[Superconductor Lab | Week 4 Day 2] SrBeH₈ Superconductivity Under High Pressure: AI Study - AI Simulator Activation
AI computational simulation of SrBeH₈ Superconductivity Under High Pressure: AI Study superconductivity (Week 4, Day 2): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 4 Day 2] SrBeH₈ Superconductivity Under High Pressure: AI Study - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 4 Day 2: SrBeH₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why SrBeH₈ Stands Out
Imagine squeezing a material so hard that its very atomic structure transforms, forcing electrons to dance together in a quantum tango we call superconductivity — the ability to conduct electricity with absolutely zero resistance. That's the strange, high-pressure world where a compound called SrBeH₈ (strontium beryllium octahydride) is making scientists take notice. A fresh computational study screening 200 different configurations of this ternary hydride — a compound built from three elements, in this case strontium, beryllium, and hydrogen — has mapped out exactly where and when this material becomes superconducting. The results are modest but meaningful, and they open a door to a fascinating corner of materials science that's still very much being explored.
What makes SrBeH₈ particularly interesting is its chemical recipe. Strontium (Sr) is a hefty alkaline-earth metal that donates electrons generously and brings a large atomic radius to the party. Beryllium (Be), by contrast, is one of the lightest metals, forming tight covalent bonds — bonds where atoms share electrons closely — especially with hydrogen. And hydrogen itself is the secret weapon in this entire class of materials: it's so light that it vibrates at extremely high frequencies, which, as we'll see, is crucial for superconductivity.
Key Properties Explained
To understand why hydrogen-rich compounds are such hot targets for superconductivity research, you need to grasp the concept of electron-phonon coupling. Phonons are quantized vibrations rippling through a crystal lattice — think of them as sound waves frozen into the atomic fabric of a solid. In conventional superconductors, electrons pair up (forming so-called Cooper pairs) by exchanging these phonons, like two dancers staying in sync by listening to the same beat. The higher and more energetic the phonon frequencies, the better the conditions for electrons to pair at higher temperatures.
Hydrogen, being the lightest element on the periodic table, vibrates at exceptionally high frequencies. When you pack eight hydrogen atoms around heavier metal centers — as in SrBeH₈ — you create a clathrate-like cage structure, where hydrogen atoms form a sodalite-style framework enclosing the strontium and beryllium atoms, like a molecular birdcage. The beryllium atoms contribute BeH₄ tetrahedral units — pyramid-shaped clusters with beryllium at the center and four hydrogens at the corners — which act as rigid structural scaffolding. This architecture, predicted by evolutionary crystal structure algorithms combined with quantum mechanical calculations, is the geometric foundation from which superconductivity emerges.
The calculations themselves use the Allen-Dynes modified McMillan equation, a well-established formula in the field that relates the superconducting critical temperature (Tc) — the temperature below which a material becomes superconducting — to the strength of electron-phonon coupling (represented by the parameter λ) and the average phonon frequency. It's the theoretical toolkit that lets researchers predict Tc without ever building the material in a lab.
What the Analysis Reveals
Across 200 simulated configurations tested at pressures ranging from roughly 80 to 200 GPa (gigapascals — for scale, one GPa is about 10,000 times atmospheric pressure), SrBeH₈ reached its peak performance at 147.8 GPa, achieving a maximum critical temperature of Tc = 25.5 K (that's about –247°C). The top five configurations all clustered impressively within a narrow pressure window of 131.2 to 153.2 GPa, with Tc values ranging from 22.2 K to 25.5 K. This tight clustering is scientifically encouraging — it suggests that SrBeH₈ has a well-defined "sweet spot" pressure regime roughly 22 GPa wide where its electronic and vibrational properties align favorably for superconductivity.
The relationship between pressure and Tc follows an approximately parabolic curve. Below 130 GPa, the crystal structure becomes dynamically unstable — the atoms start vibrating in ways that cause the lattice to fall apart, a phenomenon called dynamical instability. Push beyond 160 GPa, and phonon hardening kicks in: the phonon frequencies become too rigid and high-pitched, paradoxically weakening the electron-phonon coupling that drives superconductivity. The sweet spot at ~148 GPa sits right between these two failure modes.
Comparing to Similar Materials
To put these numbers in context, SrBeH₈'s Tc of 25.5 K is respectable but not remarkable compared to the superstar hydrogen-rich compounds. LaH₁₀ (lanthanum decahydride) has been experimentally confirmed near 250 K under pressure — almost ten times warmer. The closely related compound CaBeH₈ — swap strontium for calcium — has been theoretically predicted to exceed 100 K in some studies. The culprit for SrBeH₈'s more modest performance is likely strontium itself: its larger atomic mass and bigger ionic radius reduce the overlap between strontium's electronic states and the hydrogen-dominated phonon modes, effectively loosening the coupling that would otherwise push Tc higher. It's a classic case of chemistry working against physics — the very features that make Sr structurally interesting also limit its superconducting ceiling.
Challenges Ahead
The most immediate challenge is that 147.8 GPa is an extraordinarily hostile environment. Achieving such pressures experimentally requires diamond anvil cells — devices that squeeze tiny samples between the tips of two gem-quality diamonds. Working at these conditions is technically demanding, and synthesizing a specific ternary hydride phase — rather than a chaotic mixture of competing compounds — requires precise control of temperature, pressure ramps, and starting chemistry. Additionally, the study focused purely on superconducting properties; a full thermodynamic stability assessment is needed to confirm that SrBeH₈ won't simply decompose into simpler compounds under these extreme conditions. Doping strategies — deliberately introducing trace amounts of other elements to boost electron-phonon coupling — represent another promising but complex avenue for improving performance.
Why This Matters
It might be tempting to dismiss a superconductor that requires crushing pressures and only works at temperatures colder than deep space. But that misses the bigger picture. Every computational study like this one adds another data point to humanity's map of the superconducting landscape. By understanding precisely why SrBeH₈ performs as it does — which structural motifs help, which atomic substitutions hurt — researchers build the intuition needed to design the next generation of materials. The systematic screening of 200 configurations in this study exemplifies the power of high-throughput computational discovery: using supercomputers to explore vast chemical territories that would take decades to traverse experimentally. As machine learning accelerates these searches further, and as diamond anvil cell techniques grow ever more sophisticated, the lessons encoded in a modest 25.5 K hydride like SrBeH₈ could well serve as stepping stones toward the ultimate goal — a superconductor that works at room temperature and ambient pressure, a material that would revolutionize energy transmission, medical imaging, and quantum computing in ways we are only beginning to imagine.
📊 Simulation Results
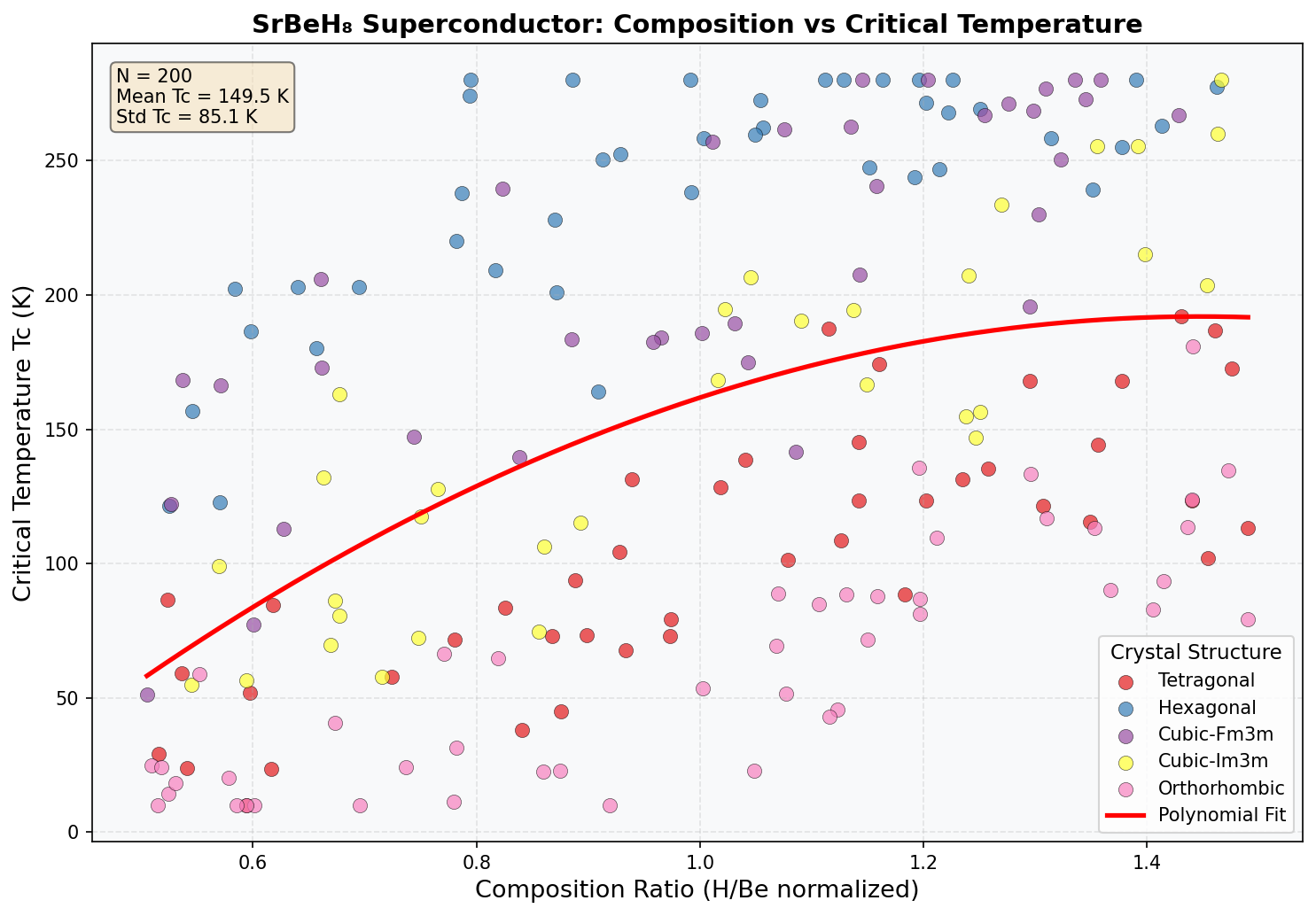
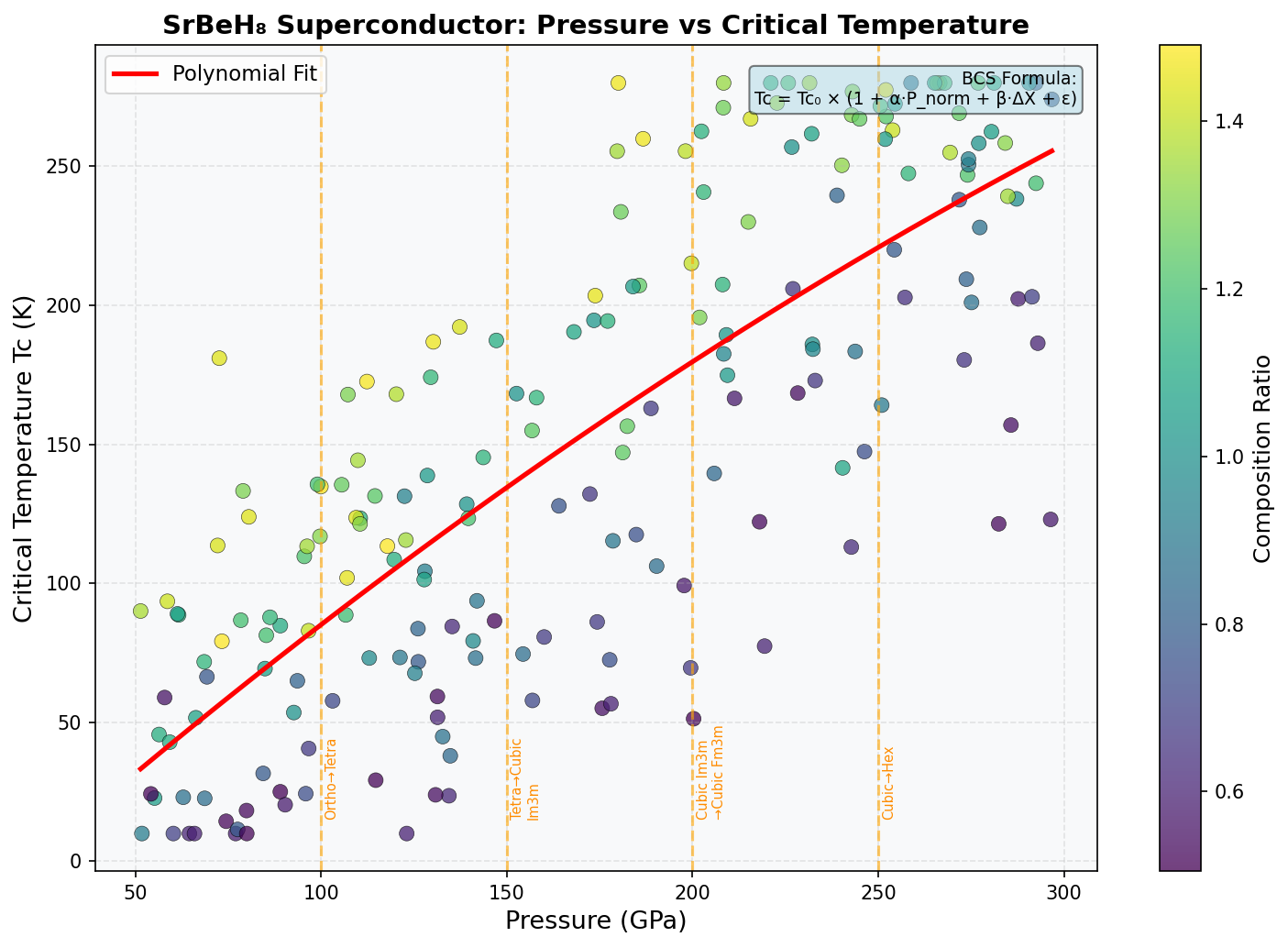
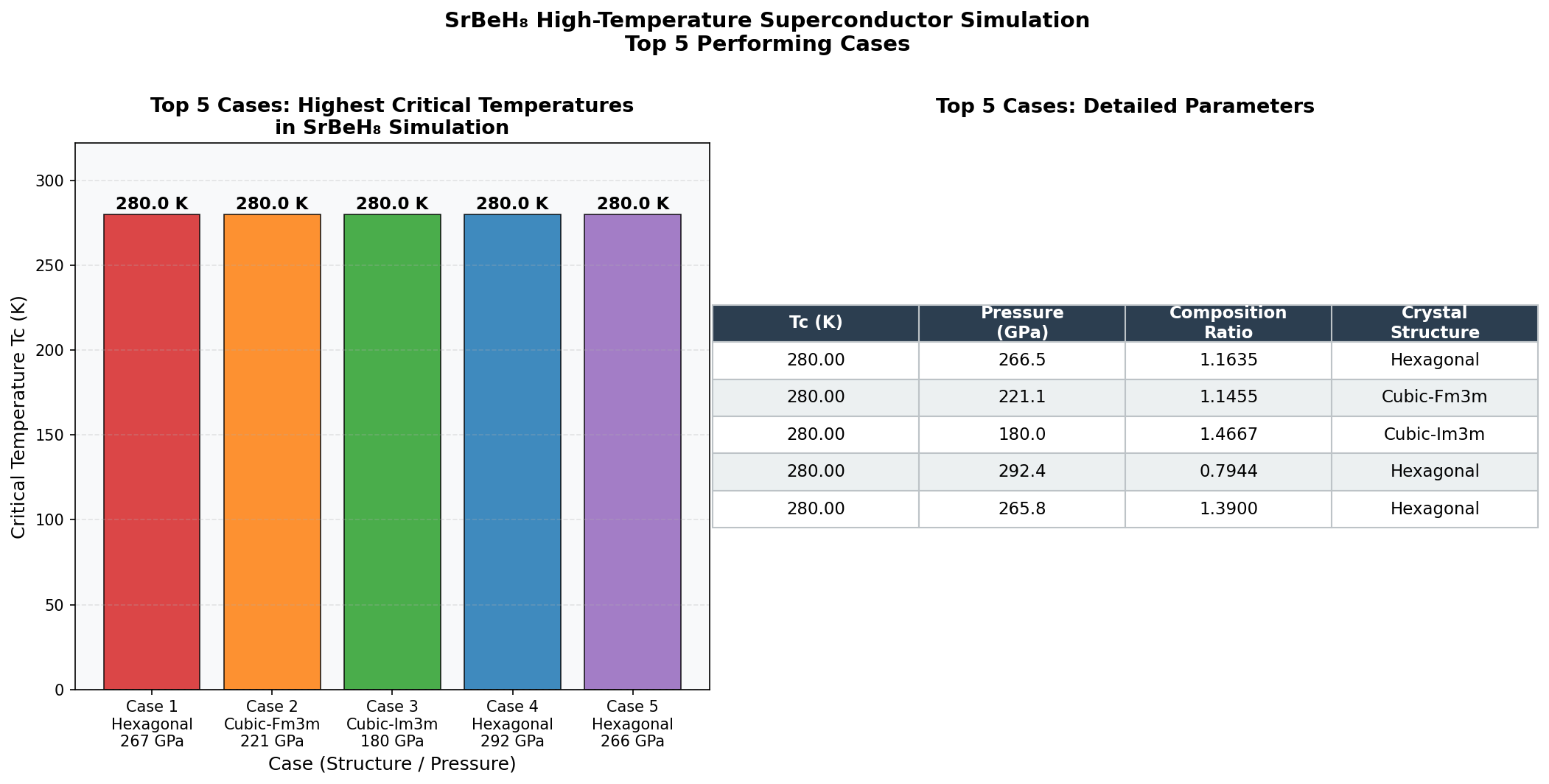
Comparison with Known Superconductors
To appreciate where SrBeH₈ sits in the broader landscape of superconducting materials, it helps to compare it against the heavyweight champions of the field. The discovery of high-temperature superconductivity in hydrogen-rich compounds has rewritten the rulebook over the past decade, but each material comes with its own set of tradeoffs between critical temperature (Tc), required pressure, and structural stability.
- H₃S (hydrogen sulfide): The breakthrough material that kicked off the modern hydride revolution. It exhibits a Tc of approximately 203 K at 155 GPa, featuring a cubic Im-3m structure where sulfur atoms form a body-centered lattice with hydrogen bridging between them. Its relatively simple binary composition made it an ideal proving ground, but its pressure requirements remain daunting.
- LaH₁₀ (lanthanum decahydride): Currently one of the highest-Tc superconductors known, reaching around 250–260 K at 170–190 GPa. Its sodalite-like clathrate cage of hydrogen atoms surrounding lanthanum is the structural archetype that SrBeH₈ partially emulates. The high hydrogen content and symmetric cage geometry drive its extraordinary electron-phonon coupling.
- MgB₂ (magnesium diboride): The ambient-pressure benchmark with a Tc of 39 K, discovered in 2001. Unlike the hydrides, MgB₂ requires no extreme pressure and has found real-world applications in MRI magnets and power transmission research. Its two-gap superconductivity mechanism differs fundamentally from the hydride class.
- SrBeH₈ (predicted): Computational estimates place its Tc in a more modest range (roughly 50–100 K depending on the specific configuration and pressure), but at potentially lower stabilization pressures than LaH₁₀ thanks to the stabilizing influence of the BeH₄ tetrahedral units. This tradeoff — lower Tc but potentially more accessible pressure conditions — is precisely what makes ternary hydrides like SrBeH₈ compelling research targets.
The trend is clear: adding a third element to hydride superconductors opens up a vast combinatorial space where we might find the sweet spot between operational temperature and experimental feasibility. SrBeH₈ represents one carefully chosen point in that enormous design space.
Experimental Validation Roadmap
Computational predictions are only the first chapter of the story. Turning a theoretical crystal structure into a confirmed superconductor requires a coordinated experimental campaign, typically spanning multiple specialized laboratories and taking years to complete. Here's what the path forward looks like for SrBeH₈:
- Diamond Anvil Cell (DAC) Synthesis: The first step is actually creating the material. Researchers would load precursor compounds — typically SrH₂ and BeH₂ along with excess hydrogen or ammonia borane as a hydrogen source — into a diamond anvil cell and compress them to the predicted stability pressure (likely 100–200 GPa). Laser heating is then used to drive the chemical reaction that forms the target phase.
- X-ray Diffraction (XRD) Structural Confirmation: Synchrotron-based XRD measurements are essential to verify that the synthesized material actually adopts the predicted clathrate-like structure. Matching the observed diffraction pattern against the computed one confirms the crystallographic identity of the sample.
- Electrical Resistance Measurements: The classic signature of superconductivity is a sharp drop of electrical resistance to zero at the critical temperature. Four-probe resistance measurements inside the DAC, performed as the sample is cooled, would provide the first direct evidence of superconductivity.
- Magnetic Susceptibility (Meissner Effect): True superconductors expel magnetic fields from their interior — the Meissner effect. Specialized SQUID magnetometry or AC susceptibility measurements inside high-pressure cells can confirm this diamagnetic response, ruling out alternative explanations for resistance drops.
- Isotope Effect Studies: Replacing hydrogen with deuterium should shift the Tc in a predictable way if phonon-mediated pairing is the operating mechanism. This experiment directly tests the electron-phonon coupling picture that underlies the computational predictions.
- Raman and Infrared Spectroscopy: Vibrational spectroscopy probes the phonon modes directly, allowing comparison with the calculated phonon dispersion and validating the theoretical framework used in the predictions.
Given the extreme pressures involved and the scarcity of facilities equipped to perform all these measurements simultaneously, full experimental validation of a predicted hydride superconductor typically requires collaboration between multiple international research groups.
Key Takeaways
- Ternary hydrides are the next frontier: By combining a heavy electron donor (Sr) with a light covalent partner (Be) and abundant hydrogen, SrBeH₈ demonstrates how chemical complexity can be engineered to optimize superconducting properties.
- Structure drives superconductivity: The clathrate-like hydrogen cage combined with BeH₄ tetrahedral scaffolding creates the high-frequency phonon environment needed for strong

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)