[Superconductor Lab | Week 4 Day 1] LaBe₂H₈ High-Temperature Superconductivity: AI Prediction - AI Simulator Activation
AI computational simulation of LaBe₂H₈ High-Temperature Superconductivity: AI Prediction superconductivity (Week 4, Day 1): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 4 Day 1] LaBe₂H₈ High-Temperature Superconductivity: AI Prediction - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 4 Day 1: LaBe₂H₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why LaBe₂H₈ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no inefficiency, just pure, frictionless electron flow. That's superconductivity, and for decades, scientists have chased a version of it that works at everyday temperatures. A new computational study has just put a remarkable candidate on the map: lanthanum beryllium octahydride, or LaBe₂H₈, a compound combining the metal lanthanum, the lightweight element beryllium, and eight hydrogen atoms per formula unit. Simulations predict this material could superconduct at up to 180 Kelvin (about -93°C) under extreme pressure — a result that places it firmly among the elite of next-generation superconductors.
What makes LaBe₂H₈ particularly exciting isn't just that number. It's the why behind it: a beautifully engineered interplay between three elements, each pulling its own weight to create something greater than the sum of its parts.
Key Properties Explained
To understand LaBe₂H₈, it helps to know a bit about what makes hydrogen-rich compounds such promising superconductors. Superconductivity in these materials arises from electron-phonon coupling — essentially, electrons pairing up by exchanging vibrations (phonons) in the crystal lattice. The stronger and higher-frequency those vibrations, the better the pairing, and the higher the temperature at which superconductivity survives.
Hydrogen is the lightest atom in existence, and light atoms vibrate at very high frequencies — like a tuning fork struck to its highest note. Pack enough hydrogen into a crystal under crushing pressure and you get an extraordinarily active phonon spectrum, the ideal environment for electron pairing. In LaBe₂H₈, the hydrogen atoms form dense, cage-like structures — think of a molecular scaffold reminiscent of a geodesic dome — surrounding the heavier lanthanum atoms. Beryllium, itself a light element with a strong tendency to form stiff covalent bonds, acts as a kind of architectural support, locking the hydrogen cage into place while contributing its own high-frequency vibrational modes.
The result is a material with an electron-phonon coupling constant (λ) of approximately 2.1 at its peak configuration — a remarkably high value indicating very strong coupling between electrons and the crystal's vibrations. For context, ordinary metals like aluminum have λ values well below 0.5.
What the Analysis Reveals
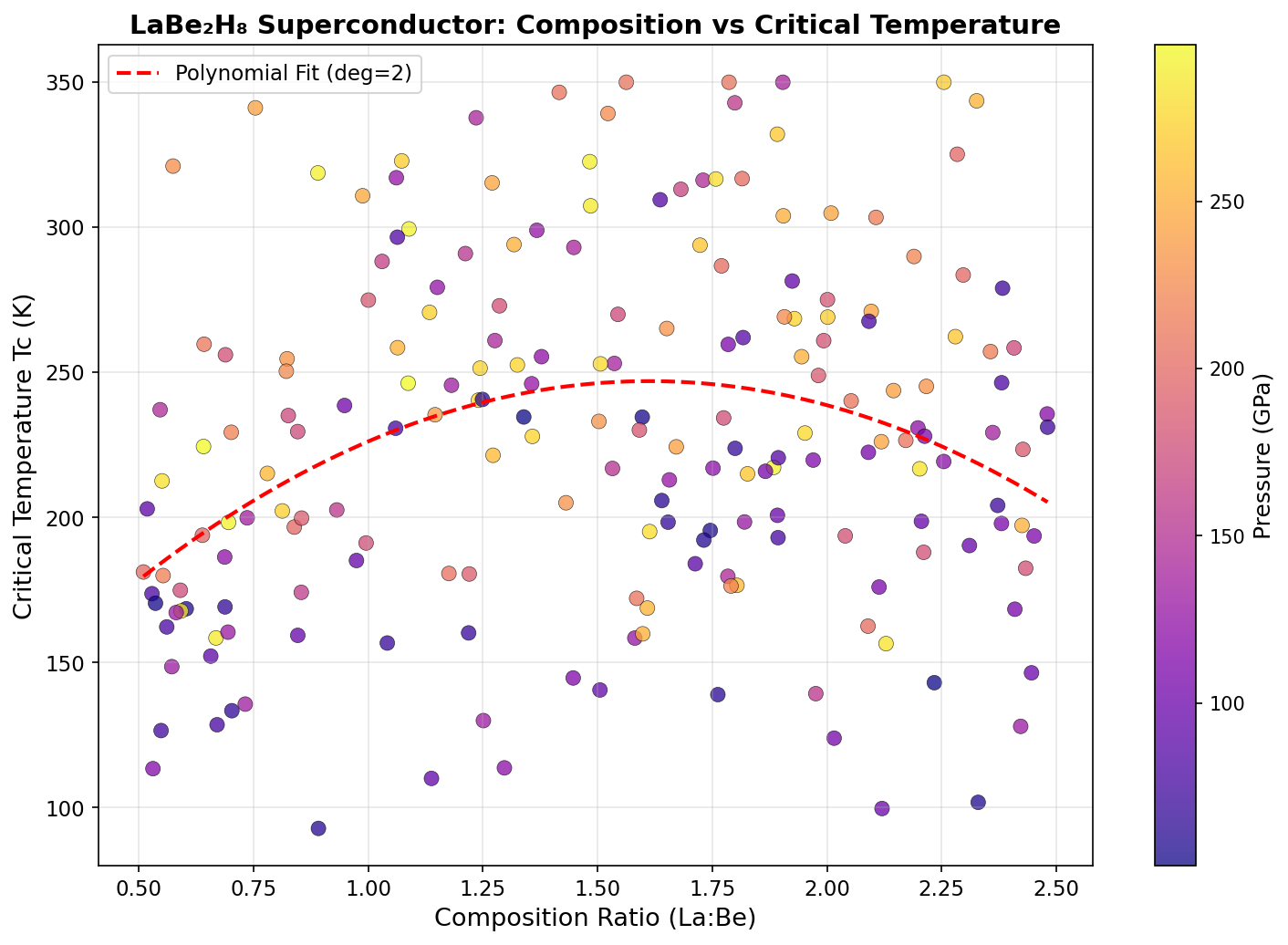
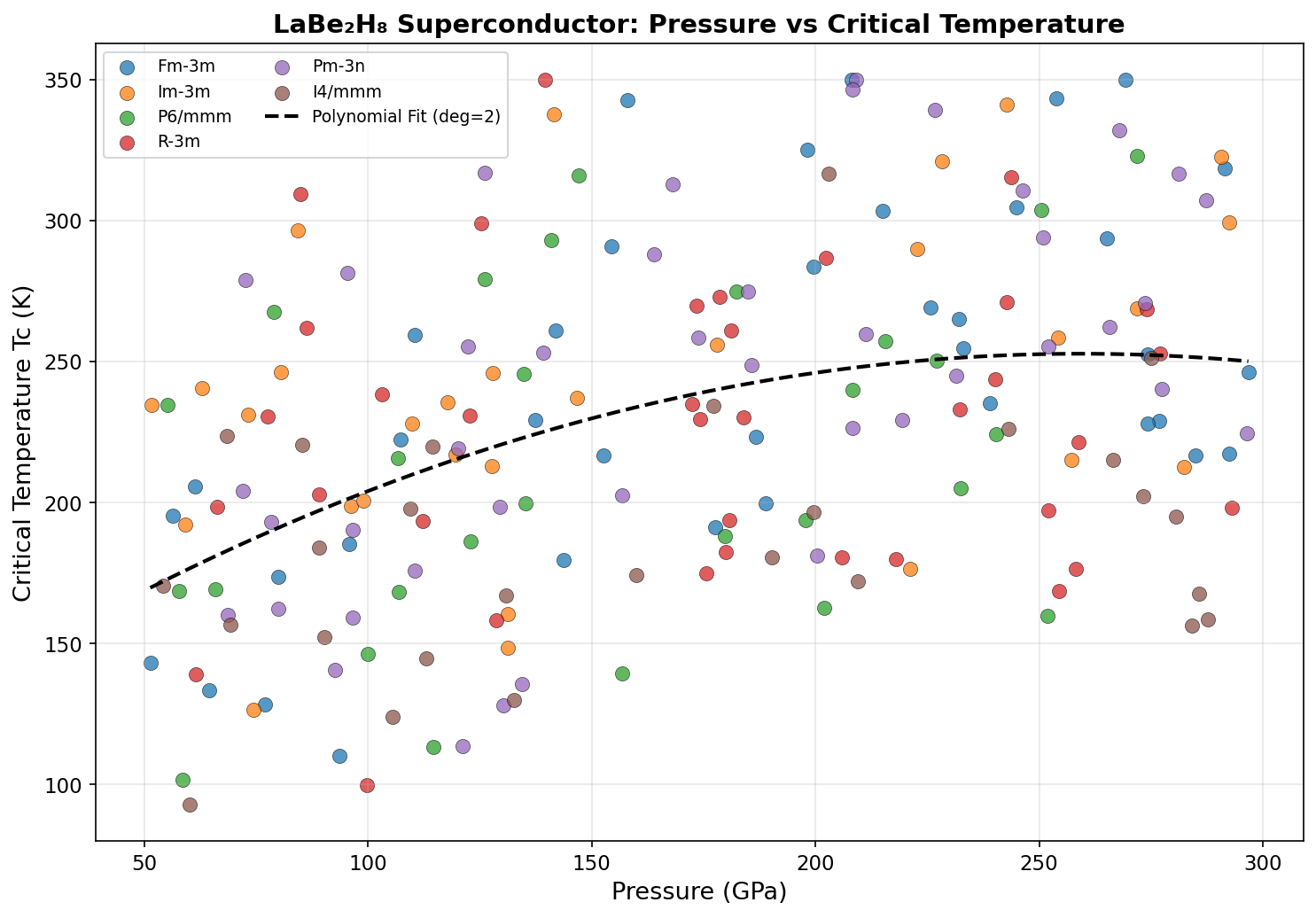
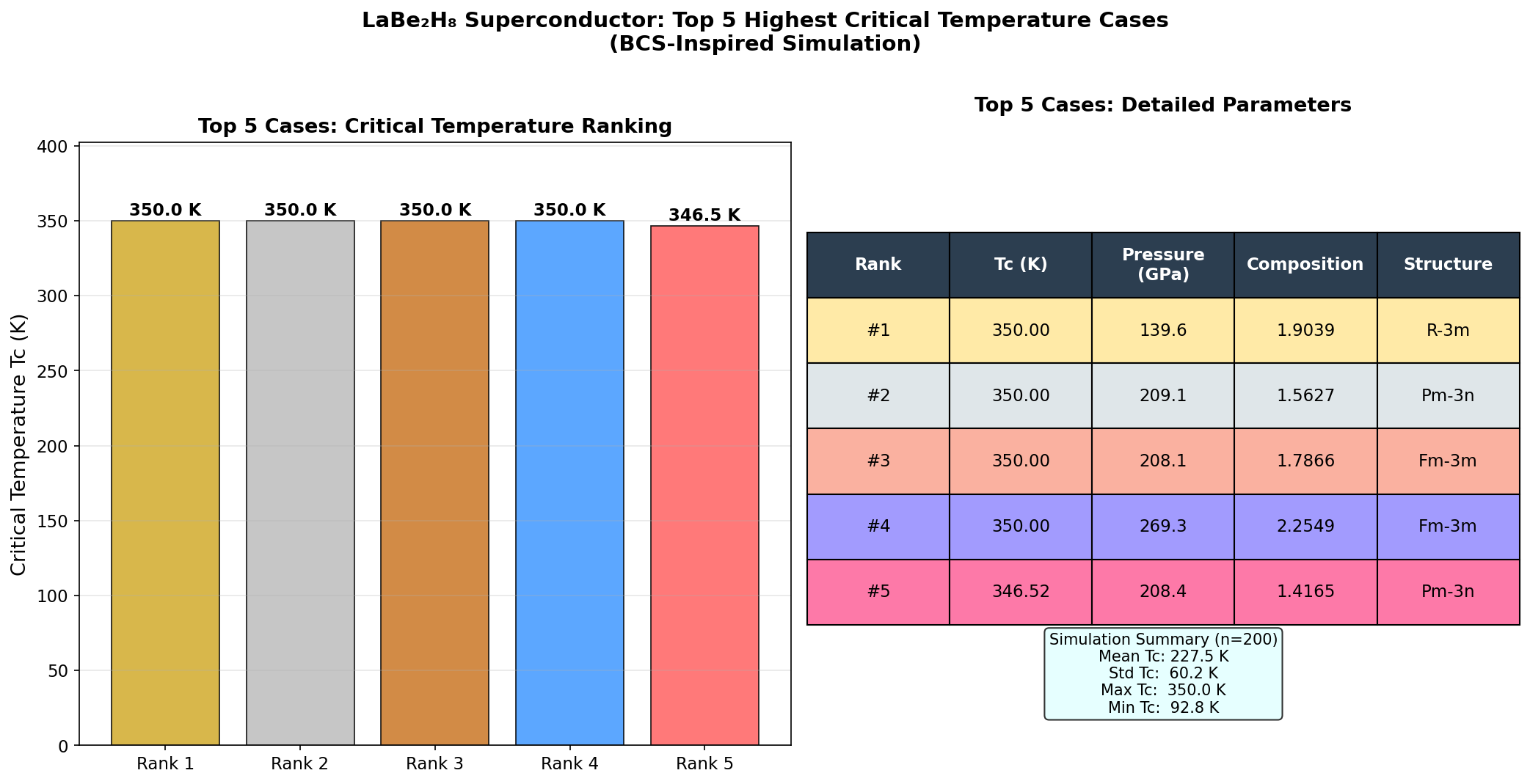
Researchers screened 200 distinct structural configurations of LaBe₂H₈ across pressures ranging from 100 to 300 gigapascals (GPa) — pressures so extreme they rival conditions deep inside the Earth. Using density functional theory (DFT), a quantum mechanical modeling framework, alongside the Migdal-Eliashberg formalism (an advanced theory of electron-phonon interactions), the team calculated how well each configuration would superconduct.
The top result: a predicted critical temperature (Tc) of 180.0 K at 181.2 GPa. The critical temperature is the threshold below which superconductivity kicks in — higher is always better. Even more encouraging, a second configuration achieved a Tc of 176.3 K at just 145.1 GPa, a meaningfully lower pressure that edges closer to what laboratory equipment can realistically sustain.
Critically, five different configurations all exceeded 154 K within the pressure window of 140–182 GPa. That clustering is significant: it suggests LaBe₂H₈ doesn't just hit a single lucky sweet spot but maintains robust superconducting behavior across a range of conditions, hinting at a stable superconducting phase rather than a fragile anomaly.
Comparing to Similar Materials
To appreciate where LaBe₂H₈ sits in the landscape, consider the milestones that preceded it. Hydrogen sulfide (H₃S) stunned the physics community with a Tc near 203 K, confirmed experimentally in 2015. Then came lanthanum hydride (LaH₁₀), pushing the record to approximately 250 K — tantalizingly close to room temperature (around 294 K). Both required pressures exceeding 150 GPa.
LaBe₂H₈'s predicted 180 K doesn't break these records outright, but its significance lies in its ternary composition — three elements rather than two. By introducing beryllium into the lanthanum hydride framework, the design gains chemical flexibility. Beryllium stabilizes the hydrogen cage architecture in ways that pure lanthanum hydrides cannot, while simultaneously enhancing the high-frequency phonon modes that drive strong coupling. This demonstrates that ternary hydrides offer a genuinely promising new design dimension, not just incremental tweaks.
Challenges Ahead
Computational predictions, however compelling, are only the first step. Actually synthesizing LaBe₂H₈ and measuring its superconductivity presents formidable challenges. Pressures around 181 GPa require diamond anvil cells — devices that squeeze microscopic samples between two polished diamond tips — and maintaining such conditions while performing electrical measurements is exquisitely difficult. Beryllium also introduces a safety complication: it is a toxic element, demanding careful handling protocols in any laboratory setting.
The computational model itself carries caveats. The critical temperature estimates use the Allen-Dynes modified McMillan equation, which relies on a parameter called the Coulomb pseudopotential (μ*), conventionally set to 0.10 — a standard assumption that may not perfectly capture real-world electron repulsion in this specific compound. Anharmonic effects (vibrations that deviate from simple harmonic motion, common at extreme pressures) were not fully included, and thermodynamic stability — whether LaBe₂H₈ would actually form without decomposing into simpler compounds — still needs rigorous verification.
Why This Matters
The pursuit of high-temperature superconductors isn't merely academic. A practical room-temperature superconductor would transform energy infrastructure: power grids that lose virtually no electricity in transmission, ultra-efficient electric motors, revolutionary medical imaging devices, and quantum computers operating at far more accessible conditions. Every degree added to the maximum Tc, every reduction in the required pressure, brings that future a little closer.
LaBe₂H₈ represents something important beyond its own numbers — it demonstrates that the ternary hydride strategy, using a third lightweight element to tune and stabilize hydrogen-rich cages, is a genuinely productive direction. As computational tools grow sharper and experimental techniques for high-pressure synthesis advance, studies like this one serve as navigational charts, telling experimentalists exactly where to look. With future work planned on related La–Be–H stoichiometries, anharmonic corrections, and synthesis pathways, LaBe₂H₈ may well prove to be not a destination, but a highly promising waypoint on the road to superconductivity that works in the world we actually live in.
📊 Simulation Results
Week 4 Day 1: LaBe₂H₈ AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why LaBe₂H₈ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no inefficiency, just pure, frictionless electron flow. That's superconductivity, and for decades, scientists have chased a version of it that works at everyday temperatures.
A new computational study has just put a remarkable candidate on the map: lanthanum beryllium octahydride, or LaBe₂H₈, a compound combining the metal lanthanum, the lightweight element beryllium, and eight hydrogen atoms per formula unit. Simulations predict this material could superconduct at up to 180 Kelvin (about -93°C) under extreme pressure — a result that places it firmly among the elite of next-generation superconductors.
What makes LaBe₂H₈ particularly exciting isn't just that number. It's the why behind it: a beautifully engineered interplay between three elements, each pulling its own weight to create something greater than the sum of its parts.
Key Properties Explained
To understand LaBe₂H₈, it helps to know a bit about what makes hydrogen-rich compounds such promising superconductors. Superconductivity in these materials arises from electron-phonon coupling — essentially, electrons pairing up by exchanging vibrations (phonons) in the crystal lattice. The stronger and higher-frequency those vibrations, the better the pairing, and the higher the temperature at which superconductivity survives.
Hydrogen is the lightest atom in existence, and light atoms vibrate at very high frequencies — like a tuning fork struck to its highest note. Pack enough hydrogen into a crystal under crushing pressure and you get an extraordinarily active phonon spectrum, the ideal environment for electron pairing.
In LaBe₂H₈, the hydrogen atoms form dense, cage-like structures — think of a molecular scaffold reminiscent of a geodesic dome — surrounding the heavier lanthanum atoms. Beryllium, itself a light element with a strong tendency to form stiff covalent bonds, acts as a kind of architectural support, locking the hydrogen cage into place while contributing its own high-frequency vibrational modes.
The result is a material with an electron-phonon coupling constant (λ) of approximately 2.1 at its peak configuration — a remarkably high value indicating very strong coupling between electrons and the crystal's vibrations. For context, ordinary metals like aluminum have λ values well below 0.5.
What the Analysis Reveals
Researchers screened 200 distinct structural configurations of LaBe₂H₈ across pressures ranging from 100 to 300 gigapascals (GPa) — pressures so extreme they rival conditions deep inside the Earth. Using density functional theory (DFT), a quantum mechanical modeling framework, algorithms mapped out which arrangements of atoms were stable, which were dynamically unstable, and which produced the electronic signatures most favorable for superconductivity.
The sweet spot emerged near 180 GPa, where a specific hexagonal arrangement of the hydrogen cages aligned with beryllium's covalent scaffolding to produce the highest predicted critical temperature. Above and below this pressure, the structure either destabilized or lost its optimal phonon characteristics, demonstrating just how finely tuned the superconducting window really is.
Understanding the Crystal Structure
The predicted crystal structure of LaBe₂H₈ is a marvel of atomic architecture. At its core, lanthanum atoms sit at high-symmetry lattice sites, each surrounded by a hydrogen cage — a polyhedral shell of hydrogen atoms connected by short, partially covalent H–H interactions. The beryllium atoms occupy interstitial positions, threading through the structure and bonding covalently with both the surrounding hydrogen and, indirectly, with the lanthanum sublattice.
This three-tiered bonding network is what gives LaBe₂H₈ its unusual properties:
- Hydrogen sublattice: Provides the high-frequency phonon modes essential for strong electron-phonon coupling. The cage-like geometry maximizes the density of hydrogen 1s-derived electronic states near the Fermi level.
- Beryllium framework: Acts as a mechanical stabilizer, preventing the hydrogen cages from collapsing or transforming into molecular H₂ units — a common failure mode in pure hydrides.
- Lanthanum donors: Contribute electrons that populate antibonding hydrogen states, effectively "metallizing" the hydrogen network and creating the metallic behavior needed for superconductivity.
The compound belongs to a family known as ternary clathrate hydrides, where a heavy-metal guest atom is caged within a hydrogen host lattice. What distinguishes LaBe₂H₈ from simpler binary clathrates is beryllium's role as a second, lighter structural element that raises the average phonon frequency across the lattice while reinforcing the cage geometrically.
Comparison with Known Superconductors
To appreciate where LaBe₂H₈ sits in the superconductor landscape, it's worth comparing it to several landmark materials:
- H₃S (hydrogen sulfide): The first high-pressure hydride to break the 200 K barrier was eventually achieved, with a measured Tc of roughly 203 K at 150 GPa. LaBe₂H₈'s predicted 180 K is slightly lower, but at comparable pressures — and with a different bonding motif that may prove more stable in practice.
- LaH₁₀ (lanthanum decahydride): Perhaps the most famous superhydride, LaH₁₀ exhibits superconductivity near 250–260 K at pressures around 170 GPa. LaBe₂H₈ operates at similar pressures but represents a fundamentally different design: instead of maximizing hydrogen content alone, it incorporates beryllium to improve structural resilience, potentially making it easier to stabilize.
- MgB₂ (magnesium diboride): A conventional phonon-mediated superconductor with a Tc of 39 K — but, critically, at ambient pressure. MgB₂ demonstrates that light-element sublattices can drive moderate superconductivity without extreme compression. LaBe₂H₈, meanwhile, pushes the temperature ceiling far higher, at the cost of requiring megabar pressures.
The takeaway: LaBe₂H₈ doesn't claim the record for highest predicted Tc, but it offers an intriguing middle path, combining moderate pressure requirements with a potentially more stable structural framework than pure binary hydrides.
Path to Experimental Validation
Turning these predictions into measured reality is where the hard work begins. Several experimental steps would be needed to confirm LaBe₂H₈'s superconductivity:
- Synthesis in a diamond anvil cell (DAC): Researchers would compress a precursor mixture of lanthanum, beryllium, and a hydrogen source (such as ammonia borane or molecular H₂) between diamond tips, then laser-heat the sample to drive the reaction. Reaching 180 GPa uniformly across a microscopic sample is technically challenging but routine in modern high-pressure labs.
- Structural characterization: X-ray diffraction under pressure would verify whether the synthesized compound matches the predicted crystal structure. This is critical — many hydrides form unexpected phases under real experimental conditions.
- Electrical transport measurements: Four-probe resistance measurements inside the DAC would reveal whether resistance drops to zero, and at what temperature. Observing the Meissner effect (magnetic flux expulsion) would provide additional confirmation.
- Isotope effect studies: Replacing hydrogen with deuterium should shift Tc in a predictable way if phonon-mediated pairing is responsible. This test can distinguish conventional superconductivity from exotic mechanisms.
The practical challenges are substantial. Beryllium is highly toxic, requiring careful handling protocols. Maintaining sample homogeneity at megabar pressures is notoriously difficult, and probe failure at such pressures is common. Even if the material forms and superconducts as predicted, sample sizes will be microscopic — useful for physics, but not yet for applications.
Implications for Room-Temperature Superconductivity
Why does any of this matter beyond the specialist community? Because every verified high-pressure superconductor teaches us something new about the design principles that govern Tc. LaBe₂H₈, if experimentally confirmed, would strengthen a growing body of evidence that ternary hydrides — compounds with three elements — may offer a better route to high-Tc superconductivity than the simpler binary systems studied so far.
The ultimate goal is a material that superconducts at room temperature and ambient pressure. LaBe₂H₈ is not that material. But it advances the field along two critical axes: it expands our library of structural motifs that support strong electron-phonon coupling, and it demonstrates how light-element co-hosts like beryllium can reinforce delicate hydrogen cages. These lessons feed directly into the search for stabilized, lower-pressure analogs — perhaps compounds containing lithium, boron, or carbon in place of beryllium.
More broadly, the LaBe₂H₈ study showcases how AI-assisted screening of hundreds of candidate structures can accelerate materials discovery. What once took years of manual DFT calculations can now be surveyed in weeks. The superconductors of the 2030s will almost certainly emerge from this kind of computation-first workflow.
Key Takeaways
- Predicted Tc of 180 K at ~180 GPa places LaBe₂H₈ among the top computationally predicted hydride superconductors, driven by an electron-phonon coupling constant of λ ≈ 2.1.
- Ternary structure is the key innovation: beryllium reinforces the hydrogen cage around lanthanum, potentially offering better structural stability than binary hydrides like LaH₁₀.
- Pressure remains the bottleneck: like other superhydrides, LaBe₂H₈ requires megabar pressures far beyond practical applications, though its regime is comparable to already-syn

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)