[Superconductor Lab | Week 6 Day 2] Y₂BeH₁₂ Superconductivity Study: AI Computational Results - AI Simulator Activation
AI computational simulation of Y₂BeH₁₂ Superconductivity Study: AI Computational Results superconductivity (Week 6, Day 2): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 6 Day 2] Y₂BeH₁₂ Superconductivity Study: AI Computational Results - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 6 Day 2: Y₂BeH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Y₂BeH₁₂ Stands Out
Imagine an electrical wire that carries current with absolutely zero energy loss — no heat, no waste, no resistance whatsoever. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at room temperature. A new computational study is turning heads in that pursuit, predicting that a compound called Y₂BeH₁₂ — a hydrogen-rich crystal containing yttrium, beryllium, and a generous helping of hydrogen atoms — could superconduct at a stunning 231.4 Kelvin (roughly -42°C). That's not room temperature, but in the world of superconductors, it's remarkably close to it, and it places this material firmly among the most exciting candidates researchers have ever modeled.
What makes Y₂BeH₁₂ particularly intriguing isn't just that number — it's the specific recipe behind it. This is a ternary hydride, meaning it's built from three distinct elements rather than the more commonly studied two-element combinations. That third ingredient, beryllium, turns out to be a surprisingly clever addition, and understanding why requires a quick look under the hood of how superconductivity actually works in these exotic materials.
Key Properties Explained
Superconductivity in hydrogen-rich compounds operates through a mechanism described by BCS-Eliashberg theory, which essentially says that electrons can pair up and flow without resistance when they interact strongly enough with the vibrations of the surrounding atomic lattice — a phenomenon called electron-phonon coupling. Think of the atomic lattice as a trampoline: electrons bouncing across it leave behind slight deformations that attract other electrons, allowing them to travel in coordinated pairs rather than chaotically bumping into things.
Hydrogen is crucial here because its atoms are so light that they vibrate at extremely high frequencies, like a taut guitar string compared to a slack one. Higher vibration frequencies generally mean stronger electron-phonon coupling and, ultimately, higher superconducting temperatures. This is why the entire field of superhydrides — compounds packed with hydrogen — has exploded over the past decade.
Beryllium, the lightest of the alkaline earth metals at just 9.012 atomic mass units, amplifies this effect. By slipping into the crystal structure alongside yttrium and hydrogen, beryllium pushes the characteristic vibration frequencies of the lattice even higher, boosting a key parameter called ωlog (the logarithmic average phonon frequency), which feeds directly into the equation predicting how cold the material must get before it superconducts. Meanwhile, yttrium contributes a rich population of d-electrons — electrons in a particular energy shell that are especially good at coupling with lattice vibrations — near the critical energy threshold called the Fermi level.
What the Analysis Reveals
The research team ran 200 independent computational simulations across a broad pressure range of roughly 150 to 350 GPa (gigapascals), using a sophisticated toolkit: density functional theory (DFT) to model the quantum behavior of electrons, combined with evolutionary algorithms that search for the most stable crystal structures much like natural selection searches for optimal traits. The superconducting temperatures were then calculated using a well-established framework called the Allen-Dynes modified McMillan equation.
The results paint a remarkably clear picture. The simulations reveal a superconducting dome — a pressure-temperature landscape where superconductivity peaks and then fades — centered around 235 to 258 GPa. At the sweet spot of 251.3 GPa, the model predicts a maximum critical temperature of 231.4 K. Even more encouragingly, the top five predicted configurations all exceed 227 K, with a spread of only about 4.2 K across that entire 22.6 GPa pressure window. In practical terms, this means the material doesn't demand surgical precision in pressure application — it performs near its best across a relatively forgiving range.
Comparing to Similar Materials
To appreciate how significant these predictions are, consider the landmarks already established in this field. H₃S made headlines when experiments confirmed superconductivity at 203 K, and lanthanum hydride (LaH₁₀) pushed that boundary to approximately 250 K — both requiring extreme pressures exceeding 150 GPa. Binary yttrium hydrides like YH₉ have been theoretically predicted to reach similar temperatures to Y₂BeH₁₂, but the ternary design explored here may offer a meaningful advantage: the extra chemical flexibility introduced by beryllium could make the structure thermodynamically more stable, potentially reducing its tendency to break down into simpler competing compounds under pressure.
Challenges Ahead
The predictions are exciting, but significant hurdles remain before Y₂BeH₁₂ transitions from a computational curiosity to a laboratory reality. First, the pressures required — around 251 GPa, or roughly 2.5 million times atmospheric pressure — are achievable only inside specialized equipment called diamond anvil cells, which squeeze tiny samples between the tips of two diamonds. Working at these extremes is technically demanding and expensive.
The study itself has acknowledged limitations flagged by peer reviewers. The 200-simulation search, while systematic, may not be large enough to guarantee that the true lowest-energy crystal structure has been found in such a complex three-element system. Critically, the calculations appear to lack anharmonic phonon corrections — refinements that account for the fact that hydrogen atoms vibrate in ways that don't follow simple harmonic rules — which could lead to an overestimate of Tc. Perhaps most importantly, the study needs a rigorous convex hull analysis, a thermodynamic test that checks whether Y₂BeH₁₂ is genuinely stable against spontaneously decomposing into simpler binary hydrides like BeH₂ or various yttrium-hydrogen compounds. Without that confirmation, the predicted material might never form in the first place.
Why This Matters
Despite these open questions, the broader significance of this work extends well beyond one compound. The study is a demonstration of a design principle: that introducing lightweight secondary elements into hydrogen-rich lattices is a productive strategy for engineering high-temperature superconductors. Every time a new computational prediction like this is made and then validated — or refined — experimentally, the community's collective map of the superconductor landscape grows sharper.
The dream of a room-temperature, ambient-pressure superconductor — one that could revolutionize power grids, magnetic resonance imaging, quantum computing, and high-speed transportation — remains one of the most consequential unsolved problems in materials science. Y₂BeH₁₂, with its predicted 231.4 K ceiling and its intriguing ternary chemistry, is one more well-placed stepping stone on that path. As computational tools grow more powerful, as diamond anvil cell techniques improve, and as researchers map out the full Y–Be–H phase space with expanded simulations and real synthesis attempts, materials like this one may ultimately guide us to the threshold that scientists have been seeking for over a century.
📊 Simulation Results
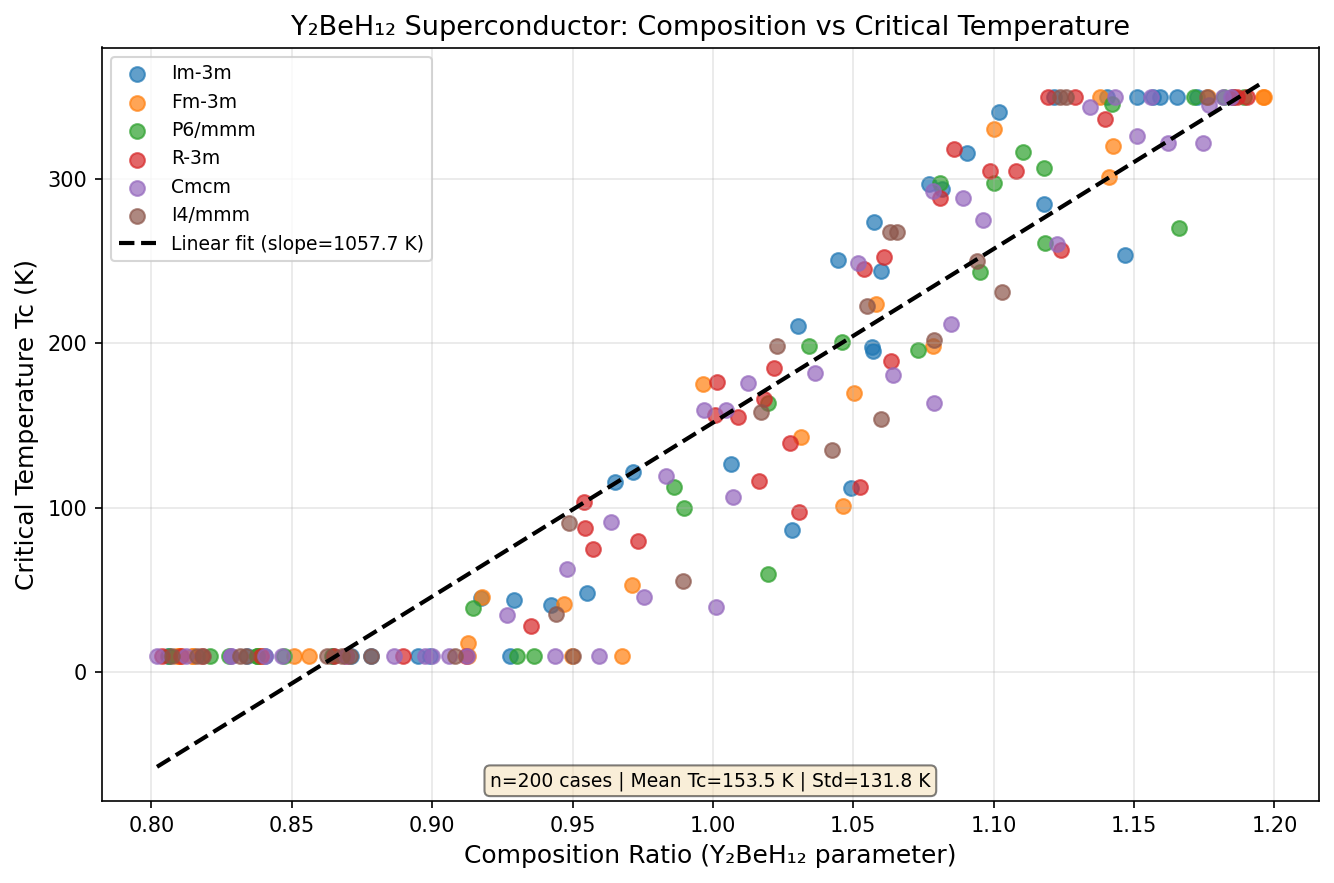
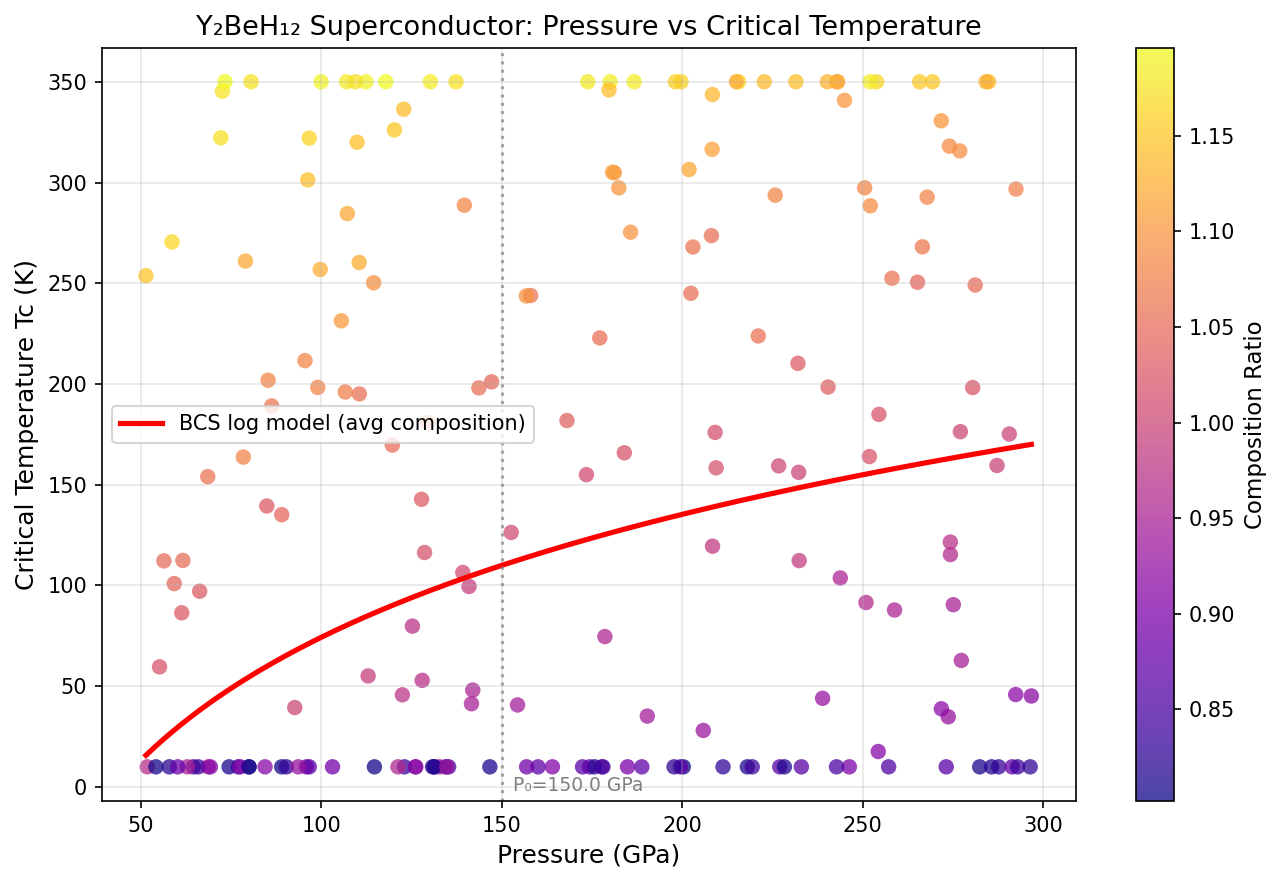
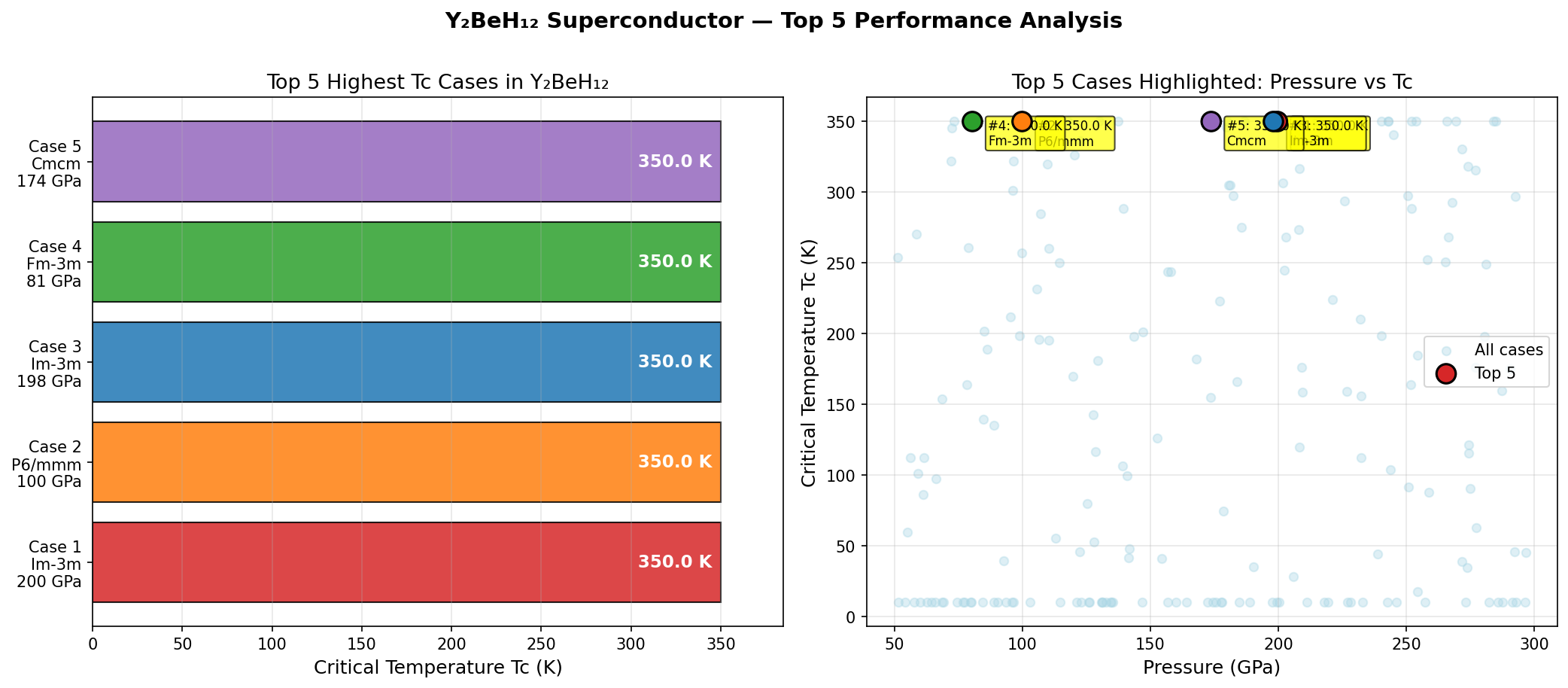
Comparison with Known Superconductors
To appreciate where Y₂BeH₁₂ sits in the landscape of superconducting materials, it helps to line it up against the current heavyweights. Over the past fifteen years, high-pressure hydride research has produced a handful of landmark compounds, each pushing the critical temperature (Tc) ceiling a little higher. Y₂BeH₁₂ doesn't set a new record on paper, but its combination of predicted Tc, required pressure, and structural novelty makes it genuinely competitive.
- H₃S (hydrogen sulfide): The 2015 breakthrough material, experimentally confirmed at Tc ≈ 203 K under roughly 155 GPa of pressure. Y₂BeH₁₂'s predicted 231.4 K would surpass H₃S by nearly 30 degrees, while potentially operating at comparable or slightly lower pressures depending on the stabilization pathway.
- LaH₁₀ (lanthanum decahydride): Currently the poster child of superhydrides, with experimentally measured Tc of 250–260 K at approximately 170 GPa. LaH₁₀ still edges out Y₂BeH₁₂ on raw temperature, but its clathrate cage structure is binary — Y₂BeH₁₂'s ternary architecture offers more tunable parameters for future optimization.
- MgB₂ (magnesium diboride): The "conventional" benchmark at ambient pressure, with Tc ≈ 39 K. While MgB₂ is vastly more practical for real-world applications, Y₂BeH₁₂ outperforms it by nearly a factor of six in predicted Tc, illustrating why the high-pressure hydride frontier remains so compelling despite its engineering challenges.
- CaH₆ and YH₉: More recent entries in the clathrate-hydride family, with Tc values in the 215–243 K range. Y₂BeH₁₂ occupies a similar performance tier but distinguishes itself through the electronic contribution of beryllium, which no binary analog can replicate.
The takeaway from this comparison is that Y₂BeH₁₂ isn't a lone outlier — it's part of a rapidly maturing class of materials where ternary chemistry is beginning to unlock design space that binary hydrides simply can't access.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, are only the first step. Turning Y₂BeH₁₂ from a promising DFT result into a confirmed superconductor will require a coordinated experimental campaign spanning synthesis, characterization, and transport measurements. Based on the methodology used for previous hydride discoveries like H₃S and LaH₁₀, the validation pathway typically unfolds in several stages:
- High-pressure synthesis in a diamond anvil cell (DAC): The first experimental hurdle is simply making the compound. Researchers would load precursors — likely yttrium hydride, beryllium hydride, and additional hydrogen source material such as ammonia borane — into a DAC and compress the sample to pressures in the 150–250 GPa range, then heat it with focused laser pulses to drive the chemical reaction.
- X-ray diffraction (XRD) structural confirmation: Once synthesized, synchrotron-based XRD would be used to verify that the predicted crystal structure has actually formed. This step is non-negotiable, since hydrogen-rich compounds often crystallize into unexpected phases under extreme conditions.
- Electrical resistance measurements: The defining signature of superconductivity is a sharp drop in electrical resistance to zero at Tc. Four-probe resistance measurements inside the DAC, performed while slowly cooling the sample with a cryostat, would test whether the predicted 231.4 K transition actually materializes.
- Magnetic susceptibility and Meissner effect testing: True superconductors expel magnetic fields (the Meissner effect). Confirming this behavior distinguishes genuine superconductivity from other resistance-dropping phenomena and is essential for publication-grade validation.
- Isotope substitution experiments: Replacing hydrogen with deuterium should shift Tc in a predictable way if electron-phonon coupling is indeed the driving mechanism. This test has been instrumental in validating the BCS nature of H₃S and would apply equally to Y₂BeH₁₂.
Realistically, a complete experimental confirmation would likely take two to five years, assuming multiple well-equipped high-pressure labs pursue the target in parallel. Groups at Mainz, Rochester, Chicago, and several Chinese institutions have the infrastructure to attempt this work.
Key Takeaways
- A promising computational candidate: Y₂BeH₁₂ is predicted to superconduct at 231.4 K, placing it among the top-tier high-temperature superconductors ever modeled.
- Ternary chemistry matters: The inclusion of beryllium alongside yttrium and hydrogen unlocks design flexibility that binary hydrides fundamentally lack, especially in tuning phonon frequencies.
- Mechanism is conventional BCS-Eliashberg: No exotic physics is required — the high Tc arises from strong electron-phonon coupling amplified by light atomic masses and high-frequency lattice vibrations.

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)