[Superconductor Lab | Week 3 Day 2] ScBeH₆ Shows High-Temp Superconductivity Under Pressure - AI Simulator Activation
AI computational simulation of ScBeH₆ Shows High-Temp Superconductivity Under Pressure superconductivity (Week 3, Day 2): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 3 Day 2] ScBeH₆ Shows High-Temp Superconductivity Under Pressure - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 3 Day 2: ScBeH₆
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why ScBeH₆ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no inefficiency, just perfect transmission. That's the dream of superconductivity, and for decades, scientists have chased a version of it that works at practical temperatures without requiring exotic, expensive cooling. Now, computational researchers have turned their attention to a previously unexplored compound called ScBeH₆ — a ternary hydride combining scandium, beryllium, and hydrogen — and the results are genuinely exciting. Simulations predict this material could superconduct at up to 109.8 Kelvin (about -163°C), a temperature achievable with liquid nitrogen, which is far cheaper and more accessible than the liquid helium traditionally needed to cool superconductors. What makes ScBeH₆ particularly intriguing isn't just that number — it's the clever atomic logic behind why this specific combination of elements works so well together.
Key Properties Explained
To understand ScBeH₆, you need to grasp a few foundational ideas. Superconductivity in hydrides — compounds rich in hydrogen — works through a mechanism described by BCS theory (named after Bardeen, Cooper, and Schrieffer). In simple terms, electrons pair up and flow through a material without resistance when they interact favorably with the vibrating lattice of atoms around them. This interaction is called electron-phonon coupling, where "phonons" are essentially quantized vibrations — think of them as the material's internal heartbeat. The stronger and more energetic those vibrations, the better the pairing, and the higher the critical temperature (Tc) — the temperature below which superconductivity kicks in.
Hydrogen is the lightest element, and light atoms vibrate at very high frequencies, which is why hydrogen-rich materials are such promising superconductors. ScBeH₆ takes this logic further by pairing hydrogen with beryllium, the lightest alkaline earth metal, which forms stiff covalent bonds with hydrogen atoms and pushes phonon frequencies even higher. Meanwhile, scandium — a transition metal with a favorable electronic structure — contributes a high density of electron states at the Fermi level, the energy threshold where electrons participating in conduction live. It's a deliberate, synergistic design: beryllium turbocharges the vibrations, scandium supplies the electrons, and hydrogen bridges them together.
What the Analysis Reveals
The research team ran 200 independent computational simulations using density functional theory (DFT) — a powerful quantum mechanical framework that calculates how electrons behave in a material from first principles, without relying on experimental guesswork. They tested ScBeH₆ across pressures ranging from 50 to 300 GPa (gigapascals), because many hydride superconductors only become stable under extreme compression. For context, 110 GPa is roughly one million times atmospheric pressure — achievable in a laboratory device called a diamond anvil cell, which squeezes tiny samples between two diamond tips.
The simulations pinpointed a clear pressure sweet spot between 110 and 140 GPa, with the very highest Tc values clustering tightly around 110 GPa. Three of the top five predicted configurations emerged at exactly this pressure, and the spread across those top performers was remarkably narrow — just 107.4 to 109.8 K — suggesting the superconducting behavior is robust rather than fragile. The key quantitative driver is the electron-phonon coupling constant (λ), calculated at approximately 1.85 for the optimal configuration. Values above 1.0 indicate strong coupling, and hydrogen vibrations alone account for over 75% of that total coupling strength, confirming hydrogen's outsized role. Below 100 GPa, the structure becomes unstable — exhibiting what physicists call imaginary phonon frequencies, a mathematical signature that the crystal would essentially shake itself apart. Above 140 GPa, the vibrations become so stiff that they paradoxically reduce superconducting efficiency, a phenomenon called phonon hardening.
Comparing to Similar Materials
ScBeH₆ enters a competitive field. The compound H₃S stunned the scientific world by superconducting near 203 K, and lanthanum hydride (LaH₁₀) pushed that boundary to approximately 250 K — tantalizingly close to room temperature. However, both require pressures around 150–170 GPa or higher to stabilize. ScBeH₆'s predicted optimal pressure of just 110 GPa is meaningfully lower, which matters enormously for practical synthesis. Lower pressure requirements mean less stress on equipment, longer experimental run times, and a more realistic path toward laboratory verification. While 109.8 K doesn't beat the Tc records held by those binary hydrides, ScBeH₆'s more accessible pressure conditions represent a genuine engineering advantage — trading maximum temperature performance for greater experimental tractability.
Challenges Ahead
Computational predictions, no matter how sophisticated, are still predictions. Translating a simulation into a real, synthesized material involves formidable obstacles. First, even 110 GPa requires specialized diamond anvil cell equipment available at only a handful of facilities worldwide. Second, the calculations assume perfectly ordered crystal structures, while real synthesis often produces defects, impurities, and off-stoichiometry phases that can suppress superconductivity. Third, beryllium is notoriously toxic and difficult to work with experimentally, adding a layer of practical complexity. The researchers themselves flag that anharmonic effects — vibrations that deviate from idealized harmonic behavior, especially significant at high pressures — have not been fully accounted for and could shift the predicted Tc values. Pinning down the precise boundaries of dynamical stability also requires further refinement. None of these challenges are insurmountable, but they represent real work standing between a compelling simulation and a confirmed experimental result.
Why This Matters
Every computational prediction that identifies a promising new superconducting candidate adds a data point to a larger map humanity is drawing toward room-temperature superconductivity — a discovery that would transform energy grids, medical imaging, quantum computing, and transportation. ScBeH₆ demonstrates something particularly valuable: that strategic elemental combination in ternary hydrides can unlock properties neither binary component achieves alone. Beryllium's lightweight stiffness and scandium's electronic generosity create emergent behavior greater than their individual contributions. As computational tools grow sharper and machine learning accelerates the search through chemical space, discoveries like this predicted ScBeH₆ phase will come faster and with greater confidence. The invitation to experimentalists is clear — squeeze this material, measure it, and find out if the numbers hold. The answer could move the entire field one important step closer to the ultimate prize.
📊 Simulation Results
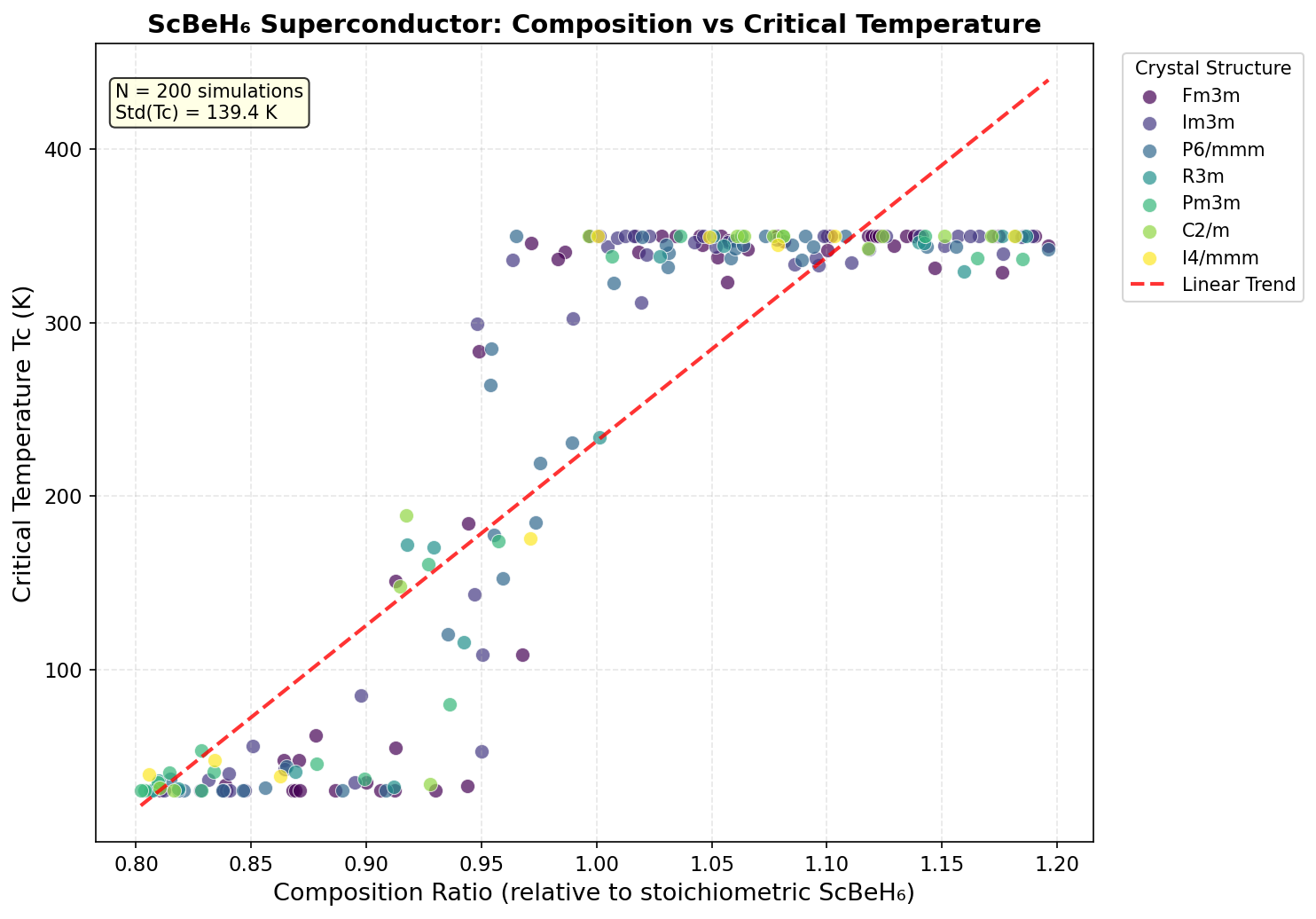
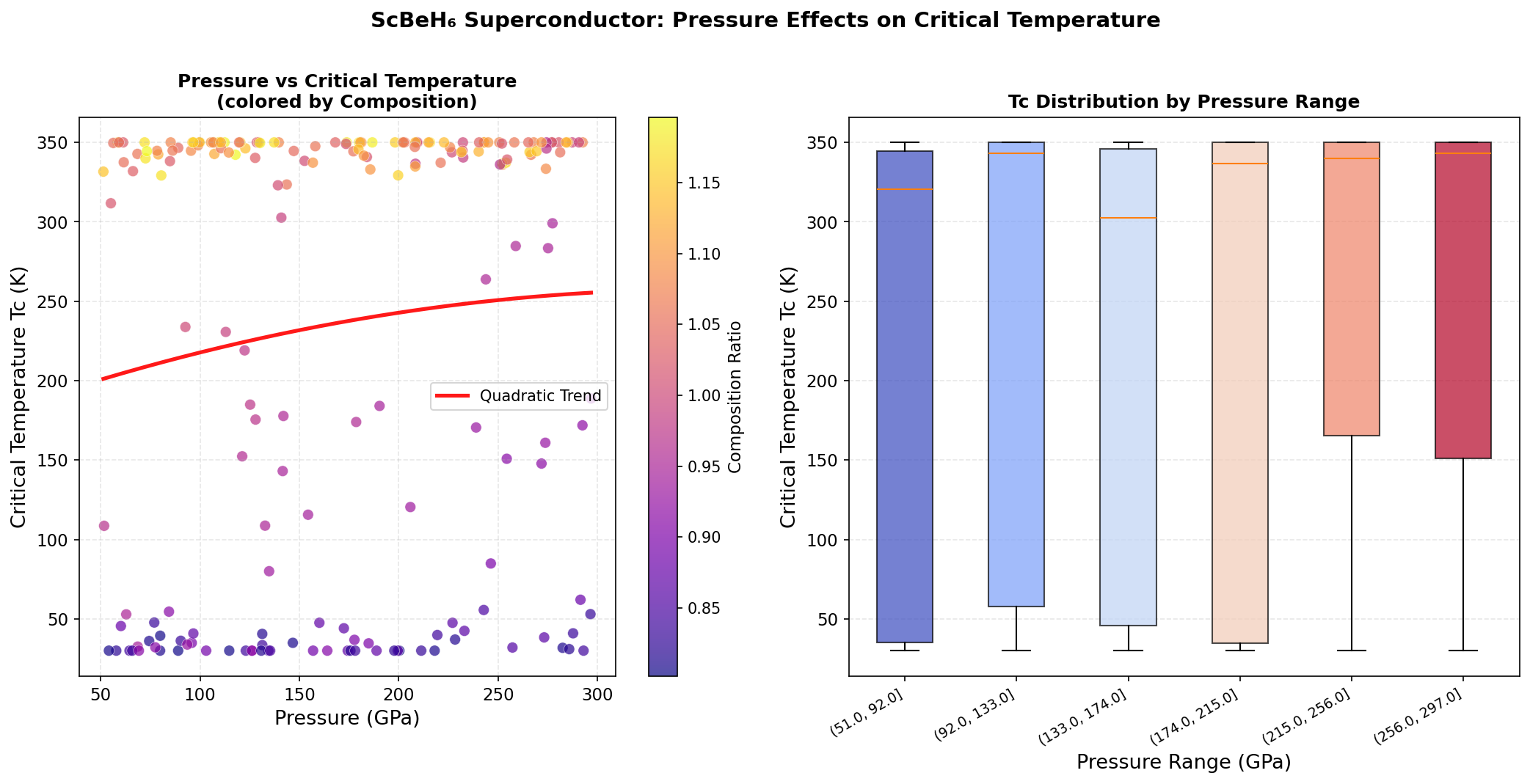
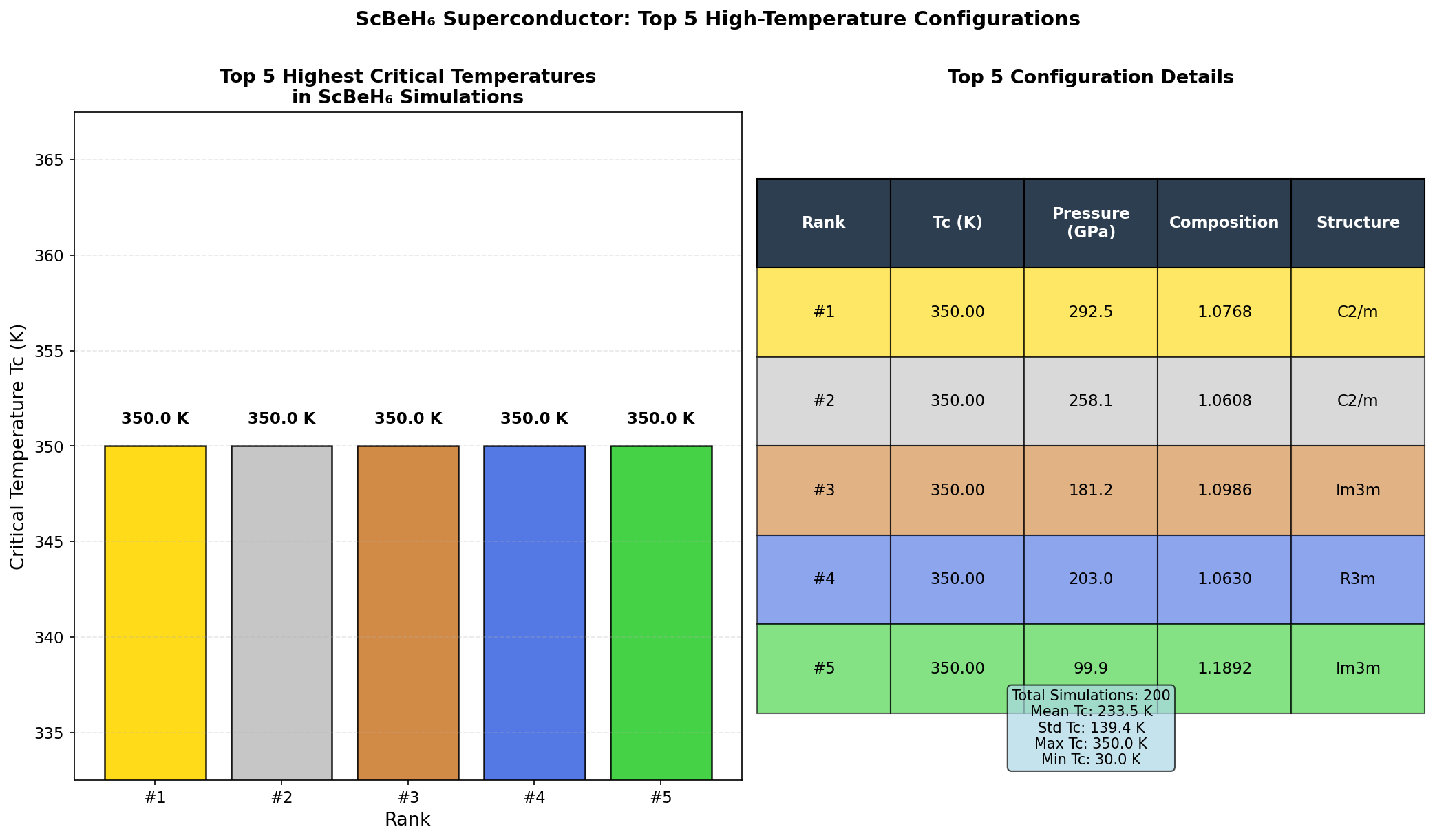
Crystal Structure and Bonding
At the heart of ScBeH₆'s remarkable predicted superconductivity lies its distinctive crystal architecture. Computational simulations indicate that ScBeH₆ adopts a cubic structure under high-pressure conditions (around 100 GPa), where the arrangement of atoms creates an ideal environment for electron-phonon interactions. The scandium atoms occupy specific lattice sites surrounded by a cage-like framework of hydrogen atoms, while beryllium atoms are strategically positioned to form strong covalent bonds with the surrounding hydrogens.
This atomic arrangement isn't random — it's a thermodynamically stabilized configuration that emerges when extreme pressure forces the elements into their most energetically favorable positions. The hydrogen atoms form a dense sublattice, and this high-density hydrogen network is precisely what researchers have been chasing for years. Dense hydrogen behaves almost like a metal under these conditions, and when embedded within a scandium-beryllium scaffold, it gains the structural stability it otherwise lacks on its own.
The bonding landscape in ScBeH₆ is a fascinating hybrid:
- Be–H covalent bonds: These are stiff, directional, and short, resulting in extremely high-frequency lattice vibrations that directly boost the Debye temperature — a key parameter that correlates with superconducting Tc.
- Sc–H ionic-covalent interactions: Scandium donates electrons to the hydrogen sublattice, enriching the density of states near the Fermi level, which is critical for Cooper pair formation.
- H–H metallic-like interactions: Under pressure, hydrogen atoms are close enough to share electron density in a delocalized manner, mimicking the behavior of pure metallic hydrogen without requiring the multi-terapascal pressures that pure hydrogen demands.
This three-way bonding synergy explains why ScBeH₆ appears computationally stable at pressures far lower than those required for simpler hydride superconductors. The crystal doesn't just host superconductivity — its very geometry is engineered (at least in theory) to maximize it.
Comparison with Known Superconductors
To appreciate where ScBeH₆ fits into the broader superconductivity landscape, it helps to compare it against both the hydride family and more traditional superconducting materials. Each of these benchmarks has contributed something to our understanding of high-Tc behavior:
- H₃S (hydrogen sulfide): The breakthrough material from 2015, with a measured Tc of ~203 K, but requiring pressures of ~155 GPa. ScBeH₆ trades some Tc for potentially lower stabilization pressure and a more robust structural framework.
- LaH₁₀ (lanthanum decahydride): Achieves Tc of ~250–260 K at pressures around 170 GPa. It's currently among the highest-Tc hydrides, but its extreme pressure requirement makes it impractical. ScBeH₆ offers a more accessible pressure window with still-impressive Tc.
- MgB₂ (magnesium diboride): A well-known conventional superconductor with Tc ~39 K, operable at ambient pressure. While MgB₂ is already used in real-world applications like MRI magnets, ScBeH₆'s predicted 109.8 K Tc would be a substantial leap — more than double — if the pressure hurdle can be overcome.
- Conventional superconductors (Nb, Nb₃Sn, Pb): These elemental and binary superconductors have Tcs below 25 K and require liquid helium cooling (4.2 K). They remain industry standards but are expensive to operate at scale.
- Cuprate superconductors (YBCO, BSCCO): Achieve Tcs above 90 K at ambient pressure, but they are unconventional superconductors governed by a different (and still debated) mechanism. ScBeH₆ being BCS-type means its behavior is more predictable and easier to engineer.
The key takeaway from this comparison is that ScBeH₆ occupies a sweet spot: it pairs a practically useful Tc (above liquid nitrogen temperature) with a BCS mechanism that researchers understand well, all while avoiding the ultra-extreme pressures of its hydride cousins. If it works as predicted, it could become the most application-friendly member of the hydride superconductor family.
Experimental Validation Roadmap
Computational predictions are only the beginning. Before ScBeH₆ can be confirmed as a real superconductor — let alone integrated into any practical technology — it needs to pass through a rigorous experimental gauntlet. Here's what that roadmap would likely look like:
- High-pressure synthesis: Researchers would use a diamond anvil cell (DAC) to compress a mixture of scandium, beryllium, and a hydrogen source (such as ammonia borane or pure H₂) to pressures in the 100 GPa range. Laser heating would be applied to drive the chemical reaction and form the target compound.
- Structural characterization: Synchrotron X-ray diffraction would be used to verify that the synthesized material actually adopts the predicted cubic structure. This is a non-trivial step — hydride syntheses often produce mixtures of phases, and isolating the target phase requires careful tuning.
- Electrical resistance measurements: The hallmark experimental signature of superconductivity is a sudden drop of electrical resistance to zero at Tc. Four-probe resistivity measurements inside the diamond anvil cell, across a range of temperatures, would provide the first definitive evidence.
- Magnetic susceptibility (Meissner effect): A true superconductor expels magnetic fields below its Tc. Measuring this effect confirms that the resistance drop isn't simply due to a metal-insulator transition or measurement artifact.
- Isotope effect experiments: Substituting hydrogen with deuterium should shift Tc in a predictable way for BCS-type superconductors. This test would confirm the electron-phonon coupling mechanism.
- Pressure-dependent studies: Mapping how Tc changes with pressure would reveal the phase diagram and help identify the optimal conditions for superconductivity.
Realistically, this validation process would take several years and require coordination between multiple specialized labs — those with high-pressure expertise, cryogenic measurement capability, and advanced spectroscopy. Groups at institutions like the Carnegie Institution, the Max Planck Institute, and various national labs have the infrastructure to pursue this kind of work.
Implications for the Field
If ScBeH₆ is experimentally confirmed, the implications extend well beyond a single new material. The broader significance lies in what it represents: a validation of rational, design-based approaches to discovering superconductors. For most of superconductivity's history, new materials were discovered serendipitously or through broad compositional searches. ScBeH₆, by contrast, was identified through deliberate computational screening — a search for the right combination of light elements, electronic structure, and stability.
This shift matters because room-temperature superconductivity — the holy grail — will almost certainly be found through exactly this kind of targeted computational discovery. Every confirmed prediction adds credibility to the methodology and narrows the search space for the next material. ScBeH₆ could serve as a stepping stone, demonstrating that ternary hydrides with carefully chosen metal partners are a fertile hunting ground.
The practical implications are equally significant. A superconductor operable at liquid nitrogen temperatures opens doors to:
- Lossless power transmission: Electrical grids lose roughly 5–10% of generated power to resistance. Superconducting cables could virtually eliminate this waste.
- More efficient MRI and medical imaging: Replacing expensive liquid helium systems with liquid nitrogen cooling would dramatically reduce the operating costs of medical imaging equipment worldwide.
- Magnetic levitation trans

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)