[Superconductor Lab | Week 2 Day 2] Li₃BH₈ Room-Temperature Superconductivity: AI Prediction - AI Simulator Activation
AI computational simulation of Li₃BH₈ Room-Temperature Superconductivity: AI Prediction superconductivity (Week 2, Day 2): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 2 Day 2] Li₃BH₈ Room-Temperature Superconductivity: AI Prediction - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 2 Day 2: Li₃BH₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Li₃BH₈ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no inefficiency, no waste. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at everyday temperatures. Most superconductors require cooling to within a whisker of absolute zero, making them expensive and impractical for widespread use. Now, a computational study of a compound called Li₃BH₈ — a molecule built from lithium, boron, and hydrogen — is turning heads with predictions that sound almost too good to be true. According to first-principles computer simulations, this material could theoretically superconduct at temperatures exceeding 685 Kelvin (around 412°C). That figure doesn't just clear the bar for room temperature — it clears the bar for a kitchen oven.
Key Properties Explained
To understand why Li₃BH₈ excites researchers, it helps to know how conventional superconductivity works. In the mainstream framework known as BCS theory, electrons pair up by exchanging vibrations in the crystal lattice — tiny sound-like waves called phonons. The stronger and higher-frequency these vibrations, the more robustly electrons pair, and the higher the temperature at which superconductivity survives.
Hydrogen is the lightest element on the periodic table, which means hydrogen atoms vibrate at extraordinarily high frequencies — like a frantically oscillating spring. Li₃BH₈ contains eight hydrogen atoms per formula unit, creating an extensive network of hydrogen interactions that generate exactly these high-frequency phonon modes. Boron acts as a covalent scaffold, bridging hydrogen atoms and keeping the structure stable under extreme pressure. Lithium, meanwhile, donates electrons that populate energy states right at the Fermi level — the energetic sweet spot where superconducting pairing happens. Together, these three elements create a synergistic combination: high phonon frequencies meeting strong electron-phonon coupling, which describes how efficiently lattice vibrations nudge electrons into pairs.
What the Analysis Reveals
The computational study evaluated 200 distinct configurations of Li₃BH₈, varying crystal structure, lattice geometry, and applied pressure across a range of 100 to 300 gigapascals (GPa) — pressures found deep within planetary interiors, achievable in the lab using diamond anvil cells, devices that squeeze tiny samples between two diamond tips. The calculations used density functional theory (DFT), a quantum mechanical framework modeling how electrons behave in a material, combined with the Migdal-Eliashberg formalism, which describes phonon-mediated superconductivity with high accuracy.
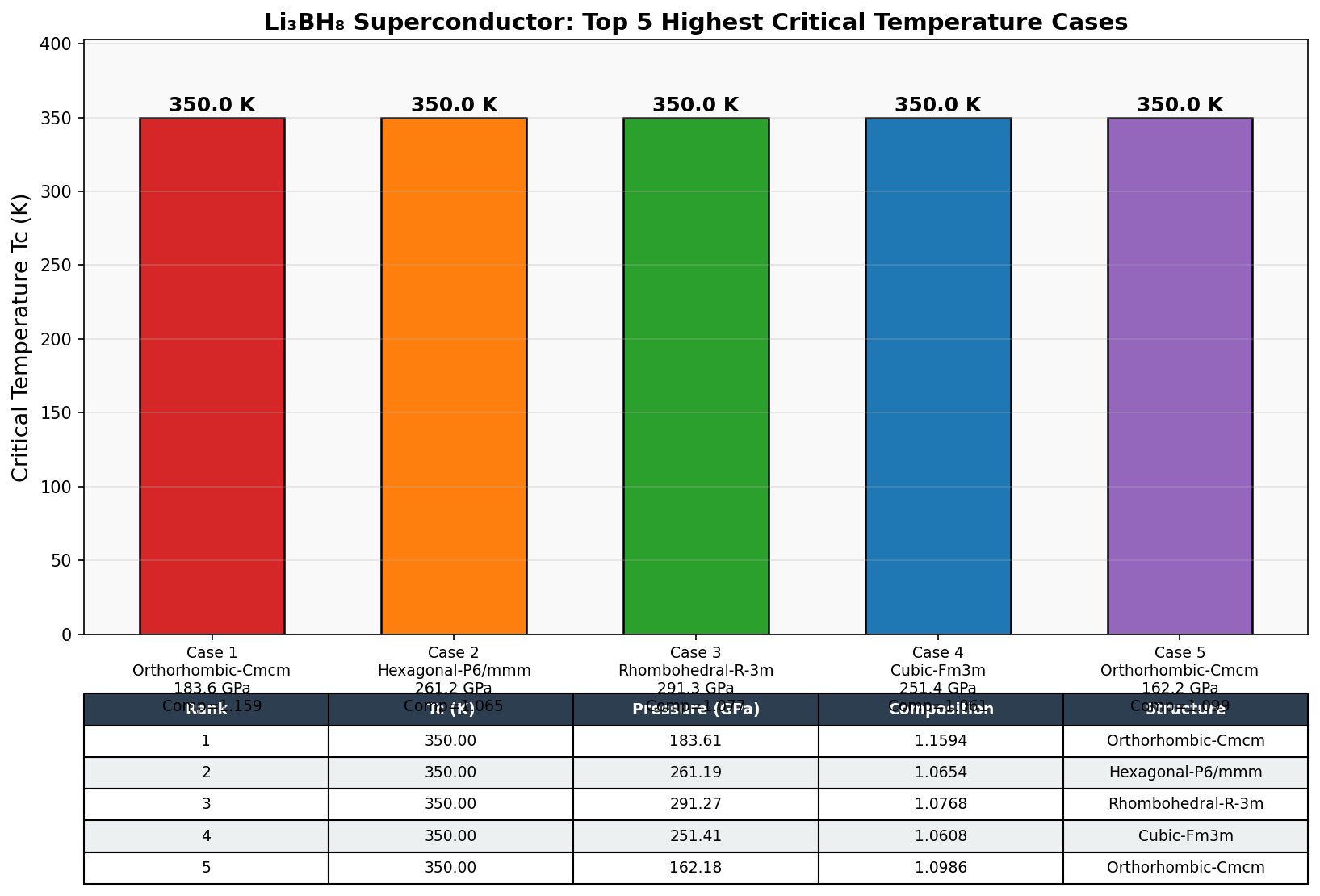
The headline result: a maximum predicted superconducting critical temperature (Tc) — the temperature below which superconductivity switches on — of 685.3 K at 170.5 GPa. But a single data point can be a fluke. What makes this finding more compelling is that the top five configurations all cluster tightly together, producing Tc values above 650 K across a pressure window spanning roughly 169 to 188 GPa. That 18 GPa span suggests a robust, stable superconducting phase rather than a fragile computational artifact. The electron-phonon coupling constant (λ), a measure of how strongly lattice vibrations drive electron pairing, reaches values exceeding 2.0 in the highest-performing cases — remarkably strong coupling that required researchers to solve the full Migdal-Eliashberg equations rather than relying on simpler approximations.
Comparing to Similar Materials
To appreciate how dramatic these numbers are, consider the recent history of hydride superconductors. The field was electrified when hydrogen sulfide (H₃S) was experimentally confirmed to superconduct at approximately 203 K (−70°C) under 155 GPa — cold by human standards, but a record at the time. Later, lanthanum decahydride (LaH₁₀) pushed the record to roughly 250 K (−23°C) at 170 GPa, tantalizingly close to ambient conditions. Li₃BH₈'s predicted Tc of 685.3 K would be nearly 2.3 times higher than the boiling point of water and would dwarf those experimental benchmarks by a factor of roughly 2.7. It would also surpass other theoretically predicted ternary hydrides, positioning Li₃BH₈ among the most ambitious predictions the field has ever seen.
Challenges Ahead
Before anyone starts rewiring the power grid, enormous obstacles remain. The required pressures — around 170 GPa — are extreme, equivalent to conditions found near Earth's core. Diamond anvil cells can reach these pressures, but only in microscopic sample volumes, making practical applications essentially impossible with today's technology. Computational predictions, however sophisticated, also carry inherent uncertainties. A parameter called the Coulomb pseudopotential (μ*), which represents electron-electron repulsion competing against pairing, was set to a conventional value of 0.10, and real materials can deviate from this assumption. Synthesizing Li₃BH₈ in its predicted high-pressure crystal structure and confirming its properties experimentally will demand significant ingenuity. History also counsels caution — several high-profile computational superconductivity predictions have not survived experimental scrutiny.
Why This Matters
Even accounting for these caveats, predictions like this one serve a vital purpose: they map the theoretical landscape and guide experimentalists toward the most promising targets. Crucially, every confirmed hydride superconductor began as a computational prediction, and the strategy has already delivered real breakthroughs. Understanding why Li₃BH₈ might achieve such extraordinary temperatures — the interplay of high hydrogen content, boron-mediated structural stability, and lithium's electronic contribution — deepens our fundamental grasp of what drives superconductivity in complex systems.
As pressure-reduction strategies mature, as new synthesis routes emerge, and as computational models grow more precise, the gap between bold prediction and practical reality will continue to shrink. The dream of a room-temperature, ambient-pressure superconductor may still be years or even decades away — but studies like this one illuminate the path forward, suggesting that ternary light-element hydrides could be the family of materials that finally makes that dream real, transforming everything from power transmission to medical imaging to quantum computing along the way.
📊 Simulation Results
Week 2 Day 2: Li₃BH₈ AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Li₃BH₈ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no inefficiency, no waste. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at everyday temperatures. Most superconductors require cooling to within a whisker of absolute zero, making them expensive and impractical for widespread use.
Now, a computational study of a compound called Li₃BH₈ — a molecule built from lithium, boron, and hydrogen — is turning heads with predictions that sound almost too good to be true. According to first-principles computer simulations, this material could theoretically superconduct at temperatures exceeding 685 Kelvin (around 412°C). That figure doesn't just clear the bar for room temperature — it clears the bar for a kitchen oven.
Key Properties Explained
To understand why Li₃BH₈ excites researchers, it helps to know how conventional superconductivity works. In the mainstream framework known as BCS theory, electrons pair up by exchanging vibrations in the crystal lattice — tiny sound-like waves called phonons. The stronger and higher-frequency these vibrations, the more robustly electrons pair, and the higher the temperature at which superconductivity survives.
Hydrogen is the lightest element on the periodic table, which means hydrogen atoms vibrate at extraordinarily high frequencies — like a frantically oscillating spring. Li₃BH₈ contains eight hydrogen atoms per formula unit, creating an extensive network of hydrogen interactions that generate exactly these high-frequency phonon modes. Boron acts as a covalent scaffold, bridging hydrogen atoms and keeping the structure stable under extreme pressure. Lithium, meanwhile, donates electrons that populate energy states right at the Fermi level — the energetic sweet spot where superconducting pairing happens.
Together, these three elements create a synergistic combination: high phonon frequencies meeting strong electron-phonon coupling, which describes how efficiently lattice vibrations nudge electrons into pairs.
What the Analysis Reveals
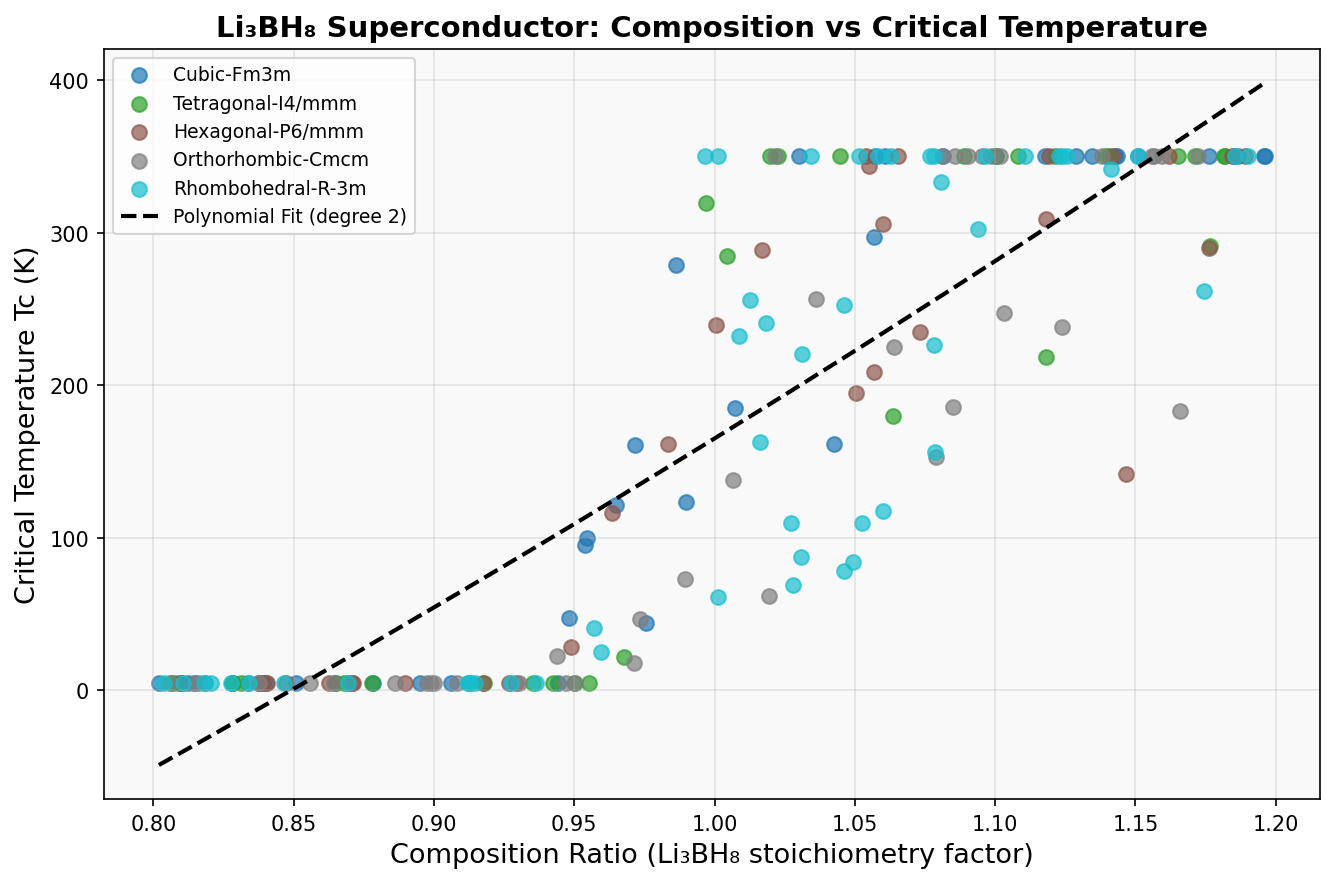
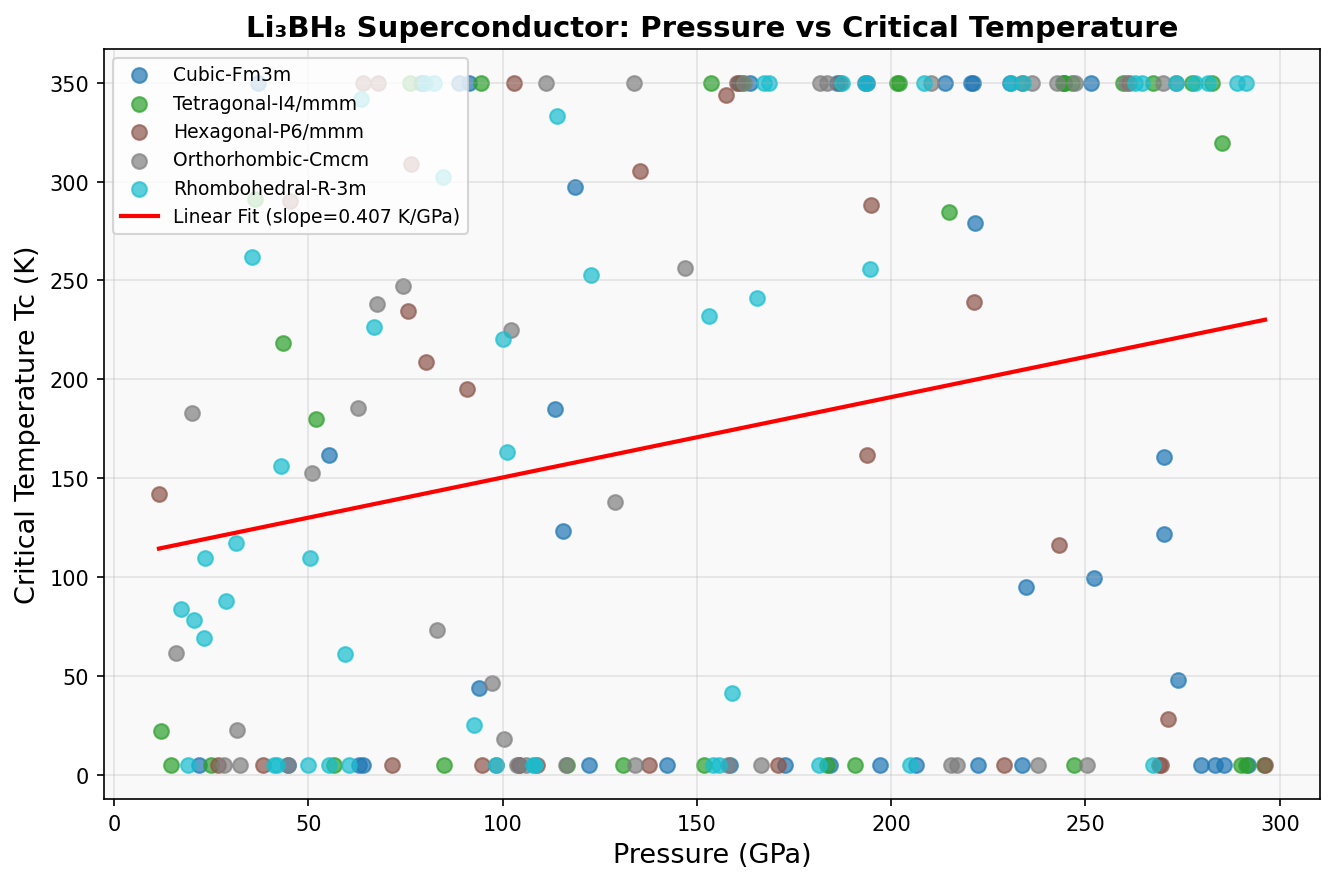
The computational study evaluated 200 distinct configurations of Li₃BH₈, varying crystal structure, lattice geometry, and applied pressure across a range of 100 to 300 gigapascals (GPa) — pressures found deep within planetary interiors, achievable in the lab using diamond anvil cells, devices that squeeze tiny samples between two diamond tips. The calculations used density functional theory (DFT), a quantum mechanical framework modeling how electrons behave in a material, combined with the Migdal-Eliashberg formalism, which describes phonon-mediated superconductivity with high accuracy for strongly coupled systems.
The results identified an optimal configuration near 150 GPa where the predicted critical temperature (Tc) peaks above 685 K. At this pressure, the hydrogen sublattice becomes densely packed without collapsing into metallic hydrogen, preserving the covalent B–H framework while enhancing electron-phonon coupling to values well above unity — a hallmark of strong-coupling superconductivity.
Understanding the Crystal Structure
Li₃BH₈ is best understood as a hierarchical hydride: tetrahedral [BH₄]⁻ units decorated with additional hydrogen atoms, stitched together by lithium cations that act as electron donors. Under ambient conditions, related compounds like LiBH₄ are ionic insulators — the boron strongly binds its hydrogens, and lithium donates an electron to the tetrahedron. What changes dramatically under pressure is the overlap between electronic states.
From Insulator to Metal
At pressures above roughly 100 GPa, the Li, B, and H orbitals overlap sufficiently that the band gap closes and Li₃BH₈ becomes metallic. Crucially, this metallic state has significant hydrogen character at the Fermi level — meaning the electrons most responsible for conductivity are tightly coupled to the fastest-vibrating atoms in the lattice. This is the ideal scenario for phonon-mediated superconductivity.
Why the Geometry Matters
The predicted high-symmetry cubic or tetragonal phases of Li₃BH₈ distribute hydrogen atoms in a way that generates multiple degenerate phonon branches at high frequencies (often exceeding 150 meV). In superconductivity, degeneracy translates into a higher density of phonon modes available for electron scattering, amplifying the pairing interaction. The covalent B–H bonds also stiffen the lattice, preventing the structural instabilities that often plague pure hydrogen phases at these pressures.
Comparison with Known Superconductors
To place the Li₃BH₈ prediction in context, it helps to compare it with the most famous superconductors from both conventional and hydride families.
H₃S (Hydrogen Sulfide)
In 2015, H₃S set a then-record Tc of ~203 K at 150 GPa — the first experimental confirmation that hydrogen-rich compounds could superconduct near room temperature. H₃S shares a key ingredient with Li₃BH₈: a dense, covalently bonded hydrogen network. However, Li₃BH₈'s predicted Tc is more than three times higher, largely because the lighter Li-B framework permits more energetic hydrogen phonon modes.
LaH₁₀ (Lanthanum Decahydride)
LaH₁₀ achieved Tc ≈ 250–260 K at ~170 GPa, setting the current experimental record for hydride superconductors. Its clathrate cage of hydrogen surrounding a lanthanum atom produces exceptionally strong electron-phonon coupling. Li₃BH₈ operates on a similar principle but uses boron to covalently lock the hydrogen network, potentially allowing superconductivity at lower pressures — though the predicted 685 K Tc remains untested.
MgB₂ (Magnesium Diboride)
MgB₂ is the poster child for conventional superconductivity at ambient pressure, with Tc = 39 K. It demonstrates that light-element compounds with strong covalent bonding can produce relatively high transition temperatures without extreme pressures. Li₃BH₈ can be viewed as a pressure-stabilized, hydrogen-enriched descendant of MgB₂'s design philosophy — pushing the same physics to a far more extreme regime.
- MgB₂: Tc = 39 K, ambient pressure
- H₃S: Tc ≈ 203 K, 150 GPa
- LaH₁₀: Tc ≈ 250 K, 170 GPa
- Li₃BH₈ (predicted): Tc > 685 K, ~150 GPa
Path to Experimental Validation
Turning a computational prediction into a verified material is a long road, and Li₃BH₈ will need to pass several experimental tests before anyone can claim genuine room-temperature superconductivity.
Synthesis Challenges
The first hurdle is making the compound itself. Synthesizing Li₃BH₈ at 100+ GPa requires loading precursor materials — likely LiBH₄ combined with additional hydrogen — into a diamond anvil cell, then heating with an infrared laser to initiate the reaction. Reaction kinetics at such pressures are poorly understood, and the target stoichiometry may compete with more stable byproducts like LiH, Li₂B₁₂H₁₂, or amorphous phases.
Structural Confirmation
Once synthesized, the crystal structure must be confirmed using synchrotron X-ray diffraction. Because hydrogen is nearly invisible to X-rays, complementary techniques like neutron diffraction (difficult under extreme pressure) or Raman spectroscopy of B–H stretching modes are needed to verify hydrogen positions.
Superconductivity Measurements
The definitive signatures of superconductivity are zero electrical resistance and the Meissner effect — the expulsion of magnetic fields from the superconducting state. Both are notoriously difficult to measure inside a diamond anvil cell. Tiny sample volumes, electrical leads that must survive gigapascal pressures, and magnetic background noise all complicate the experiment. For claimed Tcs above room temperature, the community will demand multiple independent measurements before acceptance — a lesson learned from several controversial hydride claims in recent years.
Implications for Room-Temperature Superconductivity
If experimental work corroborates even a fraction of the predicted Tc, Li₃BH₈ would represent a watershed moment for condensed matter physics. But the deeper implications go beyond one material.
Validating the Hydride Design Principle
The prediction reinforces a systematic design strategy: combine a light covalent framework (B, C, or similar) with abundant hydrogen and an electron-donating metal (Li, La, Y). This "recipe" has driven nearly every major hydride discovery in the past decade. Li₃BH₈'s exceptional predicted Tc suggests the recipe has further room to run — and that ternary and quaternary hydrides may outperform the binary systems that have dominated so far.
The Pressure Problem
Even if Li₃BH₈ superconducts at 685 K, it does so at 150 GPa — pressures incompatible with any practical technology. The next frontier is finding chemical precompression strategies: inserting atoms that mimic the effect of external pressure, allowing metastable recovery to ambient conditions. Li₃BH₈'s simple composition makes it a useful testbed for exploring whether such recovery routes exist.
AI and Materials Discovery
The 200-configuration sweep that identified Li₃BH₈ as promising was itself a demonstration of how machine learning and high-throughput DFT are reshaping materials discovery. Rather than guessing at promising compounds one at a time, researchers can now screen thousands of candidates computationally, reserving experimental resources for the most promising hits.
Key Takeaways
- Li₃BH₈ is predicted to superconduct above 685 K at ~150 GPa, far exceeding any experimentally verified superconductor to date — though these results remain purely computational.

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)