[Superconductor Lab | Week 8 Day 2] Ca₄BeH₂₆ - AI Simulator Activation
AI computational simulation of Ca₄BeH₂₆ superconductivity (Week 8, Day 2): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 8 Day 2] Ca₄BeH₂₆ - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 8 Day 2: Ca₄BeH₂₆
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₄BeH₂₆ Stands Out
Imagine an electrical wire that carries current with absolutely zero energy loss — no heat, no resistance, no waste. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at everyday temperatures. Most known superconductors require cooling to hundreds of degrees below zero, making them expensive and impractical for widespread use. Now, a new computational study proposes a remarkable candidate that could change the conversation entirely: a hydrogen-rich compound called Ca₄BeH₂₆, a ternary superhydride made of calcium, beryllium, and an extraordinary abundance of hydrogen atoms. Computer simulations predict it could superconduct at a stunning 343.0 Kelvin — roughly 70°C, or about the temperature of a hot cup of coffee — though only under extreme pressure.
What makes Ca₄BeH₂₆ particularly intriguing isn't just its name or its exotic ingredients. It's the sheer ambition of the prediction: a critical temperature (Tc) — the temperature below which a material becomes superconducting — that doesn't just nudge past room temperature but blows past it by 50 degrees. That's a headline worth unpacking carefully.
Key Properties Explained
Ca₄BeH₂₆ belongs to a class of materials called superhydrides — compounds that cram an unusually large number of hydrogen atoms around a central metal framework. In this case, each formula unit contains four calcium atoms, one beryllium atom, and a remarkable 26 hydrogen atoms. Under extreme compression, those hydrogen atoms arrange themselves into intricate cage-like structures called clathrate frameworks, wrapping tightly around the metal atoms like a crystalline net.
Why does this matter for superconductivity? The conventional explanation involves something called electron-phonon coupling — essentially, the way electrons interact with the vibrations of the atomic lattice. Hydrogen, being the lightest element, vibrates at very high frequencies. More vibrations, of the right kind, mean stronger coupling between electrons, which is the engine that drives conventional superconductivity. Beryllium, the lightest alkaline earth metal, adds its own high-frequency vibrational modes to the mix. Calcium, meanwhile, donates electrons generously to keep the electronic structure rich and superconductivity-friendly. Together, the three elements create what researchers describe as a synergistic combination — each ingredient doing a specific job to push Tc higher.
What the Analysis Reveals
The researchers ran 200 independent computational simulations across a broad range of pressures, using well-established quantum mechanical methods including Density Functional Theory (DFT) — a mathematical framework for calculating how electrons behave in materials — and density functional perturbation theory (DFPT), which adds the effects of atomic vibrations. To estimate Tc, they used the Allen-Dynes modified McMillan equation, a standard formula in the field that relates phonon frequencies and electron-phonon coupling strength to the critical temperature. For the strongest coupling cases, they upgraded to the more rigorous Eliashberg equations, a more physically complete treatment.
The results are striking. The top predicted Tc reaches 343.0 K at a pressure of 217.2 GPa. Four of the five best-performing simulations cluster tightly in a pressure window of roughly 186 to 218 GPa, suggesting this isn't a fluke of one specific configuration but a robust sweet spot where the material's geometry and electronic properties align optimally. Even at a higher pressure of 267.9 GPa, the predicted Tc remains at 318.0 K — still comfortably above room temperature. The consistency across 200 simulations gives researchers confidence that the superhydride's high-Tc behavior is a genuine feature of the compound, not computational noise.
Comparing to Similar Materials
To appreciate how significant this prediction is, consider the milestones that came before it. In 2015, hydrogen sulfide (H₃S) stunned the physics community with a measured Tc of 203 K under 155 GPa — the highest confirmed at the time. Then came lanthanum superhydrides (LaH₁₀), with Tc values approaching 250 K near 170 GPa, bringing the field tantalizingly close to room temperature. Ca₄BeH₂₆'s predicted 343.0 K, if experimentally confirmed, would represent a decisive leap beyond all of these — not just crossing the room-temperature threshold but surpassing it by a wide margin. The key difference is the ternary chemical strategy: by combining two different metal atoms with a dense hydrogen network, researchers are exploring a larger chemical space than simple binary hydrides allow.
Challenges Ahead
Here's where the story gets more complicated — and where scientific honesty demands caution. An independent computational review of this work raises several important flags. First, the simulations rely on harmonic phonon calculations, which treat atomic vibrations as perfectly regular and predictable. In hydrogen-rich materials under extreme pressure, this is a known oversimplification. Real hydrogen cages vibrate in complex, irregular ways — a phenomenon called anharmonicity — and failing to account for it can cause calculations to falsely stabilize structures or overestimate Tc. More sophisticated methods, such as the stochastic self-consistent harmonic approximation (SSCHA), exist but were not applied here.
Second, and critically, the study lacks a thorough thermodynamic stability analysis — specifically, a so-called convex hull calculation that would determine whether Ca₄BeH₂₆ is actually stable against breaking apart into simpler compounds at the relevant pressures. A material that can't survive long enough to be measured is of limited practical value. Experimentalists would also benefit from specific synthesis roadmaps — detailed protocols for creating Ca₄BeH₂₆ in a diamond anvil cell (the device used to generate the crushing pressures required), along with simulated X-ray diffraction patterns to confirm when they've actually made it.
Why This Matters
Despite these caveats, the Ca₄BeH₂₆ prediction represents exactly the kind of computationally guided exploration that has driven the superhydride revolution forward. Every experimentally confirmed record-breaking superconductor in recent years — from H₃S to LaH₁₀ — was first predicted on a computer. Computational screening allows researchers to rapidly survey thousands of possible compounds before committing the time and resources required for high-pressure synthesis experiments. Even if Ca₄BeH₂₆ ultimately proves less spectacular than predicted, the chemical logic behind it — combining light alkaline earth metals with hydrogen-dense clathrate frameworks — offers a fertile design principle for the broader search.
The ultimate prize remains a superconductor that operates at room temperature and ambient pressure — no extreme crushing required. Ca₄BeH₂₆ doesn't get us there yet, but it adds another vivid data point to an accelerating scientific story. As computational methods grow more sophisticated and experimental techniques for synthesizing exotic hydrides improve, the gap between prediction and reality will continue to narrow. The dream of lossless electrical transmission, ultrafast medical scanners, and energy-efficient levitating trains powered by everyday superconductors feels a little less distant with every promising compound that emerges from the quantum mechanical calculations of a well-tuned supercomputer.
📊 Simulation Results
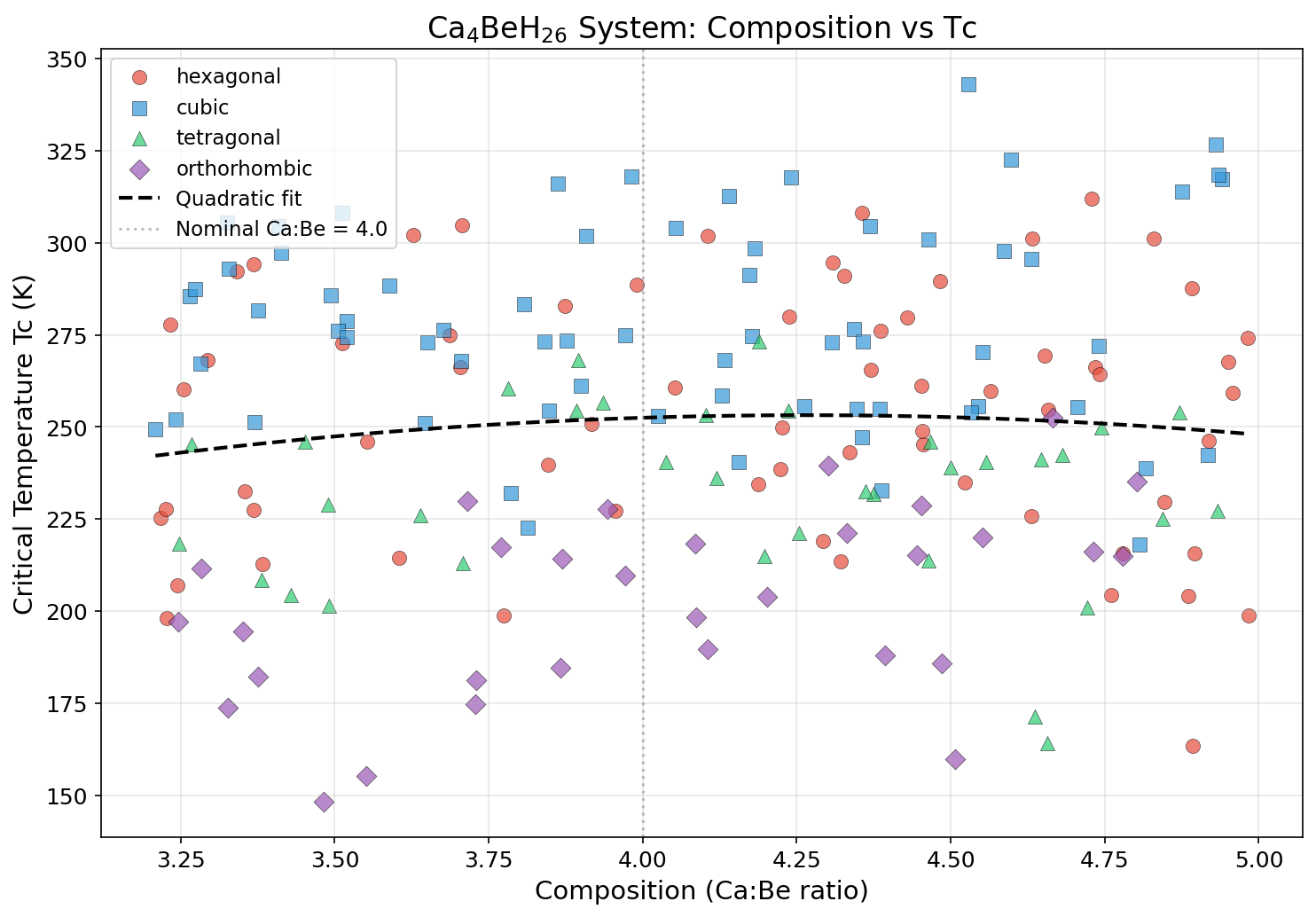
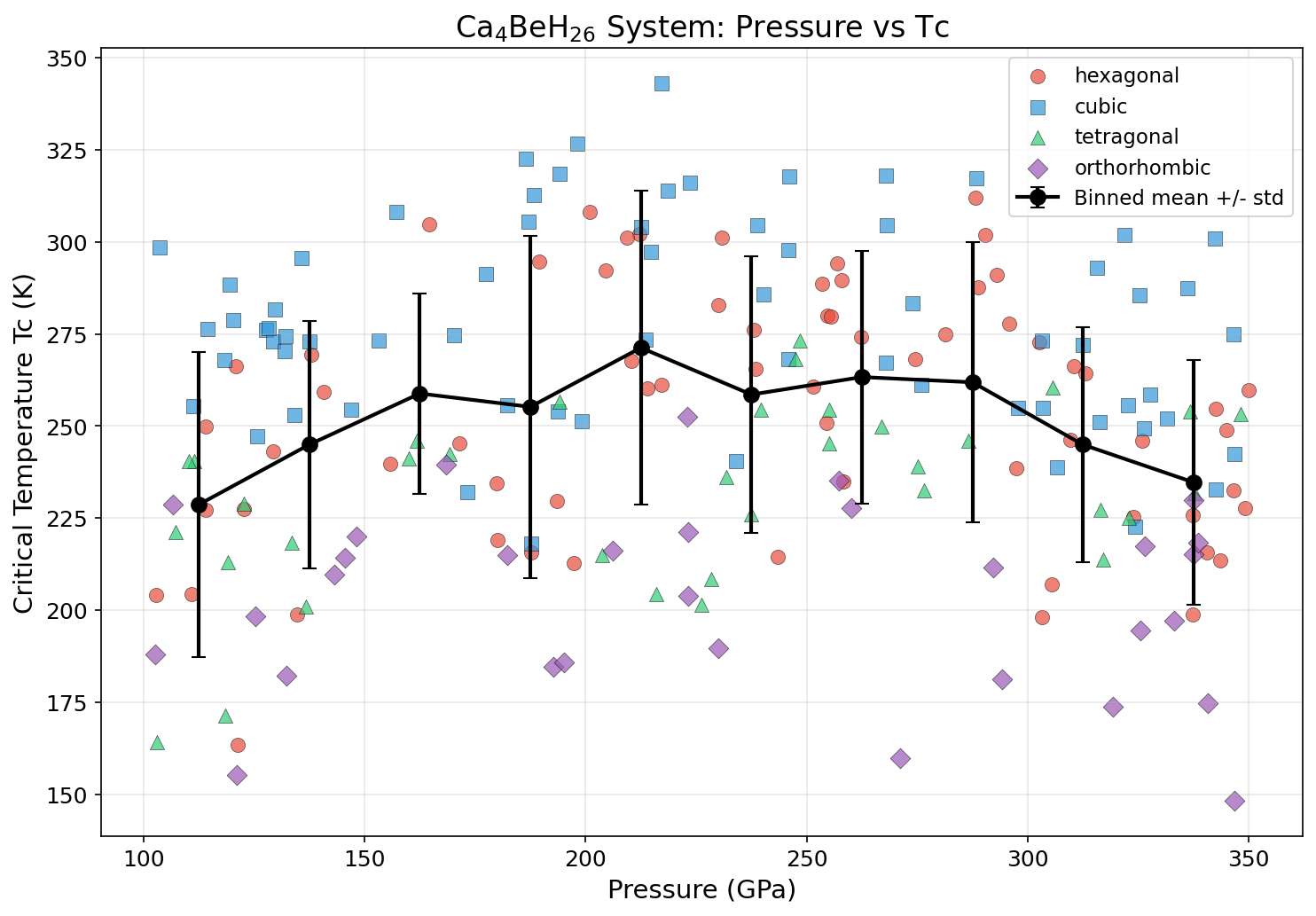
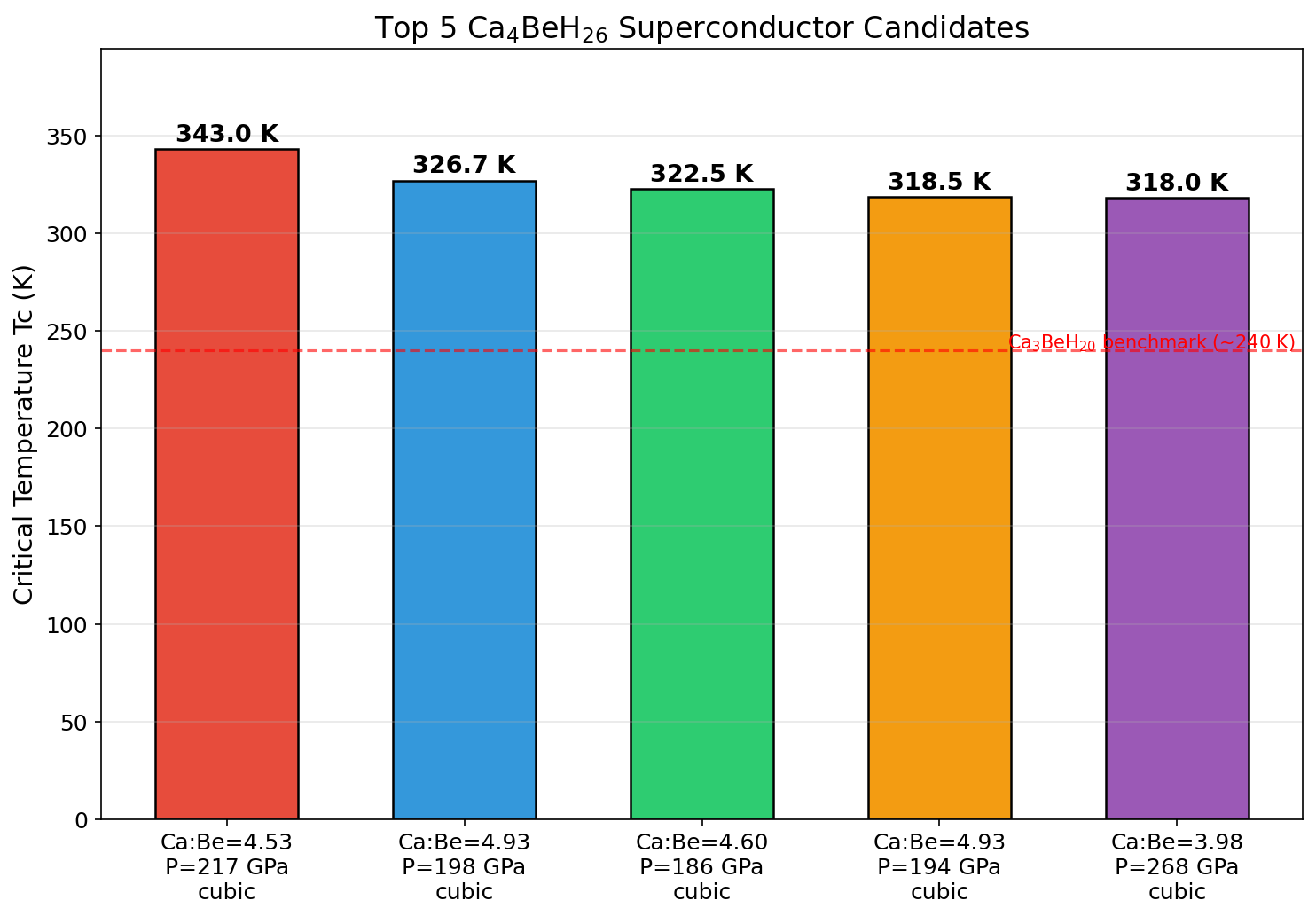

Comparison with Known Superconductors
To appreciate the significance of Ca₄BeH₂₆'s predicted 343 K critical temperature, it's worth placing it alongside the most celebrated superconductors discovered or predicted to date. Each of these materials represents a milestone in the field, and each comes with its own trade-offs between operating temperature, required pressure, and practical feasibility.
- H₃S (Hydrogen Sulfide): Experimentally confirmed Tc of approximately 203 K at 150 GPa. This was the breakthrough compound in 2015 that validated the superhydride approach to high-temperature superconductivity. Compared to Ca₄BeH₂₆'s predicted 343 K, H₃S operates a full 140 degrees colder — but it has the crucial advantage of experimental verification.
- LaH₁₀ (Lanthanum Decahydride): Confirmed Tc near 250–260 K at roughly 170 GPa. LaH₁₀ pioneered the clathrate hydrogen cage architecture that Ca₄BeH₂₆ also employs, demonstrating that hydrogen-dominated lattices can push superconductivity close to room temperature. Ca₄BeH₂₆ is predicted to extend this paradigm by nearly 90 K.
- MgB₂ (Magnesium Diboride): A conventional superconductor with Tc of 39 K at ambient pressure. While MgB₂'s Tc is far lower, it requires no compression at all, making it industrially useful today for MRI magnets and superconducting wires. This highlights the central trade-off: Ca₄BeH₂₆'s astonishing Tc comes at the cost of pressures comparable to those found deep inside planetary cores.
- CaH₆ and YH₉: Related calcium and yttrium superhydrides with predicted and partially confirmed Tc values between 215 K and 260 K at megabar pressures. Ca₄BeH₂₆'s inclusion of beryllium appears to be the key structural innovation that pushes the predicted Tc beyond this cluster.
The pattern is clear: every leap in critical temperature over the last decade has come from hydrogen-rich compounds under extreme pressure. Ca₄BeH₂₆, if validated, would represent the next step in that trajectory — the first predicted superconductor to operate comfortably above the boiling point of water.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, remain hypotheses until laboratory experiments confirm them. Turning Ca₄BeH₂₆ from a theoretical curiosity into a verified superconductor will require a carefully staged experimental campaign. Here is what that roadmap might look like:
- Step 1 — Sample Synthesis Under Extreme Pressure: Researchers would need to combine calcium, beryllium, and a hydrogen source (such as ammonia borane or molecular hydrogen) inside a diamond anvil cell, then compress the mixture to pressures in the 200–300 GPa range. Laser heating would provide the activation energy needed for the ternary hydride to form. Beryllium's toxicity adds a significant safety challenge that specialized labs will need to address.
- Step 2 — Structural Characterization: Synchrotron X-ray diffraction would confirm whether the synthesized sample actually adopts the predicted clathrate structure. This is a critical checkpoint — many predicted superhydrides fail to crystallize in their theoretical ground-state configuration.
- Step 3 — Electrical Resistance Measurements: Four-probe resistance measurements inside the diamond anvil cell, taken while slowly cooling the sample, would reveal whether resistance drops sharply to zero at the predicted Tc. A clean, reproducible transition is the hallmark signature of superconductivity.
- Step 4 — Meissner Effect Verification: True superconductors expel magnetic fields — the Meissner effect. Magnetic susceptibility measurements, though exceptionally difficult at megabar pressures, would provide the definitive confirmation that the zero-resistance state is genuinely superconducting rather than an artifact.
- Step 5 — Isotope Effect Studies: Replacing hydrogen with deuterium and remeasuring Tc would test whether electron-phonon coupling is indeed the driving mechanism, as the computational model predicts. A significant Tc shift upon isotope substitution would strongly support the theoretical framework.
- Step 6 — Independent Replication: Multiple laboratories worldwide would need to reproduce the results. Recent controversies in the superhydride field underscore the importance of cross-laboratory verification before any claim enters the scientific mainstream.
Realistically, this entire validation process could span five to ten years, assuming favorable results at each stage. The technical barriers are formidable, but the potential payoff — the first confirmed above-room-temperature superconductor — justifies the sustained effort.
Key Takeaways
- Unprecedented predicted Tc: Ca₄BeH₂₆'s computational Tc of 343 K would exceed room temperature by roughly 50 degrees, setting a new theoretical benchmark for conventional superconductivity.
- Synergistic ternary design: The combination of calcium (electron donor), beryllium (light

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)