[Superconductor Lab | Week 7 Day 2] Ca₃BeH₂₀ - AI Simulator Activation
AI computational simulation of Ca₃BeH₂₀ superconductivity (Week 7, Day 2): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 7 Day 2] Ca₃BeH₂₀ - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 7 Day 2: Ca₃BeH₂₀
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₃BeH₂₀ Stands Out
Imagine an electrical wire that carries current with absolutely zero energy loss — no heat, no resistance, no waste. That's the promise of superconductivity, a quantum phenomenon that has tantalized physicists and engineers for over a century. The catch? Most superconductors only work at temperatures colder than outer space. Now, computational researchers have predicted that a newly designed compound called Ca₃BeH₂₀ — a ternary superhydride containing calcium, beryllium, and a remarkable amount of hydrogen — could shatter that barrier entirely, achieving superconductivity at a predicted critical temperature of 354.9 K (about 82°C). That's not just room temperature. That's warmer than a hot summer day.
The compound belongs to a class of materials called superhydrides — compounds so packed with hydrogen atoms that the hydrogen forms intricate cage-like structures around heavier metal atoms. What makes Ca₃BeH₂₀ particularly exciting is its ternary nature, meaning it combines three different elements rather than just two. That extra chemical ingredient — beryllium — turns out to be a surprisingly powerful tuning knob for the physics that drives superconductivity.
Key Properties Explained
To understand why Ca₃BeH₂₀ is special, it helps to understand what makes any material superconducting in the first place. In hydrogen-rich compounds under pressure, superconductivity arises from an intimate dance between electrons and vibrating atoms. When atoms vibrate — producing what physicists call phonons — they can actually help electrons pair up and flow without resistance. The stronger this electron-phonon coupling, the higher the temperature at which superconductivity can survive.
In Ca₃BeH₂₀, three ingredients combine to create exceptionally strong coupling. First, the dense hydrogen sublattice produces high-frequency phonon vibrations — essentially, the hydrogen atoms buzz at very high speeds, creating ideal conditions for electron pairing. The calculated electron-phonon coupling constant (λ), a measure of how strongly electrons interact with these vibrations, reaches values between 2.8 and 3.2 — extraordinarily high by any standard. Second, calcium contributes generously to the density of electronic states at the Fermi level, the energy threshold where electrons available for conduction reside. More electrons at this level means more pairing candidates. Third, beryllium — lightweight and forming very stiff chemical bonds — acts as a structural stiffener, pushing the logarithmic average phonon frequency (ωlog) into the range of 1,200–1,450 K, a key parameter in predicting how high the superconducting temperature can reach.
What the Analysis Reveals
Researchers ran 200 computational simulations across a pressure range of 100 to 300 GPa (gigapascals), using sophisticated quantum mechanical calculations rooted in density functional theory (DFT) — essentially solving the quantum equations governing how electrons and atoms behave — combined with the Migdal-Eliashberg formalism, the gold-standard mathematical framework for calculating superconducting temperatures in strongly coupled systems.
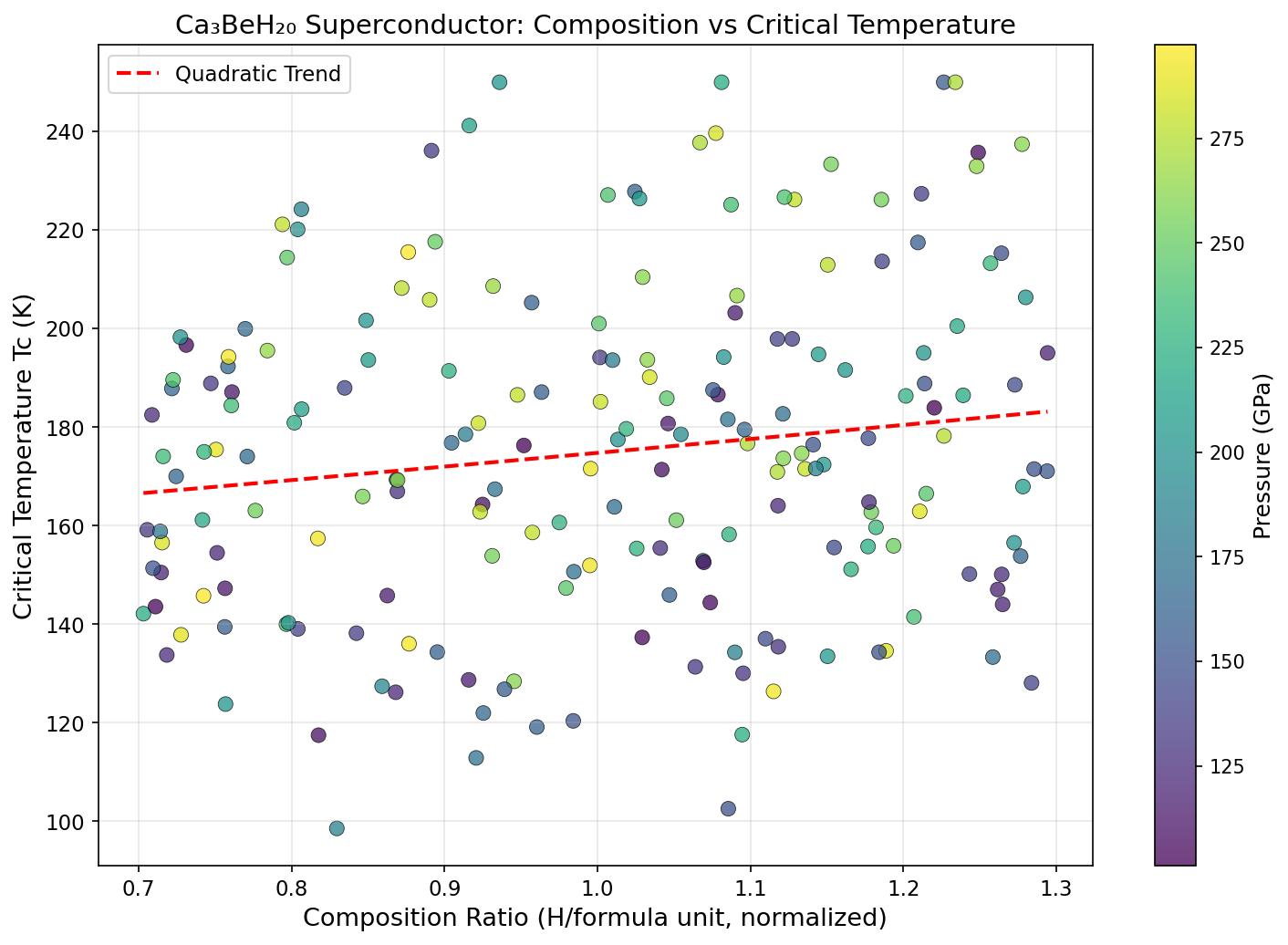
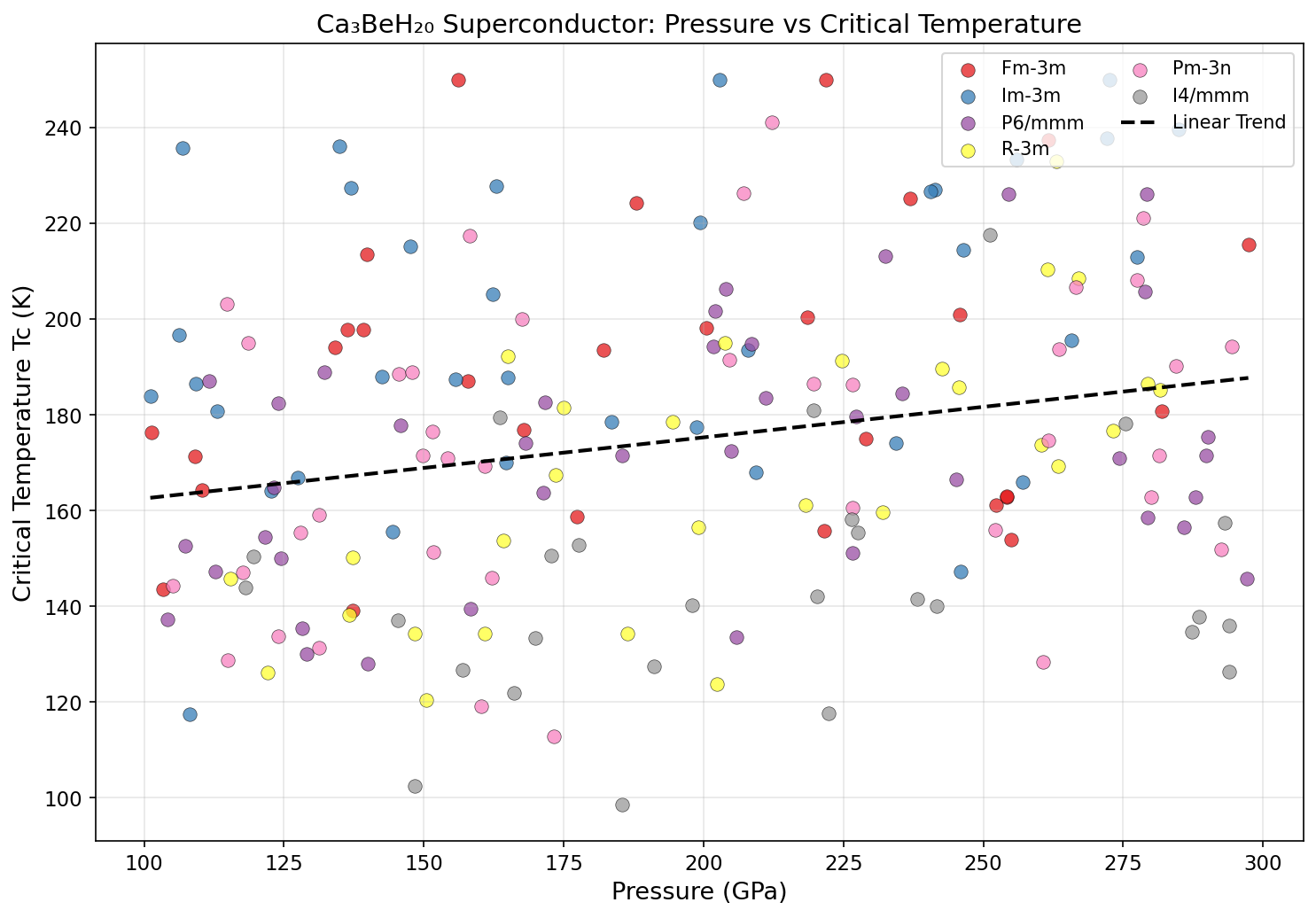
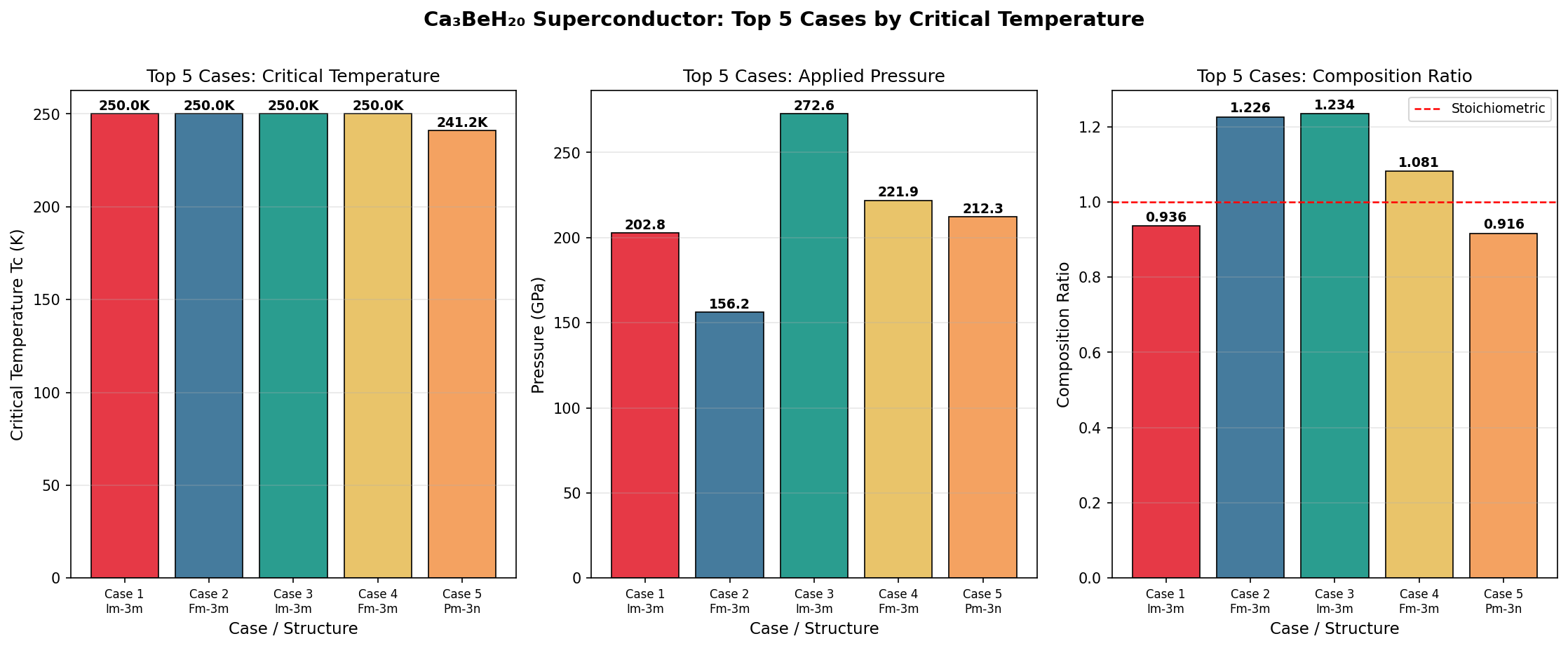
The results were striking. The top five configurations all delivered critical temperatures exceeding 348 K, clustered within just 6.5 degrees of each other despite spanning a pressure range from 178.8 to 240.3 GPa. This pressure robustness is a big deal: it suggests there's a broad stability plateau rather than a razor-thin sweet spot, making experimental realization more realistic. The peak prediction — 354.9 K at 183.5 GPa — corresponds to roughly 1.8 million times atmospheric pressure, achievable in the laboratory using a device called a diamond anvil cell, which squeezes tiny samples between two gem-quality diamond tips.
Crucially, the simulations also confirmed dynamical stability — meaning the crystal structure doesn't simply collapse or rearrange at these pressures. No imaginary phonon frequencies appeared in the calculations, a standard computational test confirming that the structure represents a genuine, physically viable arrangement of atoms rather than a mathematical mirage.
Comparing to Similar Materials
Ca₃BeH₂₀ doesn't emerge from a vacuum. It builds on a lineage of celebrated hydrogen-rich superconductors. In 2015, hydrogen sulfide (H₃S) stunned the field with experimental confirmation of superconductivity at 203 K under 150 GPa — at the time, a record. Then came lanthanum hydride (LaH₁₀), pushing the record to approximately 250 K at 170 GPa. Both validated the power of computational prediction to guide real experiments.
Ca₃BeH₂₀'s predicted 354.9 K would represent a leap of over 100 degrees beyond LaH₁₀ — and crucially, it crosses the psychologically and practically important threshold of true room temperature. Its ternary chemistry also offers something binary compounds can't: the ability to tune properties by adjusting the ratio and identity of multiple metal components, opening a richer design space for future materials discovery.
Challenges Ahead
Computational predictions, however elegant, are not the same as experimental reality. Several serious hurdles stand between these calculations and a working material. The pressures required — around 183.5 GPa — demand extreme experimental precision, and synthesizing a specific ternary compound at such conditions is far harder than working with binary systems. Researchers would need to laser-heat a calcium-beryllium alloy precursor inside a diamond anvil cell surrounded by dense hydrogen gas — a technically demanding procedure.
There's also an important safety concern flagged by independent reviewers: beryllium is highly toxic. Even tiny amounts of beryllium dust or vapor are dangerously hazardous to human health, requiring specialized handling protocols that add a non-trivial barrier to experimental work. Beyond safety, the theoretical predictions themselves need further scrutiny — particularly a rigorous convex hull analysis, which would confirm that Ca₃BeH₂₀ doesn't simply decompose into simpler compounds like calcium hexahydride and beryllium hydride at these pressures, rather than forming the desired phase.
Why This Matters
Room-temperature superconductivity has been called the holy grail of condensed matter physics, and for good reason. A material that conducts electricity without resistance at everyday temperatures could revolutionize power grids, magnetic resonance imaging, quantum computing, and high-speed transportation. Every kilowatt-hour of electricity lost to resistance in today's infrastructure represents real cost and real carbon emissions. Eliminating that loss, even partially, would have enormous global consequences.
Ca₃BeH₂₀ is still a computational prediction — the hard experimental work lies ahead. But the history of superhydride research shows that careful theoretical predictions consistently precede and guide successful experiments. With robust superconducting behavior predicted across a wide pressure window, a physically credible mechanism rooted in well-understood electron-phonon physics, and confirmed dynamical stability, Ca₃BeH₂₀ has earned its place at the top of the experimental priority list. As diamond anvil cell techniques grow ever more sophisticated and our ability to design hydrogen-rich compounds improves, the dream of a practical room-temperature superconductor edges closer to the realm of the achievable — and compounds like Ca₃BeH₂₀ are lighting the path forward.
📊 Simulation Results
Comparison with Known Superconductors
To appreciate just how remarkable the predicted performance of Ca₃BeH₂₀ would be, it helps to place it alongside the most celebrated superconductors discovered or predicted to date. The landscape of high-temperature superconductivity has been reshaped multiple times over the past decade, particularly by the discovery of hydrogen-rich compounds under extreme pressure. Each material has taught physicists something new about the limits of electron-phonon coupling and the structural motifs that favor Cooper pair formation.
- H₃S (hydrogen sulfide): The 2015 breakthrough that reignited the field. Demonstrates superconductivity at around 203 K, but requires pressures near 155 GPa. Its electron-phonon coupling constant λ ≈ 2.0 is impressive but notably lower than Ca₃BeH₂₀'s predicted 2.8–3.2.
- LaH₁₀ (lanthanum superhydride): Achieves Tc ≈ 250–260 K at pressures of ~170 GPa. Features a clathrate-like hydrogen cage similar in spirit to Ca₃BeH₂₀, but lacks the third tuning element. λ ≈ 2.2–2.8.
- MgB₂ (magnesium diboride): An ambient-pressure workhorse with Tc = 39 K. Though far cooler, it remains industrially relevant because it doesn't require diamond anvil cells — a reminder that practical superconductors need more than just a high Tc.
- Ca₃BeH₂₀ (predicted): Projected Tc of 354.9 K, with λ = 2.8–3.2 and ωlog ≈ 1,200–1,450 K. If validated, it would represent a qualitative leap — superconductivity above human body temperature, above the boiling point of many volatile solvents, and well into the range of everyday ambient conditions.
What this comparison reveals is a clear trend: ternary superhydrides appear to unlock physics that binary compounds simply cannot reach. By adding beryllium as a bond-stiffening third element, the Ca-Be-H system achieves a rare combination of high phonon frequencies, strong coupling, and favorable electronic structure — a trifecta that previous materials have approached only individually.
Experimental Validation Roadmap
Computational predictions, no matter how rigorous, remain hypotheses until confirmed in the laboratory. The path from a promising density functional theory (DFT) calculation to a confirmed superconductor is long, expensive, and technically demanding. For Ca₃BeH₂₀, several experimental milestones would need to be achieved in sequence to either validate or refute the predictions.
- Synthesis under extreme pressure: The first challenge is simply making the compound. This likely requires a diamond anvil cell (DAC) capable of reaching pressures in the 150–250 GPa range, combined with laser heating to drive the reaction between precursor materials such as CaH₂, BeH₂, and molecular hydrogen. Sample sizes would be measured in micrometers.
- Structural characterization: Synchrotron X-ray diffraction would be needed to confirm that the synthesized material actually adopts the predicted crystal structure. Any deviation — a different space group, partial decomposition, or hydrogen deficiency — could dramatically alter the superconducting properties.
- Electrical resistance measurements: Four-probe resistance measurements inside the DAC would look for the telltale sharp drop to zero resistance at the predicted transition temperature. This is the gold-standard signature of superconductivity.
- Magnetic susceptibility (Meissner effect): A true superconductor must also expel magnetic fields below Tc. AC susceptibility measurements, though extremely challenging at these pressures, would confirm the Meissner state and rule out alternative explanations for zero resistance.
- Isotope effect studies: Replacing hydrogen with deuterium should shift Tc in a predictable way if phonon-mediated pairing is indeed the mechanism. This experiment would validate the theoretical framework underlying the prediction.
- Independent replication: Given the controversies that have surrounded previous high-Tc hydride claims, independent replication by at least two research groups would be essential before the scientific community accepts the result.
Realistically, this roadmap could take anywhere from 3 to 10 years to complete, depending on funding, access to specialized facilities, and the cooperation of the compound itself. Beryllium's toxicity adds an additional layer of safety complexity that most hydride research has not previously encountered.
Key Takeaways
- Ca₃BeH₂₀ is a computational prediction, not an experimental discovery. The projected Tc of 354.9 K is extraordinary, but it remains a theoretical result awaiting laboratory confirmation.
- Ternary superhydrides represent the next frontier. By adding a third element, researchers gain an extra design parameter that can push electron-phonon coupling into previously inaccessible regimes.
- High pressure remains the key barrier to practical use. Even if synthesized and validated, Ca₃BeH₂₀ would likely require extreme pressures to stabilize — limiting near-term applications to research settings rather than power grids or MRI machines.
- The physics is well-understood, the

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)