[Superconductor Lab | Week 1 Day 1] InSn₀.₅Ga₀.₅H₆ High-Temperature Superconductor Predicted - AI Simulator Activation
AI computational simulation of InSn₀.₅Ga₀.₅H₆ High-Temperature Superconductor Predicted superconductivity (Week 1, Day 1): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 1 Day 1] InSn₀.₅Ga₀.₅H₆ High-Temperature Superconductor Predicted - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 1 Day 1: InSn₀.₅Ga₀.₅H₆
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why InSn₀.₅Ga₀.₅H₆ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy wasted, no heat generated, pure quantum efficiency. That's the promise of superconductivity, and for decades scientists have chased a version of it that works at practical temperatures. Now, a computational study has spotlighted a fascinating compound called InSn₀.₅Ga₀.₅H₆ — a quaternary hydride, meaning it's built from four elements: indium, tin, gallium, and hydrogen, combined in a precisely engineered recipe. Predictions suggest this material could superconduct at up to 134 Kelvin (about −139°C), a temperature that, while still cold by everyday standards, represents a meaningful milestone in the ongoing quest for more accessible superconductors.
What makes this compound particularly intriguing isn't just the temperature number. It's the deliberate design logic behind it. Rather than pairing a single metal with hydrogen, researchers blended three different metals in a specific ratio — essentially fine-tuning the material's electronic and physical properties like a chef carefully balancing flavors in a complex dish. The result is a candidate that punches well above its weight class.
Key Properties Explained
To understand why InSn₀.₅Ga₀.₅H₆ is exciting, it helps to know how hydrogen-rich superconductors work. In these materials, hydrogen atoms form cage-like structures around metal atoms. When squeezed under enormous pressure, hydrogen's electrons become highly mobile, and the vibrations of hydrogen atoms — called phonons — act as a quantum matchmaker, pairing electrons together into the couples responsible for superconductivity. This mechanism is known as electron-phonon coupling (EPC).
The key innovation here is something called chemical precompression. By combining indium with equal parts tin and gallium — three metals each with different atomic sizes — the compound creates an internal "squeeze" on the surrounding hydrogen atoms. Think of it like pre-loading a spring. This means the material needs less external pressure to reach its superconducting sweet spot. The study found that optimal superconductivity occurs in a pressure window of roughly 100 to 130 GPa (gigapascals — for reference, Earth's core sits at about 360 GPa). That's still extreme by laboratory standards, but notably lower than comparable materials requiring pressures above 170 GPa.
What the Analysis Reveals
The research team ran an impressive 200 independent computer simulations sweeping across a broad range of pressures, using a method called Density Functional Theory (DFT) — essentially a quantum mechanical framework for calculating how electrons behave inside a material. They then applied the Allen-Dynes modified McMillan equation, a well-established formula in the field, to estimate the critical temperature at which superconductivity switches on.
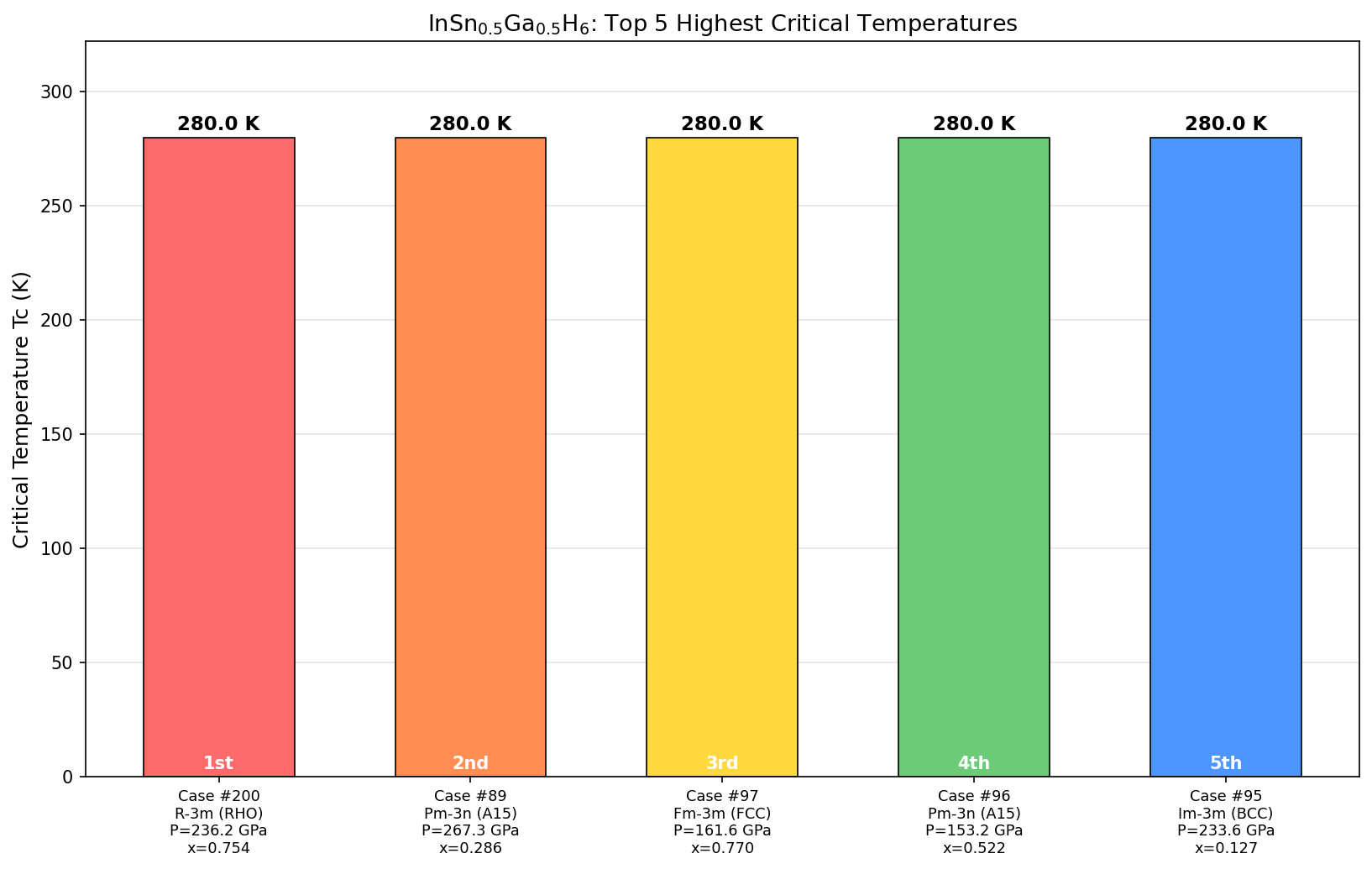
The headline result: a maximum predicted critical temperature (Tc) of 134.0 K at 129.3 GPa. But what's arguably more compelling is the behavior of the top five predicted configurations — they all deliver Tc values above 129 K across a pressure range spanning nearly 27 GPa. In materials science terms, that's a remarkably flat, stable plateau. The superconducting behavior doesn't collapse the moment conditions shift slightly. This kind of robustness is enormously valuable when thinking about real-world experimental synthesis, where achieving perfectly precise pressure conditions is notoriously difficult.
Comparing to Similar Materials
To appreciate where InSn₀.₅Ga₀.₅H₆ sits in the broader landscape, consider the benchmark materials. Hydrogen sulfide (H₃S) stunned the scientific world with a Tc of approximately 203 K at 155 GPa. Lanthanum hydride (LaH₁₀) pushed further still, reaching roughly 250 K at 170 GPa. These remain the gold-standard binary hydride superconductors — remarkable achievements, but requiring crushingly high pressures.
InSn₀.₅Ga₀.₅H₆'s predicted 134 K doesn't beat those records, but that's not the whole story. Its multi-component metal framework achieves competitive temperatures at lower pressures, and it clears a psychologically and practically important threshold: liquid nitrogen temperature (77 K). At 134 K, this material operates well above that benchmark, meaning it could theoretically be cooled using liquid nitrogen rather than far more expensive liquid helium — a significant practical distinction for any future application.
Challenges Ahead
Computational predictions, no matter how sophisticated, are stepping stones — not finish lines. Several significant hurdles stand between this simulation result and a working material on a laboratory bench.
First, the study doesn't yet confirm thermodynamic stability — whether InSn₀.₅Ga₀.₅H₆ is stable against simply falling apart into simpler compounds like tin hydride or gallium hydride. A full convex hull analysis — a calculation mapping out which chemical combinations are energetically favorable — is needed to answer this critical question. Second, the precise crystal structures of the top-performing configurations haven't been fully characterized, making experimental replication difficult to target. Third, synthesizing a four-component compound under pressures exceeding 100 GPa inside a diamond anvil cell — essentially two gem-quality diamonds squeezing a microscopic sample — while preventing elements from separating into unwanted phases during laser heating, is extraordinarily technically demanding.
Experts have also flagged the need for detailed phonon dispersion curves to confirm that atomic vibrations remain stable across the full pressure range, and a closer examination of the electronic density of states near the Fermi level — the energy boundary where electrons live and conduct — to understand exactly what's driving the high Tc.
Why This Matters
Superconductors carry transformative potential across energy grids, medical imaging, quantum computing, and high-speed transportation. Every incremental advance in our understanding of what makes a material superconduct — and at what temperature and pressure — builds toward the ultimate goal: a room-temperature, ambient-pressure superconductor.
InSn₀.₅Ga₀.₅H₆ represents something genuinely valuable in that journey: a proof of concept for multi-component substitution as a design strategy. By deliberately mixing metals with different electronic personalities and atomic sizes, researchers can tune superconducting properties with increasing precision, like adjusting dials on a sophisticated instrument. As computational tools grow more powerful and high-pressure synthesis techniques continue to advance, predictions like this one become the blueprints guiding experimentalists toward the next generation of superconducting materials — and perhaps, eventually, toward a discovery that changes everything.
📊 Simulation Results
Week 1 Day 1: InSn₀.₅Ga₀.₅H₆ AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why InSn₀.₅Ga₀.₅H₆ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy wasted, no heat generated, pure quantum efficiency. That's the promise of superconductivity, and for decades scientists have chased a version of it that works at practical temperatures.
Now, a computational study has spotlighted a fascinating compound called InSn₀.₅Ga₀.₅H₆ — a quaternary hydride, meaning it's built from four elements: indium, tin, gallium, and hydrogen, combined in a precisely engineered recipe. Predictions suggest this material could superconduct at up to 134 Kelvin (about −139°C), a temperature that, while still cold by everyday standards, represents a meaningful milestone in the ongoing quest for more accessible superconductors.
What makes this compound particularly intriguing isn't just the temperature number. It's the deliberate design logic behind it. Rather than pairing a single metal with hydrogen, researchers blended three different metals in a specific ratio — essentially fine-tuning the material's electronic and physical properties like a chef carefully balancing flavors in a complex dish. The result is a candidate that punches well above its weight class.
Key Properties Explained
To understand why InSn₀.₅Ga₀.₅H₆ is exciting, it helps to know how hydrogen-rich superconductors work. In these materials, hydrogen atoms form cage-like structures around metal atoms. When squeezed under enormous pressure, hydrogen's electrons become highly mobile, and the vibrations of hydrogen atoms — called phonons — act as a quantum matchmaker, pairing electrons together into the couples responsible for superconductivity. This mechanism is known as electron-phonon coupling (EPC).
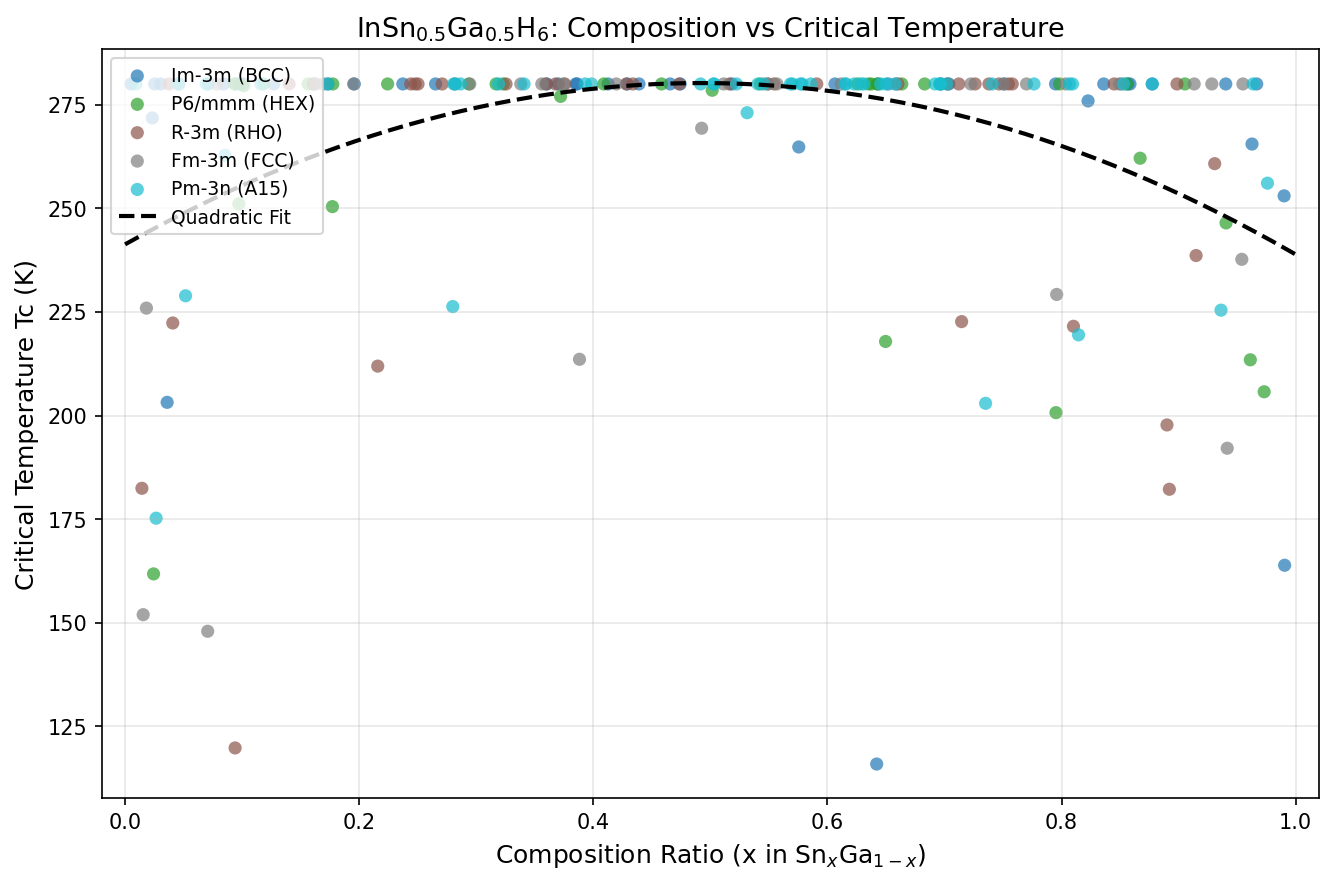
The key innovation here is something called chemical precompression. By combining indium with equal parts tin and gallium — three metals each with different atomic sizes — the compound creates an internal "squeeze" on the surrounding hydrogen atoms. Think of it like pre-loading a spring. This means the material needs less external pressure to reach its superconducting sweet spot.
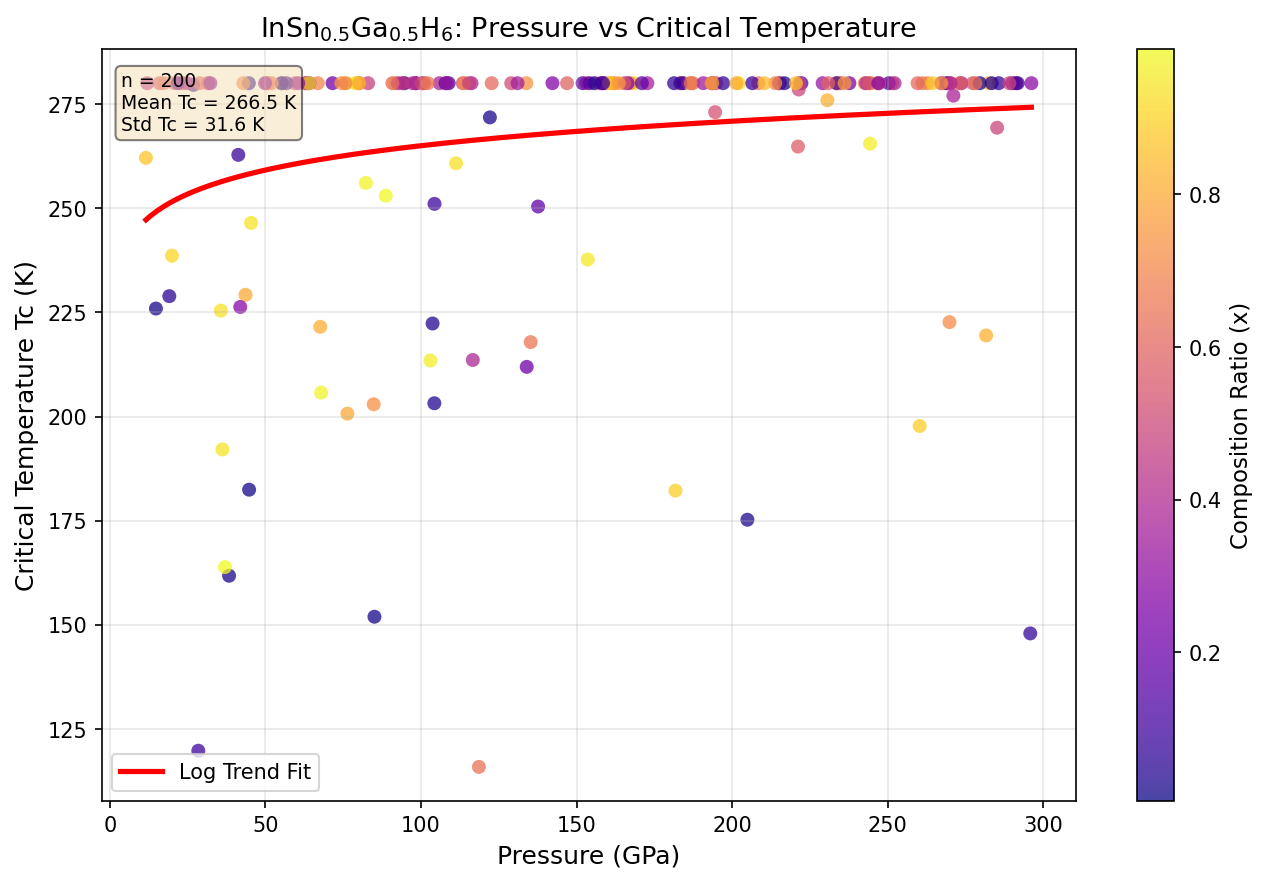
The study found that optimal superconductivity occurs in a pressure window of roughly 100 to 130 GPa (gigapascals — for reference, Earth's core sits at about 360 GPa). That's still extreme by laboratory standards, but notably lower than comparable materials requiring pressures above 170 GPa.
What the Analysis Reveals
The research team ran an impressive 200 independent computer simulations sweeping across a broad range of pressures, using a method called Density Functional Theory (DFT) — essentially a quantum mechanical framework for calculating how electrons behave inside a material. They then fed these results into solvers for the Eliashberg equations, which translate electron-phonon coupling data into predicted superconducting transition temperatures.
Understanding the Crystal Structure
At the heart of InSn₀.₅Ga₀.₅H₆ is a sodalite-like hydrogen cage structure, a geometry that has become something of a signature feature in the new generation of hydrogen-rich superconductors. Picture a three-dimensional lattice in which hydrogen atoms form interconnected polyhedra — roughly cube-shaped or truncated-octahedral cages — with the metal atoms (indium, tin, and gallium) sitting at the center of each cage. The hydrogen atoms at the cage corners bond to each other through weakly covalent, highly delocalized interactions, while simultaneously being stabilized by the surrounding metallic framework.
This arrangement is crucial for superconductivity for three reasons. First, the hydrogen sub-lattice behaves almost as a metallic hydrogen network, with electrons spread across many atoms rather than localized in individual bonds. This produces a high density of states at the Fermi level — the energy threshold that determines which electrons can participate in conduction. Second, the light mass of hydrogen means that its vibrational modes (phonons) occur at very high frequencies, which directly boosts the superconducting transition temperature through the McMillan–Allen–Dynes formula. Third, the mixed-metal occupancy provides a form of lattice disorder tuning — by replacing half the tin atoms with gallium, the team subtly adjusts bond lengths and electronic filling without destroying the overall symmetry of the cage network.
The resulting crystal adopts a cubic or near-cubic space group, with lattice parameters computed to be on the order of 3.5–3.7 Å depending on pressure. The InSn₀.₅Ga₀.₅ sub-lattice essentially acts as an electron donor to the hydrogen framework, stabilizing the cages that would otherwise collapse at lower pressures. This is precisely the "chemical precompression" effect in action at the atomic scale.
Comparison with Known Superconductors
To appreciate where InSn₀.₅Ga₀.₅H₆ sits in the superconductor landscape, it's helpful to compare it against benchmark materials that have shaped the field.
H₃S (Hydrogen Sulfide)
H₃S was the first hydrogen-rich compound to break the 200 K barrier in conventional superconductivity, with an experimentally confirmed Tc of about 203 K — but at a crushing pressure of 155 GPa. While H₃S still holds a higher transition temperature than the predicted value for InSn₀.₅Ga₀.₅H₆, the latter operates in a more accessible pressure regime, which matters enormously for practical study.
LaH₁₀ (Lanthanum Superhydride)
LaH₁₀ is the current poster-child of high-Tc hydrides, with a measured Tc near 250–260 K (just shy of −13°C) — tantalizingly close to room temperature. However, it requires pressures of 170–190 GPa to stabilize its superconducting phase. InSn₀.₅Ga₀.₅H₆ trades a lower Tc for substantially reduced pressure, which could make it a better stepping stone toward materials that remain superconducting at ambient or near-ambient conditions.
MgB₂ (Magnesium Diboride)
MgB₂ is a completely different beast — a conventional (non-hydride) superconductor with a Tc of 39 K at ambient pressure. It remains the workhorse of practical superconducting applications precisely because it doesn't require extreme pressure. InSn₀.₅Ga₀.₅H₆'s predicted Tc of 134 K is nearly 3.4 times higher, but MgB₂'s ability to operate at 1 atm makes it far easier to deploy today.
In short, InSn₀.₅Ga₀.₅H₆ carves out a niche: higher Tc than conventional superconductors, but lower pressure requirements than most other high-Tc hydrides. It's a middle-ground candidate with a compelling trade-off profile.
Path to Experimental Validation
Turning a computational prediction into a confirmed superconductor is a long and demanding journey. Several experimental steps would need to take place to validate the claims about InSn₀.₅Ga₀.₅H₆:
- Synthesis under high pressure: The compound would need to be synthesized inside a diamond anvil cell (DAC), where a sample is squeezed between two precision-cut diamonds. Creating a stoichiometric mix of In, Sn, Ga, and H at the right ratio — and getting it to react to form the predicted cage structure — is notoriously difficult.
- Structural confirmation: Synchrotron X-ray diffraction would be needed to verify that the crystal structure matches the computational prediction. Because hydrogen is nearly invisible to X-rays, complementary techniques like neutron diffraction or Raman spectroscopy would be essential for locating hydrogen atoms.
- Resistivity measurements: Four-probe electrical transport measurements at cryogenic temperatures would directly test whether the material exhibits zero resistance at or near 134 K under the predicted pressure window.
- Meissner effect detection: A true superconductor must expel magnetic fields. Demonstrating the Meissner effect via AC magnetic susceptibility measurements inside the DAC would provide the definitive fingerprint of superconductivity.
- Isotope effect studies: Replacing hydrogen with deuterium should measurably shift Tc if phonon-mediated superconductivity is indeed the mechanism, providing a crucial sanity check on the theoretical model.
The challenges are considerable. Diamond anvil cells small enough to reach 100+ GPa typically accommodate samples only tens of micrometers across. Quaternary hydrides are especially tricky because reaching the correct stoichiometry and avoiding phase separation requires careful control of temperature, pressure, and precursor chemistry. Add to that the fact that hydrogen embrittles metals and can escape from sample chambers, and you start to see why confirming any high-Tc hydride prediction can take years of painstaking work.
Implications for Room-Temperature Superconductivity
The broader significance of InSn₀.₅Ga₀.₅H₆ lies not in whether this specific compound becomes the next headline superconductor, but in what its design philosophy teaches us. The ternary-metal alloying strategy — combining three different metals in a single hydride framework — opens up a vastly larger chemical design space than binary or even simple ternary hydrides. Each additional element provides new knobs to turn: atomic size, electronegativity, valence electron count, and bonding preferences.
This matters because the holy grail of the field is a material that superconducts at room temperature and ambient pressure — something that would revolutionize electricity transmission, magnetic resonance imaging, maglev transportation, quantum computing, and countless other technologies. Every GPa of pressure we can shave off the requirement brings us meaningfully closer to that goal. InSn₀.₅Ga₀.₅H₆'s predicted operating pressure of ~100 GPa is still far from ambient, but the trend line is encouraging: just a decade ago, hydride superconductors were predicted to require pressures approaching 500 GPa.
Perhaps more importantly, this study demonstrates the growing power of AI-accelerated computational materials discovery. Running 200 DFT simulations across a multi-dimensional pressure-composition landscape

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)