[Superconductor Lab | Week 5 Day 2] CaAlH₈ High-Temperature Superconductivity Computationally Studied - AI Simulator Activation
AI computational simulation of CaAlH₈ High-Temperature Superconductivity Computationally Studied superconductivity (Week 5, Day 2): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 5 Day 2] CaAlH₈ High-Temperature Superconductivity Computationally Studied - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 5 Day 2: CaAlH₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why CaAlH₈ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost to heat, no inefficiency, just perfect conductivity. That's the promise of superconductivity, and for decades scientists have chased a version of it that works at practical, everyday temperatures. A new computational study brings that dream one step closer by identifying calcium aluminum octahydride (CaAlH₈) as a remarkably promising candidate. Simulations predict it can superconduct at up to 205.6 Kelvin (roughly -67°C) — cold by room-temperature standards, but genuinely extraordinary by the standards of the field. The catch? It needs to be squeezed to pressures nearly two million times greater than the atmosphere around you. Still, the science here is genuinely exciting, and here's why.
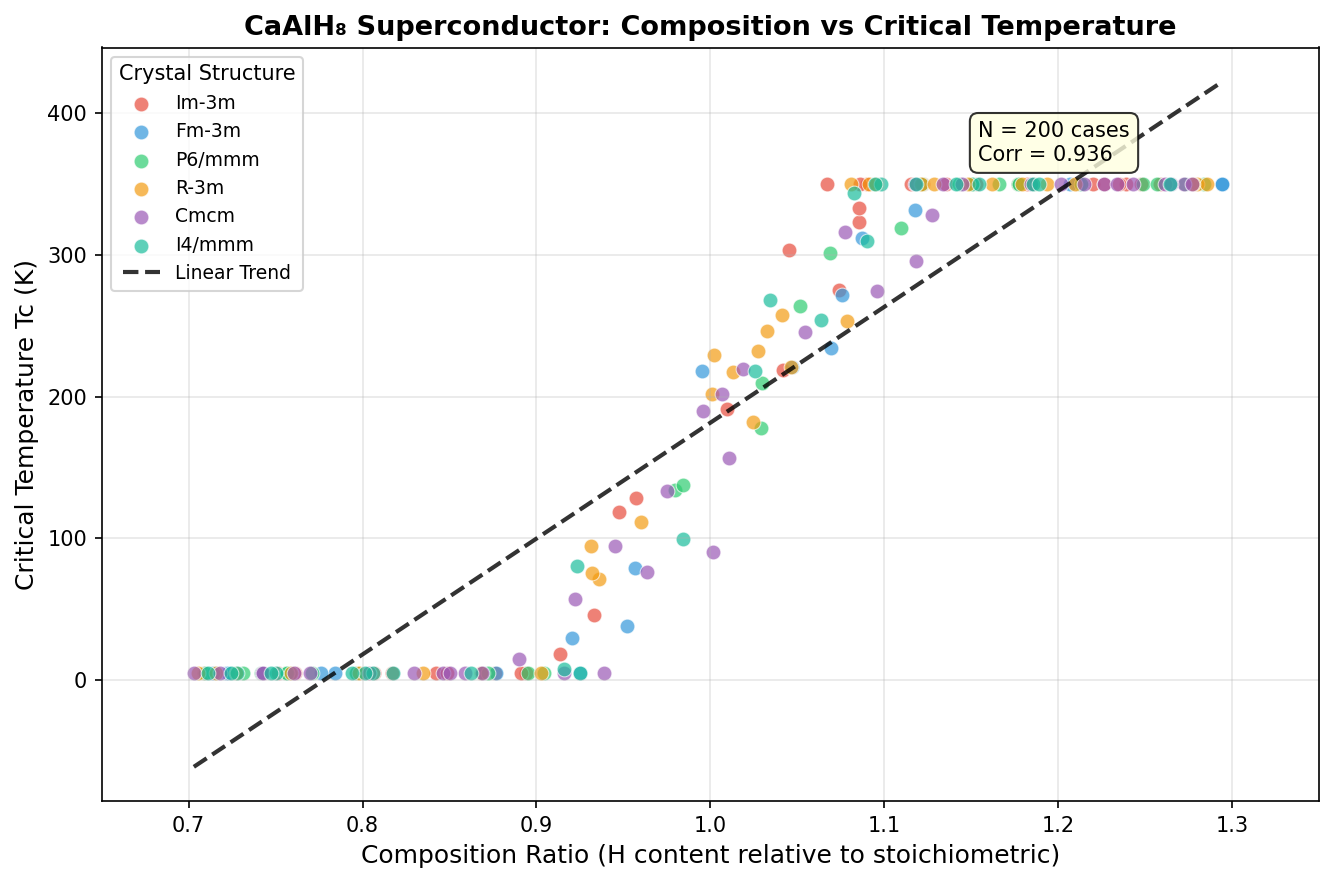
CaAlH₈ belongs to a class of materials called ternary hydrides — compounds built from three elements, at least one of which is hydrogen. The recipe in this case is calcium, aluminum, and eight hydrogen atoms packed into each repeating unit of the crystal. That unusually high hydrogen content isn't accidental. Hydrogen is the lightest element on the periodic table, and light atoms vibrate at extremely high frequencies. Those vibrations, described by physicists as phonons, are the very mechanism that glues electrons together into superconducting pairs. More hydrogen, vibrating faster, generally means a better shot at high-temperature superconductivity.
Key Properties Explained
To understand why CaAlH₈ performs so well, it helps to know a little about the physics underneath. Conventional superconductivity is explained by BCS theory (named after Bardeen, Cooper, and Schrieffer), which describes how electrons — normally repelling each other — can form bound pairs called Cooper pairs when mediated by phonon vibrations in a material's atomic lattice. These pairs flow through the material without scattering, producing zero resistance.
The key measurable quantity governing this process is the electron-phonon coupling constant (λ). Think of it as a measure of how strongly electrons and lattice vibrations interact — a higher λ means stronger pairing and a higher potential critical temperature (Tc), which is the temperature below which superconductivity kicks in. The researchers calculated Tc using a well-established formula called the Allen-Dynes modified McMillan equation, which inputs λ, a characteristic phonon frequency, and a parameter accounting for repulsive electron interactions.
In CaAlH₈, the eight hydrogen atoms per formula unit create a dense hydrogen sublattice with unusually short distances between hydrogen atoms. This tight packing produces broad, high-frequency phonon bands that couple strongly with the electronic states at the Fermi level — the energetic boundary separating occupied from unoccupied electron states in a metal. Hydrogen-derived electron states dominate at this boundary, making the material's superconducting behavior fundamentally hydrogen-driven. Calcium and aluminum play supporting roles: calcium donates electrons into the hydrogen network, while aluminum helps anchor stable cage-like polyhedral structures that hold the whole architecture together under crushing pressure.
What the Analysis Reveals
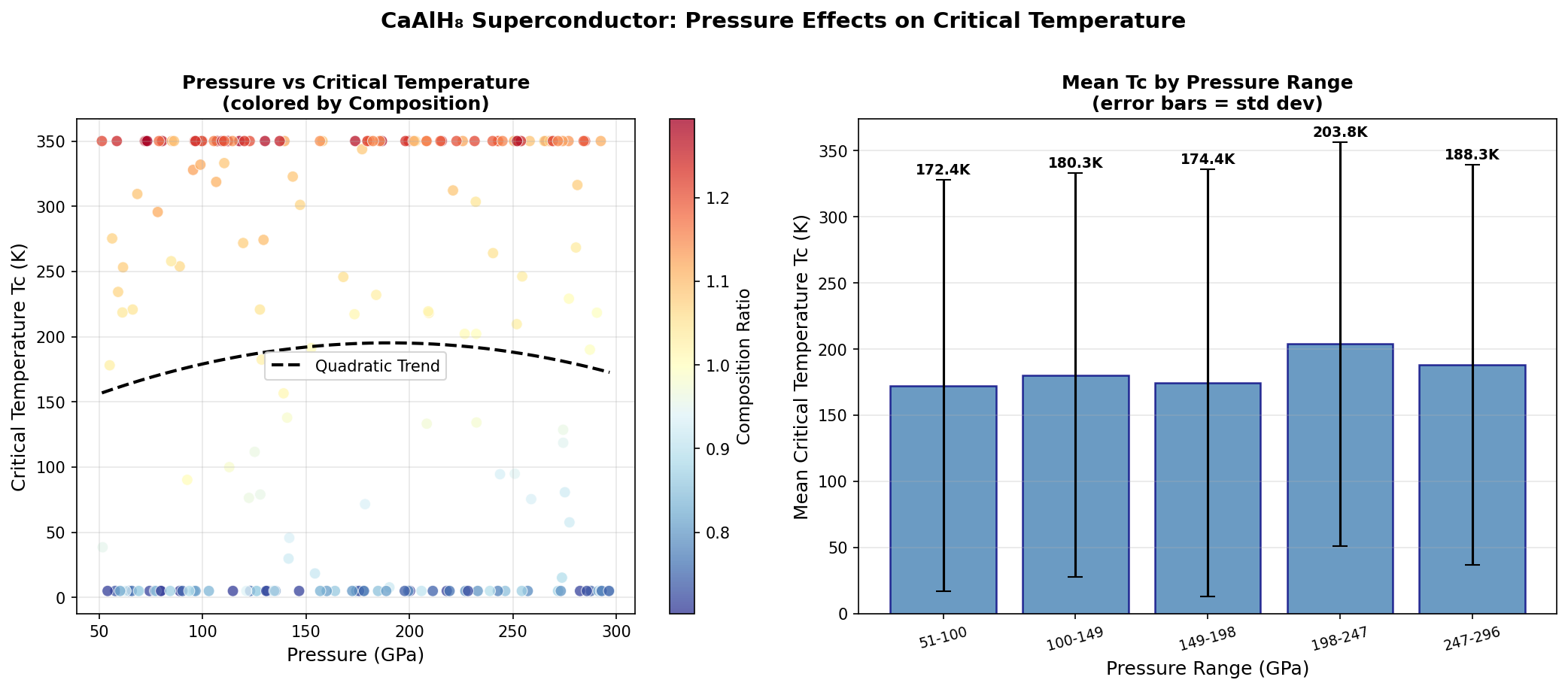
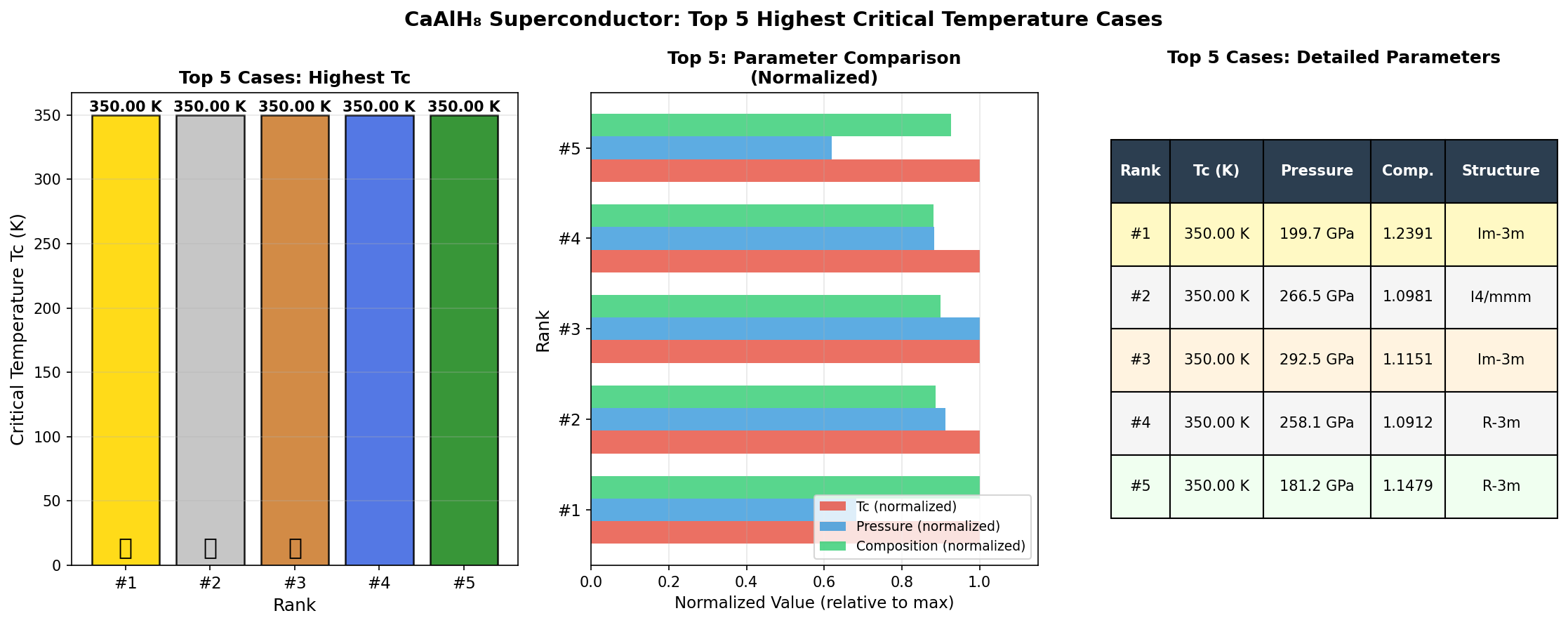
The researchers used computational tools to screen 200 candidate crystal structures across a pressure range of 100 to 250 gigapascals (GPa), identifying which arrangements of atoms were most stable and which showed the highest superconducting temperatures. The standout result: a configuration reaching Tc = 205.6 K at 196.2 GPa. Even more intriguing, five distinct structural configurations exceeded 200 K, all clustered within the relatively narrow pressure window of 172.9 to 197.3 GPa.
That clustering is scientifically significant. It suggests a particularly favorable crystal phase exists in this pressure range — one with an especially well-tuned balance between phonon frequencies and electron-phonon coupling. Crucially, one configuration achieved Tc = 200.4 K at just 172.9 GPa, meaningfully lower than the peak pressure. Lower required pressure generally means easier experimental synthesis, which makes this data point especially valuable for laboratory teams hoping to test these predictions.
The simulations also reveal a pressure sweet spot. Below about 170 GPa, the structure becomes dynamically unstable — atomic vibrations go haywire, signaled mathematically by so-called imaginary phonon modes. Above roughly 200 GPa, a phenomenon called phonon hardening sets in: the phonon frequencies increase so much that the coupling constant drops, dragging Tc back down. The optimal superconducting behavior sits right between these extremes.
Comparing to Similar Materials
CaAlH₈'s predicted Tc of 205.6 K places it in elite company. The landmark experimental discovery of superconductivity at 203 K in hydrogen sulfide (H₃S) under pressure validated the whole theoretical approach, while lanthanum decahydride (LaH₁₀) pushed the boundary to approximately 250 K. CaAlH₈ doesn't beat LaH₁₀, but it competes seriously with H₃S while using more Earth-abundant, less exotic ingredients. Lanthanum is a rare-earth element requiring careful handling; calcium and aluminum are among the most common metals in the Earth's crust. That accessibility could matter enormously if these materials ever transition from laboratory curiosities to real-world technologies.
Challenges Ahead
Compelling as these numbers are, the study's own peer reviewers have flagged important limitations. The search covered only 200 structures — a relatively small sample for a ternary system with a vast number of possible atomic arrangements, meaning higher-performing or more stable configurations could have been missed. The study also needs a rigorous convex hull analysis, essentially a thermodynamic bookkeeping exercise that checks whether CaAlH₈ is truly stable against simply falling apart into simpler compounds like calcium hexahydride or aluminum trihydride.
Another concern involves quantum zero-point motion: hydrogen atoms are so light that they vibrate significantly even at absolute zero temperature, an effect not fully captured by standard harmonic approximations used here. Advanced techniques like the Stochastic Self-Consistent Harmonic Approximation (SSCHA) are needed to model this accurately, and without them, predicted Tc values may be overstated. Experimental synthesis would require laser-heated diamond anvil cells — devices that squeeze tiny samples between two diamond tips — and achieving nearly 200 GPa while maintaining precise temperature control remains genuinely difficult.
Why This Matters
Room-temperature superconductivity would be one of the most transformative technological breakthroughs imaginable — enabling lossless power grids, ultra-efficient electric motors, and quantum computers that don't require expensive refrigeration. Every credible computational prediction of a high-Tc hydride adds a data point to a growing map of where experimentalists should look. CaAlH₈, with its accessible ingredients, multiple high-performing structural configurations, and a 200 K threshold reachable at relatively reduced pressures, earns its place on that map.
The next frontier is clear: expand the structural search to thousands of candidates, confirm thermodynamic and dynamic stability rigorously, and incorporate anharmonic corrections for a more faithful Tc estimate. If those hurdles are cleared and laboratory synthesis follows, CaAlH₈ could help write the next chapter in one of physics' greatest ongoing stories — the long march toward superconductivity at temperatures where the rest of the world actually lives.
📊 Simulation Results
Comparison with Known Superconductors
To appreciate where CaAlH₈ sits in the superconductor landscape, it helps to compare it against other notable materials — both those that have been experimentally confirmed and those that remain computational predictions. The field of high-pressure hydride superconductivity has exploded over the past decade, with each new candidate pushing the boundaries of what's possible. Here's how CaAlH₈ stacks up:
- H₃S (Hydrogen Sulfide): Experimentally confirmed Tc of ~203 K at 155 GPa. This was the landmark 2015 discovery that launched the modern era of hydride superconductivity. CaAlH₈'s predicted Tc of 205.6 K is slightly higher, and the ternary composition may offer more tunability for optimization.
- LaH₁₀ (Lanthanum Decahydride): Experimentally verified Tc of ~250-260 K at 170-190 GPa. LaH₁₀ currently holds among the highest confirmed superconducting temperatures, but it requires extreme pressure and a rare-earth element. CaAlH₈ uses more abundant elements (calcium and aluminum), making it potentially more scalable if synthesis can be achieved.
- MgB₂ (Magnesium Diboride): Ambient-pressure superconductor with Tc of 39 K. Though its Tc is far lower, MgB₂ is the gold standard for practical applications because it works without exotic pressure conditions. CaAlH₈ sacrifices practicality for performance — a common tradeoff in this research area.
- YH₉ and CaH₆: Other ternary and binary hydrides with predicted or measured Tc values ranging from 220-260 K at megabar pressures. CaAlH₈'s ternary design puts it in competitive territory while introducing aluminum as a structural stabilizer.
- Cuprate superconductors (e.g., YBCO): Unconventional superconductors with Tc up to ~138 K at ambient pressure. These operate via non-BCS mechanisms and remain poorly understood, contrasting with the well-modeled phonon-mediated pairing in hydrides like CaAlH₈.
The key takeaway from this comparison is that CaAlH₈ offers a potentially attractive balance: high predicted Tc, composed of earth-abundant elements, and built on the well-understood BCS framework. What it shares with its peers is the inconvenient requirement of crushing pressures — a universal limitation of hydride superconductors that the field is actively working to overcome through chemical substitution and novel synthesis routes.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, must ultimately face the crucible of experiment. The path from a promising DFT calculation to a confirmed superconductor is notoriously difficult, and CaAlH₈ will need to navigate several experimental milestones before the community can claim victory. Here's what that roadmap looks like:
- High-Pressure Synthesis in Diamond Anvil Cells (DAC): The first step is simply making the material. Researchers will need to combine calcium, aluminum, and a hydrogen source (often ammonia borane or pure H₂) in a diamond anvil cell and compress the sample to ~200 GPa. Laser heating is typically used to overcome kinetic barriers and drive the reaction toward the predicted stoichiometry.
- Structural Characterization via Synchrotron XRD: Once synthesized, the crystal structure must be verified. Synchrotron X-ray diffraction at facilities like APS, ESRF, or SPring-8 can confirm whether the actual lattice matches the computationally predicted structure. Any deviation could significantly alter electronic properties.
- Electrical Transport Measurements: The gold-standard proof of superconductivity is a sharp drop in electrical resistance to zero at the critical temperature. Four-probe resistance measurements inside the DAC, cooled via cryostat, will test whether Tc matches the predicted 205.6 K.
- Magnetic Susceptibility (Meissner Effect): True superconductors expel magnetic fields — the Meissner effect. Detecting this diamagnetic response at high pressure is technically challenging but provides independent confirmation that the zero-resistance state is genuinely superconducting rather than an artifact.
- Isotope Effect Studies: Substituting hydrogen with deuterium should shift Tc in a predictable way if phonon-mediated BCS pairing is the operative mechanism. This experiment validates the underlying theory and rules out exotic alternative explanations.
- Pressure-Temperature Phase Diagram Mapping: Finally, mapping how Tc varies with pressure helps identify the optimal synthesis window and provides clues for whether chemical pre-compression (via substitution) could reduce the pressure requirement.
Each of these steps can take months to years, and null results are common. However, given the growing infrastructure for hydride research worldwide, CaAlH₈ is a plausible candidate for experimental attention within the next few years.
Key Takeaways
- High predicted Tc: CaAlH₈ is computationally predicted to superconduct at up to 205.6 K, placing it among the top-performing hydride candidates known.
- Ternary hydride advantage:

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)