium diboride): A conventional superconductor discovered in 2001, with a Tc of 39 K at ambient pressure. MgB₂ remains a practical workhorse for applications because it works without extreme compression, but its Tc is far below what Ca₂BeH₈ offers in principle.
- Cuprate superconductors (YBCO, BSCCO): Copper-oxide materials that superconduct above 77 K (liquid nitrogen temperature) at ambient pressure. These remain the current industrial standard, but their unconventional pairing mechanism is still not fully understood, and fabricating them into reliable wires is notoriously difficult.
- Conventional low-Tc superconductors (Nb, NbTi, Nb₃Sn): The materials used in MRI machines and particle accelerators today, with critical temperatures below 20 K. Reliable but thirsty for liquid helium.
Against this backdrop, Ca₂BeH₈'s predicted 144.7 K Tc at 145.6 GPa positions it as a compelling mid-tier candidate: not the highest Tc on record, but notable for combining strong superconductivity with lighter elemental constituents and, crucially, a pressure that — while still extreme — is at the lower end of the hydride superconductor spectrum.
Experimental Validation Roadmap
Computational predictions, no matter how elegant, only tell half the story. The other half belongs to experimentalists working with diamond anvil cells, synchrotron beamlines, and cryogenic measurement systems. Confirming Ca₂BeH₈'s predicted properties will require a sequenced series of experiments, each answering a specific question:
- Synthesis under pressure: The first step is actually creating Ca₂BeH₈. Researchers would typically load precursor materials — perhaps calcium hydride, beryllium hydride, and excess hydrogen — into a diamond anvil cell and compress them to the target pressure. Laser heating would then drive the reaction to form the predicted stoichiometry.
- Structural characterization: X-ray diffraction performed at synchrotron facilities would verify that the synthesized material adopts the predicted crystal structure. Any deviation in lattice parameters or symmetry would prompt a re-examination of the theoretical model.
- Electrical transport measurements: The hallmark test for superconductivity is a resistance drop to zero below Tc. Four-probe resistance measurements inside the diamond anvil cell, performed as temperature is lowered through the predicted critical region, would provide the first direct evidence.
- Magnetic susceptibility and the Meissner effect: True superconductors expel magnetic fields. Detecting diamagnetic screening through sensitive magnetometry would rule out alternative explanations like a metal-insulator transition or contact artifacts.
- Isotope effect experiments: Replacing hydrogen with deuterium should shift Tc in a predictable way if electron-phonon coupling is indeed the driving mechanism. This is a classic test for conventional phonon-mediated superconductivity.
- Spectroscopic probes: Techniques like inelastic X-ray scattering can directly measure phonon dispersions, allowing experimentalists to compare observed lattice vibrations with those predicted by theory.
Historically, the full validation cycle for a new hydride superconductor takes anywhere from two to five years after initial prediction, depending on synthesis difficulty and access to specialized equipment. The community will be watching closely.
Implications for the Field
Beyond the specific case of Ca₂BeH₈, this prediction carries broader significance for the trajectory of superconductor research. For most of the 20th century, progress in raising critical temperatures came in incremental steps — a few degrees here, a new material family there. The advent of hydride superconductors has dramatically accelerated that progress, and computational-first discovery has become the engine driving it.
Ternary hydrides like Ca₂BeH₈ represent a particularly exciting frontier because they vastly expand the compositional search space. Binary hydrides such as H₃S and LaH₁₀ combine hydrogen with just one other element, which inherently limits the tuning knobs available to optimize properties. By introducing a third element, researchers gain new degrees of freedom: they can balance electronegativity, tune electronic structure, and engineer phonon spectra more precisely. Ca₂BeH₈'s beryllium-mediated phonon bridging is a perfect example of how a carefully chosen third element can enable physics that neither binary component could achieve alone.
This prediction also feeds into the grand quest for ambient-pressure, room-temperature superconductivity — the holy grail that would revolutionize power transmission, transport

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)