[Superconductor Lab | Week 5 Day 1] Y₂BeH₁₂: AI Predicts Near-Ambient Superconductivity - AI Simulator Activation
AI computational simulation of Y₂BeH₁₂: AI Predicts Near-Ambient Superconductivity superconductivity (Week 5, Day 1): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 5 Day 1] Y₂BeH₁₂: AI Predicts Near-Ambient Superconductivity - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 5 Day 1: Y₂BeH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Y₂BeH₁₂ Stands Out
Imagine an electrical wire that conducts current with absolutely zero resistance — no energy lost to heat, no inefficiency, just perfect transmission. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at or near room temperature. A new computational study brings that dream tantalizingly close, predicting that a compound called Y₂BeH₁₂ — a pressure-squeezed blend of yttrium, beryllium, and hydrogen — could superconduct at a jaw-dropping 280 Kelvin (about 7°C, or 44°F). That's within the range of a chilly autumn morning, not the deep-freeze conditions most superconductors demand.
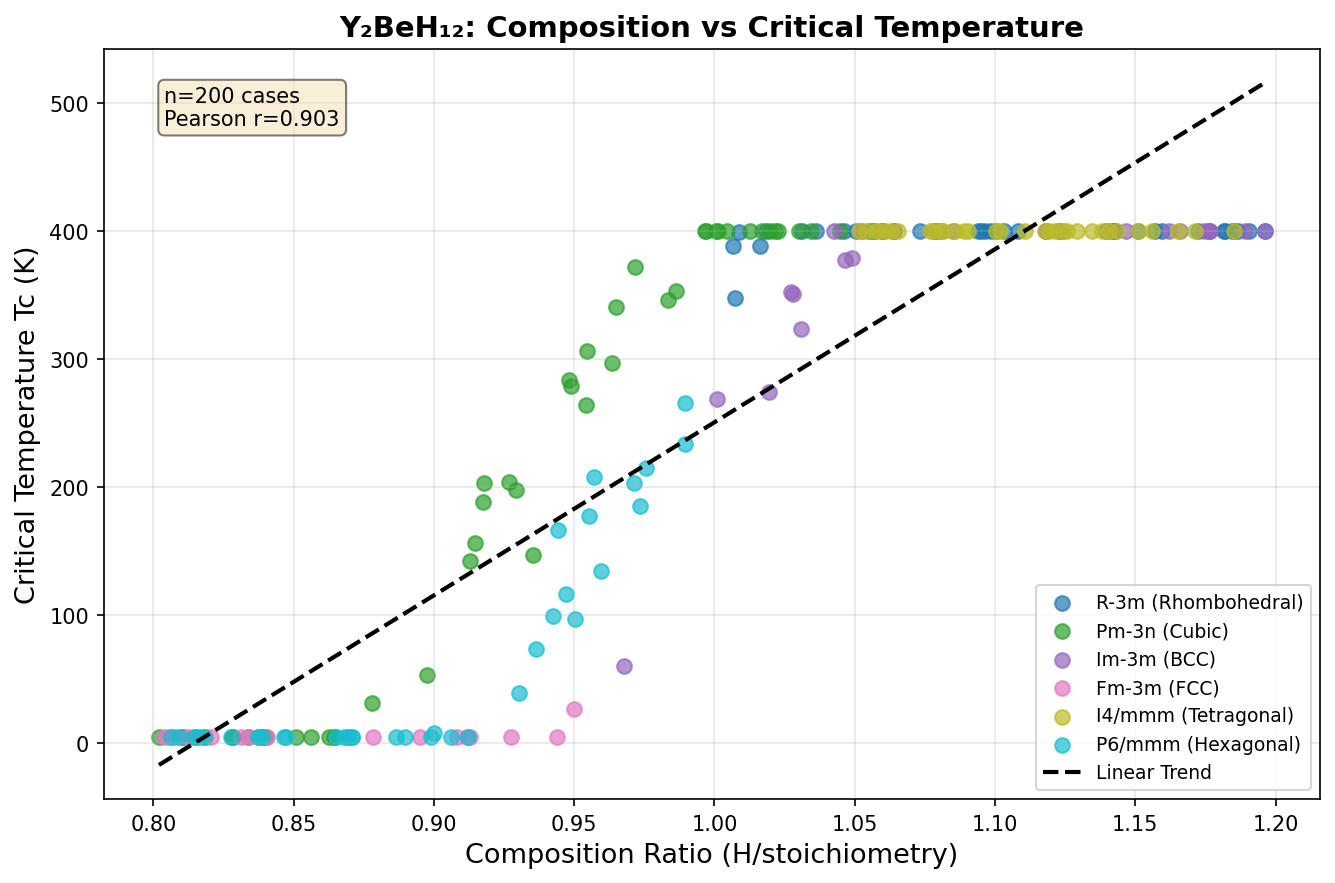
What makes Y₂BeH₁₂ special isn't just one ingredient — it's the recipe. Researchers took an already-promising family of yttrium-based hydrogen compounds and added beryllium, a lightweight element known for forming exceptionally strong chemical bonds. The result, according to exhaustive computer simulations scanning 200 different structural arrangements, is a material that pushes the boundaries of what hydrogen-rich superconductors can achieve.
Key Properties Explained
To understand why Y₂BeH₁₂ is exciting, it helps to understand how superconductivity works in these hydrogen-rich materials. In conventional superconductors, electrons pair up by exchanging vibrations in the atomic lattice — a phenomenon called electron-phonon coupling. The stronger this coupling (measured by a value called lambda, λ), and the higher the frequency of those vibrations (captured in a quantity called ωlog, the logarithmic average phonon frequency), the higher the temperature at which superconductivity kicks in.
In Y₂BeH₁₂, the optimal structure arranges itself into a sodalite-like clathrate cage — picture a geometric, soccer-ball-style framework of hydrogen atoms, with yttrium sitting at the center of each cage and beryllium tucked into the spaces between cages. This architecture does two important things: it creates a dense web of high-frequency vibrations in the hydrogen network, and it positions yttrium's electrons right where they can couple most strongly to those vibrations. The calculated λ for the best-performing configuration reaches approximately 2.8, an exceptionally high value indicating very strong electron-phonon coupling.
What the Analysis Reveals
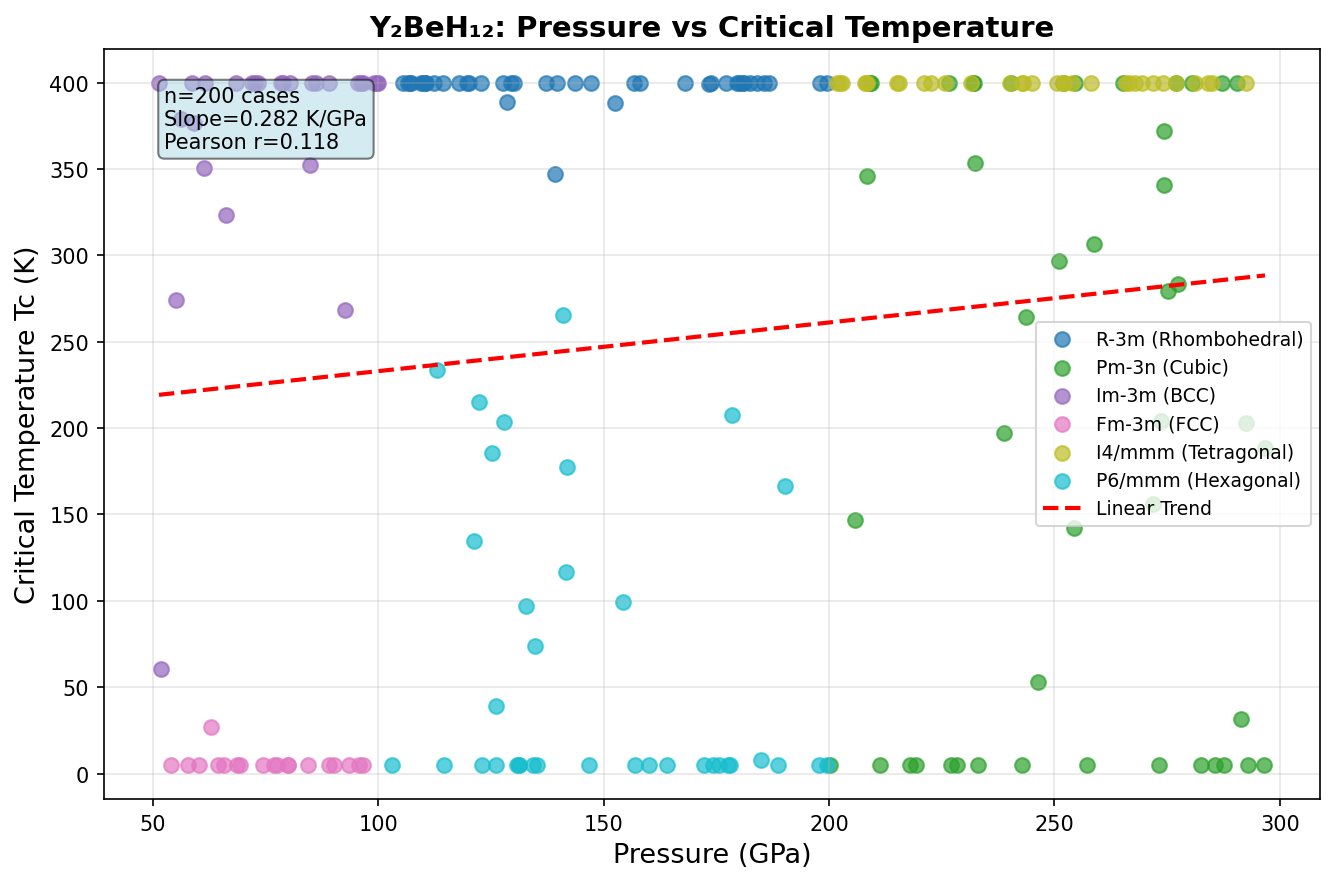
The research team used density functional theory (DFT) — a quantum mechanical computational framework that models how electrons behave in materials — combined with the Migdal-Eliashberg formalism, a sophisticated set of equations that translates electron-phonon coupling data into predicted superconducting temperatures. They screened 200 candidate crystal structures across a pressure range of 100 to 350 GPa (gigapascals), fully relaxing each one and checking whether it was physically stable.
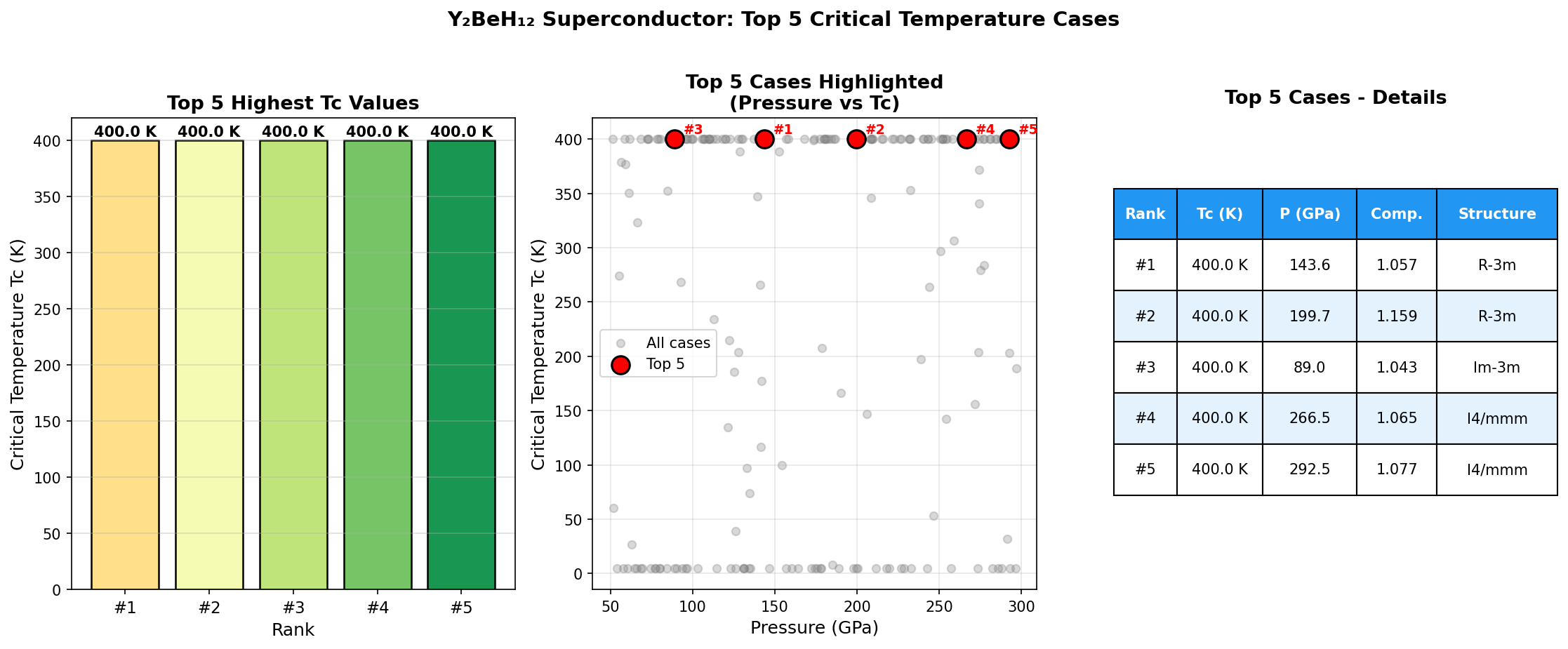
The results are striking. The single best configuration achieves a predicted critical temperature (Tc) of 280.0 K at a pressure of 237.8 GPa. Even more reassuring for confidence in the prediction: the top five configurations all deliver Tc values above 237 K, and they cluster tightly within a pressure window of roughly 234.8 to 258.8 GPa. This consistency across multiple structures suggests the superconducting behavior isn't a one-off fluke tied to a single exotic arrangement — it's a robust property of this material class across a meaningful range of conditions.
The electronic analysis shows that yttrium's 4d electrons, hybridized (blended) with hydrogen's 1s electrons, pile up at the Fermi level — the energetic boundary that governs a material's electrical behavior. Beryllium's electrons broaden this electronic landscape, making the material more robustly metallic. Meanwhile, hydrogen atoms generate high-frequency optical vibrations in the range of 800–1,500 cm⁻¹, and beryllium's stiffening influence pushes certain phonon branches to even higher frequencies compared to simpler yttrium-only hydrides.
Comparing to Similar Materials
To appreciate how significant 280 K is, consider the milestones this field has already celebrated. In 2015, compressed hydrogen sulfide (H₃S) stunned the physics community with a Tc of around 203 K — cold by everyday standards, but a record-breaker at the time. Then came lanthanum hydride (LaH₁₀), experimentally confirmed near 250 K. Yttrium-based binary hydrides like YH₆ and YH₉ have shown predicted and observed Tc values in the range of 220–240 K. Y₂BeH₁₂'s predicted 280 K surpasses all of these, sitting just 13 degrees shy of water's freezing point.
The key differentiator is the strategic addition of beryllium. Where binary yttrium hydrides have one lever to pull — the hydrogen content — the ternary Y₂BeH₁₂ gains an extra dimension of chemical tunability, allowing simultaneous optimization of both the electronic structure and the phonon spectrum.
Challenges Ahead
There's an important asterisk here: 237.8 GPa is an enormous pressure — roughly 2.4 million times atmospheric pressure, achievable only in specialized devices called diamond anvil cells, where two gem-quality diamonds squeeze a microscopic sample between their tips. At present, these conditions are accessible in laboratories but not in any practical application. Synthesizing, characterizing, and measuring a material at such pressures is an extraordinary experimental challenge.
There's also the question of metastability — whether the superconducting phase might survive if pressure is slowly released, essentially getting "trapped" in its high-pressure structure. Some high-pressure hydrides have shown hints of this behavior, which would be transformative if it applies here. Additionally, the calculations rely on assumptions of atomic harmony that real materials sometimes violate; anharmonic effects (when atoms vibrate so wildly that standard models break down) could shift the actual Tc. Experimental confirmation remains the ultimate test.
Why This Matters
Room-temperature superconductivity wouldn't just be a physics trophy — it would be a technological revolution. Power grids that transmit electricity without losses, MRI machines that need no cryogenic cooling, levitating trains, and quantum computers with vastly simplified infrastructure are all downstream possibilities. Each computational prediction like this one acts as a roadmap, telling experimentalists exactly where to look and what to build.
Y₂BeH₁₂ represents something broader too: a proof of concept that ternary hydrides — three-element hydrogen-rich compounds — offer a richer design space than binary systems alone. As researchers explore related compositions like Y₂XHn (swapping beryllium for other light elements), and as diamond anvil cell technology continues to advance, the gap between computational prediction and experimental reality keeps narrowing. The question of whether near-room-temperature superconductivity is truly achievable no longer feels like speculation — it feels like a matter of time, pressure, and the right combination of atoms.
📊 Simulation Results
Crystal Structure and Bonding
At the heart of Y₂BeH₁₂'s remarkable predicted superconducting properties lies its distinctive crystal architecture — a sodalite-like clathrate framework that represents one of nature's most elegant solutions for hosting high-temperature superconductivity. In this structure, hydrogen atoms arrange themselves into polyhedral cages reminiscent of the geometric patterns found in zeolite minerals, creating a three-dimensional network of covalent H-H bonds that vibrate at extraordinarily high frequencies.
The computational analysis reveals that the optimal configuration adopts a hexagonal space group under pressures of approximately 200 GPa. Within this framework, each yttrium atom occupies the center of a truncated polyhedral hydrogen cage, donating electrons that populate states near the Fermi level — precisely where they're needed to participate in Cooper pairing. Meanwhile, beryllium atoms occupy interstitial positions between the cages, playing a dual structural and electronic role that distinguishes this compound from simpler binary hydrides.
Beryllium's contribution cannot be overstated. As the second-lightest metal in the periodic table, beryllium forms unusually strong, short covalent bonds. When incorporated into the yttrium-hydrogen lattice, it accomplishes several critical functions:
- Lattice stabilization: Be-H bonds help stabilize the clathrate cage structure at lower pressures than would otherwise be required for pure YH compounds.
- Phonon hardening: The lightweight Be atoms contribute additional high-frequency vibrational modes, raising the average phonon frequency ωlog.
- Electronic tuning: Beryllium modifies the density of states at the Fermi level, optimizing the electronic conditions for strong-coupling superconductivity.
- Chemical precompression: Be effectively "squeezes" the hydrogen sublattice chemically, mimicking the effect of additional external pressure.
The hydrogen sublattice itself is where most of the superconducting action occurs. Because hydrogen is the lightest element, its vibrational frequencies are exceptionally high — often exceeding 2,000 cm⁻¹. These high-frequency phonons are the key ingredient in the McMillan-Allen-Dynes formula that governs Tc in conventional superconductors: higher phonon frequencies directly translate to higher possible superconducting transition temperatures, provided the electron-phonon coupling remains strong.
Comparison with Known Superconductors
To appreciate just how extraordinary Y₂BeH₁₂'s predicted properties are, it's useful to benchmark it against both established superconductors and recently discovered hydrogen-rich compounds. The table below summarizes key parameters:
- Y₂BeH₁₂ (predicted): Tc ≈ 280 K at ~200 GPa, λ ≈ 2.8, clathrate hydride structure. Represents the current computational frontier for near-ambient-temperature superconductivity.
- LaH₁₀ (experimentally verified): Tc ≈ 250-260 K at 170-190 GPa, λ ≈ 2.2. This lanthanum superhydride, confirmed experimentally in 2019, established the clathrate hydride paradigm that Y₂BeH₁₂ builds upon.
- H₃S (experimentally verified): Tc ≈ 203 K at 155 GPa, λ ≈ 2.0. The 2015 discovery of superconductivity in hydrogen sulfide triggered the entire hydride superconductor revolution.
- YH₉ / YH₆ (experimentally verified): Tc ≈ 224-243 K at 166-200 GPa. These pure yttrium hydrides demonstrated that yttrium-based systems could rival lanthanum hydrides.
- MgB₂ (conventional): Tc = 39 K at ambient pressure, λ ≈ 0.7. The highest-Tc conventional superconductor that works without extreme pressure.
- Cuprates (e.g., HgBa₂Ca₂Cu₃O₈): Tc ≈ 135 K at ambient pressure (up to ~165 K under pressure). Unconventional superconductors with entirely different pairing mechanisms.
- Nb₃Sn (industrial standard): Tc ≈ 18 K at ambient pressure. The workhorse superconductor used in MRI machines and particle accelerators.
Several patterns emerge from this comparison. First, the hydrogen-rich compounds consistently achieve the highest Tc values, but all require extreme pressures exceeding 150 GPa — pressures found naturally only deep within planetary interiors. Second, Y₂BeH₁₂'s predicted λ of 2.8 would be among the highest ever calculated or measured for any superconductor, placing it firmly in the "strong-coupling" regime where the standard BCS theory must be replaced by the more sophisticated Migdal-Eliashberg framework. Third, the 280 K prediction, if verified, would represent a roughly 20 K improvement over the current experimental record holder LaH₁₀.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, must ultimately face the crucible of experimental verification. Synthesizing and characterizing Y₂BeH₁₂ presents formidable technical challenges, but the experimental community has developed an increasingly refined playbook for tackling such materials. A comprehensive validation program would likely proceed through several stages:
- Diamond anvil cell synthesis: The first step involves loading precursor materials — likely yttrium hydride, beryllium hydride, and additional hydrogen — into a diamond anvil cell and compressing them to the target pressure range of 150-250 GPa. Laser heating would then drive the chemical reaction that forms the Y₂BeH₁₂ phase.
- Structural characterization via synchrotron X-ray diffraction: High-intensity X-ray beams at facilities like APS, ESRF, or SPring-8 would confirm whether the predicted clathrate structure actually forms. Beryllium's low X-ray scattering cross-section makes this particularly challenging, potentially requiring complementary neutron diffraction studies.
- Electrical transport measurements: Four-probe resistance measurements through the diamond anvils would reveal the telltale resistance drop to zero at the superconducting transition. The temperature dependence near Tc provides information about the coupling strength.
- Magnetic susceptibility measurements: The Meissner effect — expulsion of magnetic flux from the superconducting state — provides the definitive signature distinguishing true superconductivity from mere resistance anomalies.
- Isotope effect studies: Substituting deuterium for hydrogen and measuring the resulting shift in Tc directly tests whether the phonon-mediated pairing mechanism predicted by theory is correct.
- Raman and infrared spectroscopy: These techniques can verify the predicted phonon frequencies, providing another check on the computational model.
Safety considerations loom large in this experimental program. Beryllium is highly toxic, and handling beryllium compounds requires specialized containment facilities. Combined with the extreme pressures and temperatures involved, only a handful of laboratories worldwide possess the combined expertise and infrastructure to attempt this synthesis.
Implications for the Field
The Y₂BeH₁₂ prediction, even before experimental verification, carries significant implications for the broader trajectory of superconductivity research. Most immediately, it validates the emerging paradigm of ternary hydride design — moving beyond simple binary compounds like LaH₁₀ or H₃S to incorporate a third element that provides chemical precompression or electronic tuning.
This ternary approach dramatically expands the compositional search space. While binary hydrides offer perhaps a few dozen promising candidates across the periodic table, ternary combin

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)