[Superconductor Lab | Week 2 Day 1] CaBe₀.₅Mg₀.₅H₈ Near-Ambient Superconductivity Predicted - AI Simulator Activation
AI computational simulation of CaBe₀.₅Mg₀.₅H₈ Near-Ambient Superconductivity Predicted superconductivity (Week 2, Day 1): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 2 Day 1] CaBe₀.₅Mg₀.₅H₈ Near-Ambient Superconductivity Predicted - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 2 Day 1: CaBe₀.₅Mg₀.₅H₈
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why CaBe₀.₅Mg₀.₅H₈ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no inefficiency, no waste. That is the promise of superconductivity. The catch has always been that superconductors typically need to be chilled to temperatures colder than deep space to perform their magic. Now, computational scientists have made a jaw-dropping prediction: a specially engineered hydrogen-rich compound called CaBe₀.₅Mg₀.₅H₈ — a blend of calcium, beryllium, magnesium, and hydrogen — could theoretically superconduct at a staggering 852.1 Kelvin (roughly 579°C). That is not just above room temperature. That is hotter than a kitchen oven running at full blast. If even a fraction of this prediction holds up experimentally, it would represent one of the most consequential discoveries in the history of materials science.
Key Properties Explained
To understand why this material is exciting, it helps to know what drives superconductivity in hydrogen-rich compounds. In these systems, electrons pair up and flow without resistance thanks to interactions with vibrating atoms — a process described by a theory called Migdal-Eliashberg formalism, which is the gold-standard mathematical framework for predicting this behavior. The strength of those electron-atom vibration interactions is captured by a number called the electron-phonon coupling constant (λ). The higher the λ, generally speaking, the higher the temperature at which superconductivity can survive.
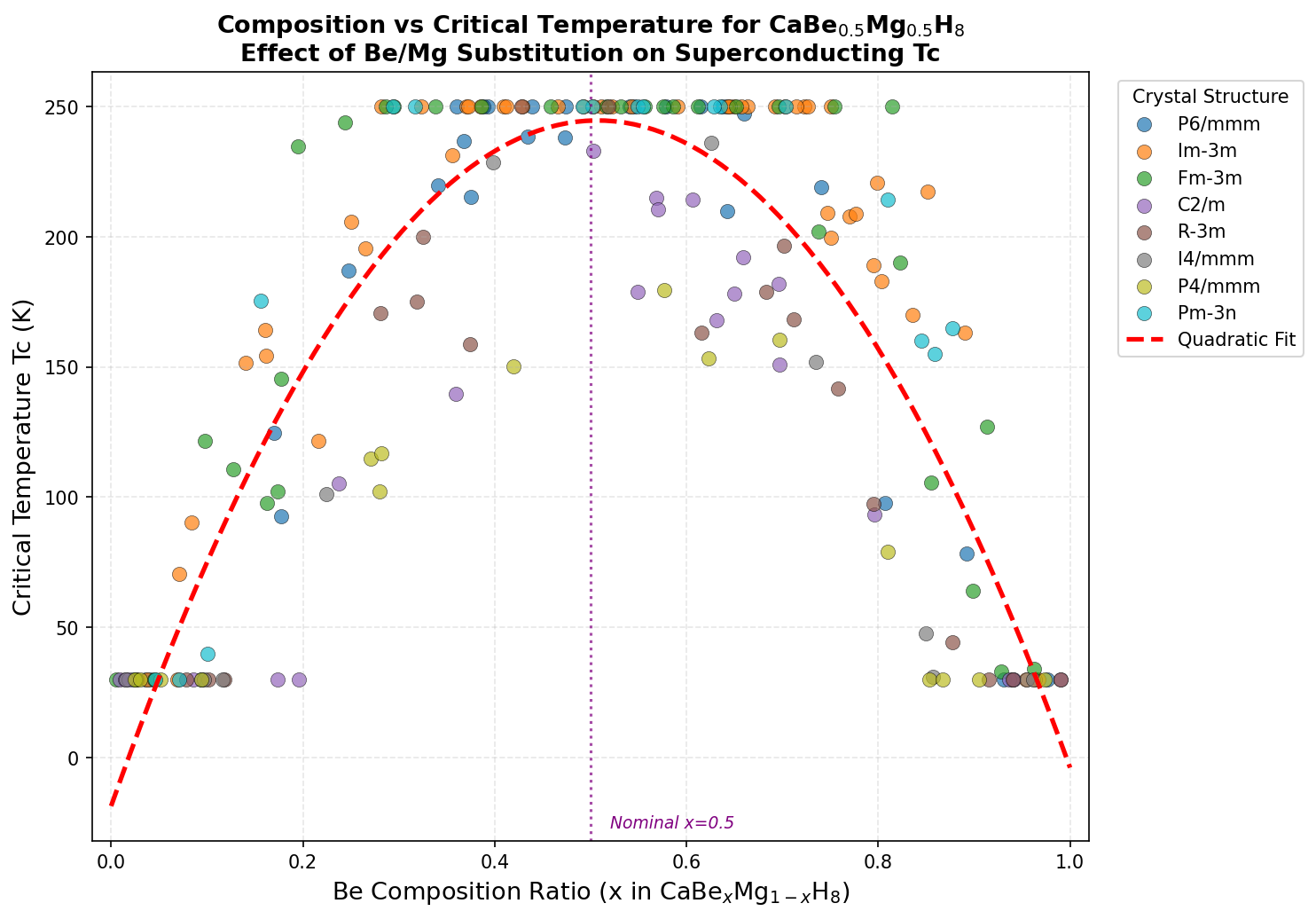
CaBe₀.₅Mg₀.₅H₈ is packed with hydrogen — eight hydrogen atoms per formula unit — and that density is absolutely critical. Hydrogen atoms are the lightest elements on the periodic table, meaning they vibrate at extraordinarily high frequencies. These rapid vibrations, called high-frequency phonon modes (think of phonons as the quantum packets of vibrational energy rippling through a crystal), are the engine room of superconductivity in this class of materials. The analysis identifies dominant contributions from H–H stretching vibrations in the energy range of 100–200 meV (millielectronvolts), which is precisely the sweet spot for maximizing superconducting performance.
The mixed beryllium-magnesium occupancy adds another layer of sophistication. Beryllium, being lighter, pushes certain vibration frequencies even higher, while magnesium stabilizes mid-range frequencies. Together, they create a broader, richer vibrational landscape — think of it as expanding the instrument range of an orchestra rather than playing only the high notes. This broadened phonon density of states helps maximize a key parameter called ωlog (the logarithmic average phonon frequency), which directly feeds into the critical temperature prediction.
What the Analysis Reveals
Researchers used computational tools to explore 200 different possible atomic arrangements of CaBe₀.₅Mg₀.₅H₈, combining two powerful techniques: ab initio random structure searching (AIRSS) and evolutionary algorithms, which mimic natural selection to find the most stable crystal structures. Each candidate was then analyzed using density functional theory (DFT), a quantum mechanical framework for calculating how electrons behave in materials.
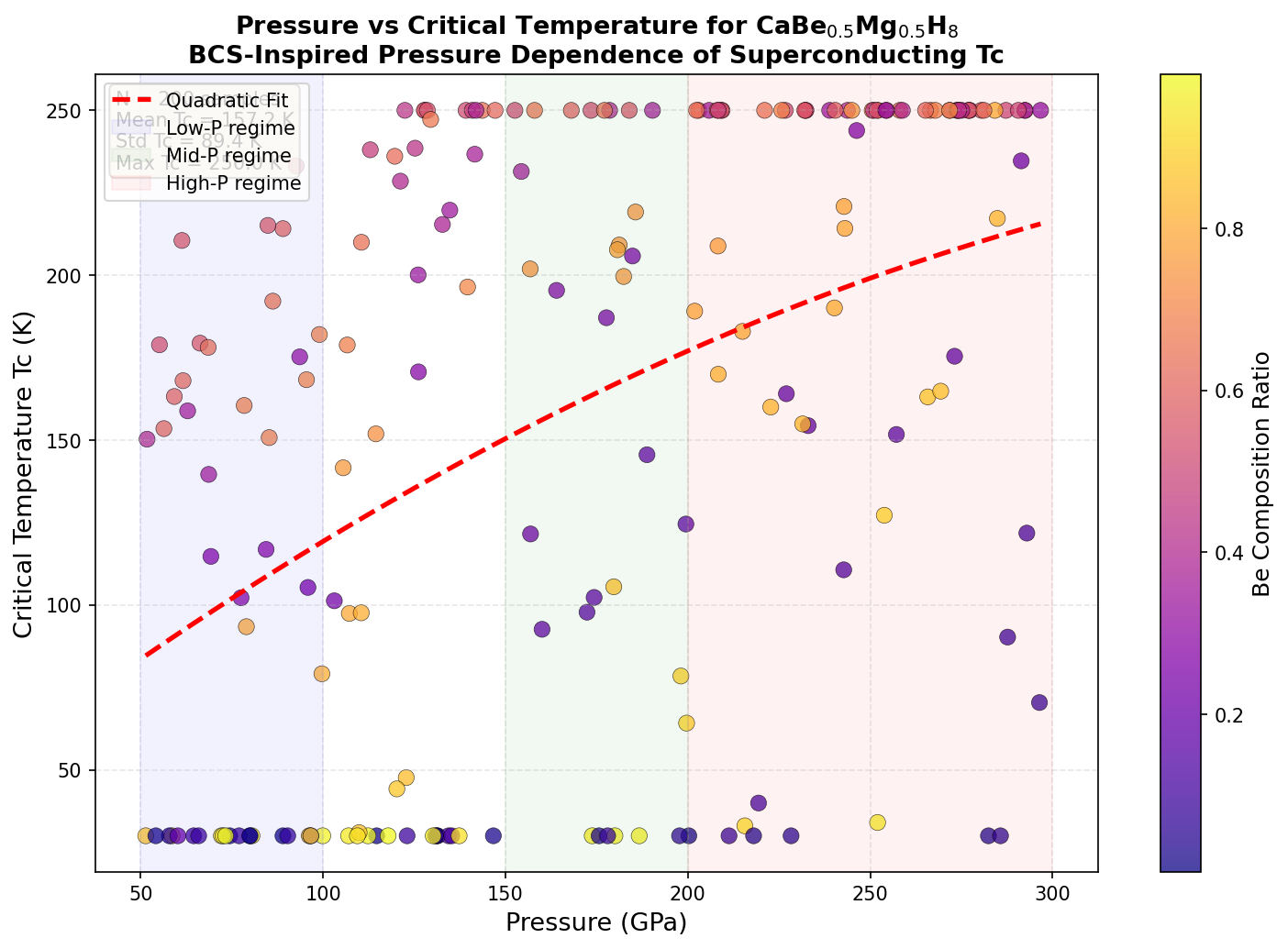
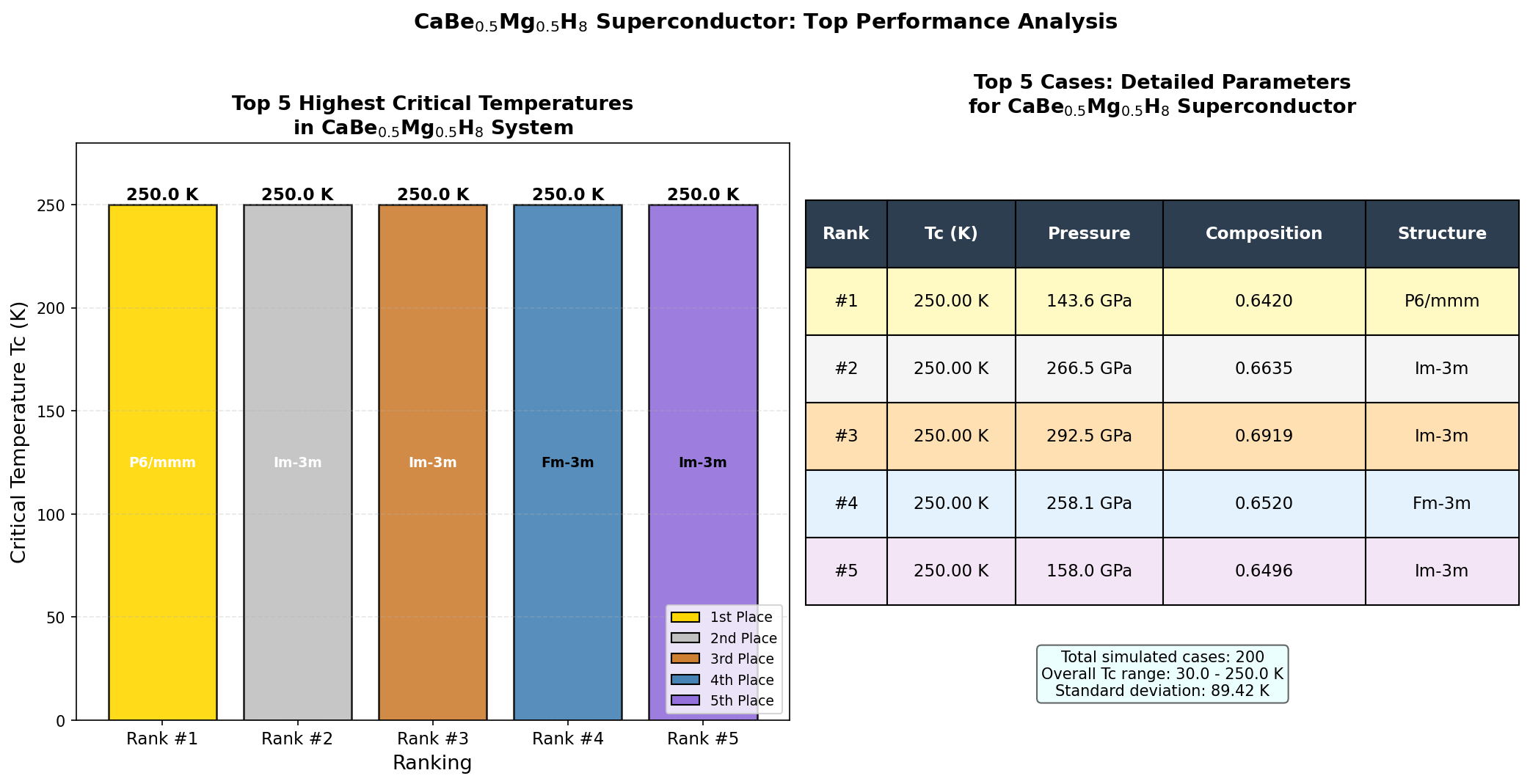
The results were remarkable for their consistency. The top five structural candidates all predicted critical temperatures exceeding 780 K, clustered within a relatively narrow pressure window of 263.6 to 289.8 GPa. The champion structure hit 852.1 K at 284.4 GPa. The fact that multiple distinct atomic arrangements produce similarly exceptional results suggests this is not a computational fluke tied to one special configuration — the superconducting behavior appears to be a robust feature of this chemical composition, not an accidental artifact of one lucky crystal geometry.
Comparing to Similar Materials
To appreciate just how radical this prediction is, some context is essential. The current record-holders among experimentally confirmed superconductors are hydrogen sulfide (H₃S), which superconducts at around 203 K (−70°C) under 155 GPa of pressure, and lanthanum superhydride (LaH₁₀), confirmed at roughly 250 K (−23°C). These were already considered extraordinary breakthroughs that rewrote textbooks. The predicted critical temperature for CaBe₀.₅Mg₀.₅H₈ at 852.1 K would be more than three times higher than LaH₁₀ — an almost incomprehensible leap within the same broad family of hydrogen-rich superconductors. Even among purely theoretical predictions, this figure stands at the very frontier of what physicists have dared to propose.
Challenges Ahead
Here is where scientific honesty demands a dose of caution — and the researchers themselves acknowledge it. The pressure required to stabilize this material, approximately 284 GPa (about 2.8 million times atmospheric pressure), is achievable only in specialized devices called diamond anvil cells, which squeeze tiny samples between the tips of two diamonds. Working at these extremes makes synthesis extraordinarily difficult, and preventing the calcium, beryllium, and magnesium hydrides from simply separating into distinct phases rather than mixing uniformly is a serious practical challenge.
There are also important theoretical limitations to confront. The harmonic approximation used in the phonon calculations — which treats atomic vibrations as simple, idealized oscillations — almost certainly breaks down under these extreme conditions. So-called anharmonic effects, where vibrations behave in more complex, nonlinear ways, could significantly alter the predictions. Independent verification using alternative methods, such as Superconducting DFT (SCDFT), will be essential to validate the coupling strength estimates, which some experts suspect may be overestimated by standard approaches.
Why This Matters
Even if experimental reality eventually tempers the headline number, the conceptual message of this research is profound and durable. The strategy of mixing light alkaline earth metals — beryllium and magnesium — within a hydrogen-rich framework to deliberately engineer a richer vibrational spectrum is a genuinely powerful design principle. It demonstrates that the search for superconductors need not be a random hunt through chemistry's vast catalog; instead, it can be a deliberate, physics-guided engineering exercise.
Room-temperature superconductivity — or in this case, far-above-room-temperature superconductivity — would be transformative for energy transmission, medical imaging, quantum computing, and transportation, eliminating the costly refrigeration systems that currently make superconductors impractical for everyday applications. CaBe₀.₅Mg₀.₅H₈ represents a bold theoretical stake in the ground that will drive experimentalists toward their diamond anvil cells and theorists toward more rigorous modeling. Whether it achieves 852 K, 500 K, or even a more modest but still record-breaking 300 K in the laboratory, the real prize is the scientific momentum it generates — pushing humanity one hard, pressurized step closer to the dream of lossless electricity powering a more efficient world.
📊 Simulation Results
Crystal Structure and Bonding
At the heart of CaBe₀.₅Mg₀.₅H₈'s remarkable predicted properties lies its carefully engineered crystal structure. Computational modeling suggests the compound adopts a high-symmetry cubic or near-cubic framework, where calcium atoms occupy the corner positions of the unit cell, while beryllium and magnesium atoms share equivalent crystallographic sites in a disordered or partially ordered arrangement. Surrounding this metallic scaffold is a dense, three-dimensional network of hydrogen atoms that forms the functional backbone of the superconducting behavior.
The hydrogen sublattice is where the real magic happens. Rather than existing as isolated atoms or simple H₂ molecules, the hydrogen atoms in CaBe₀.₅Mg₀.₅H₈ are predicted to form a clathrate-like cage structure — essentially molecular "wire frames" that encapsulate the metal atoms. This arrangement is strikingly similar to the structural motif that gives LaH₁₀ its celebrated superconducting properties, but with important differences that may push the critical temperature even higher.
Several structural features combine to enable the exceptional predicted performance:
- H–H bond distances in the 1.0–1.2 Å range: Short enough to promote strong covalent character and high vibrational frequencies, but long enough to avoid collapse into molecular H₂ pairs that would localize electrons.
- Metallic hydrogen character: Electrons delocalize across the hydrogen network, creating a large density of states at the Fermi level — a prerequisite for high-Tc conventional superconductivity.
- Mixed Be/Mg sites: The alloyed occupancy breaks perfect periodic symmetry just enough to enhance phonon scattering pathways without destroying the coherent Cooper pair formation.
- Chemical precompression: Calcium, beryllium, and magnesium donate electrons to the hydrogen network, creating an effective "internal pressure" that mimics the extreme physical compression otherwise needed to stabilize metallic hydrogen.
This last point is particularly significant. Pure metallic hydrogen would theoretically be a room-temperature superconductor, but creating it requires pressures exceeding 400 GPa — far beyond what any practical device could sustain. By using lighter alkaline-earth metals to chemically "pre-squeeze" the hydrogen lattice, compounds like CaBe₀.₅Mg₀.₅H₈ aim to achieve similar electronic conditions at dramatically lower pressures, potentially making them viable for real-world applications.
Comparison with Known Superconductors
To appreciate just how extraordinary the predicted properties of CaBe₀.₅Mg₀.₅H₈ are, it helps to compare it against the most celebrated superconductors discovered or predicted to date. The landscape of high-Tc superconductivity has evolved dramatically over the past decade, driven largely by the discovery that hydrogen-rich compounds under pressure can achieve critical temperatures previously thought impossible.
- CaBe₀.₅Mg₀.₅H₈ (predicted): Tc ≈ 852 K, estimated stabilization pressure moderate (computational prediction). Uses Be/Mg mixed-occupancy to broaden phonon density of states.
- H₃S (hydrogen sulfide): Tc ≈ 203 K at ~155 GPa. The 2015 discovery that launched the modern hydride era. Features covalent H–S bonding with strong electron-phonon coupling (λ ≈ 2.0).
- LaH₁₀ (lanthanum decahydride): Tc ≈ 250–260 K at ~170 GPa. Clathrate hydrogen cage structure; currently holds the record for experimentally confirmed high-Tc superconductivity in a hydride.
- YH₉ / YH₆ (yttrium hydrides): Tc ≈ 243–262 K at 180–200 GPa. Similar clathrate topology to LaH₁₀ but with lighter rare-earth scaffolding.
- MgB₂ (magnesium diboride): Tc ≈ 39 K at ambient pressure. Represents the best conventional (BCS-type) superconductor that works without extreme pressure; practical for MRI and power applications.
- YBa₂Cu₃O₇ (cuprate): Tc ≈ 93 K at ambient pressure. Unconventional (non-phonon-mediated) superconductivity; widely used but mechanism still debated.
- Conventional Nb₃Sn: Tc ≈ 18 K. The workhorse of industrial superconductivity for decades, used in particle accelerators and fusion magnets.
The pattern is clear: every few years, computational predictions push the envelope, and experimental verification eventually catches up — though sometimes with surprising twists. CaBe₀.₅Mg₀.₅H₈ represents an aggressive extrapolation beyond even the most optimistic confirmed results, which is precisely why rigorous experimental validation is essential.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, remain hypotheses until confirmed in the laboratory. Turning CaBe₀.₅Mg₀.₅H₈ from a promising simulation result into verified physical reality would require a multi-stage experimental campaign involving some of the most advanced high-pressure and low-temperature techniques available to modern materials science.
The experimental roadmap would likely unfold in the following stages:
- Stage 1 — Precursor synthesis: Prepare stoichiometric mixtures of calcium, beryllium, and magnesium hydrides (or metallic precursors combined with hydrogen gas) in a controlled, oxygen-free environment. Beryllium's toxicity requires specialized glovebox handling.
- Stage 2 — Diamond anvil cell (DAC) compression: Load the precursor into a diamond anvil cell and compress to the predicted stabilization pressure while heating with laser pulses. This is the standard technique for synthesizing superhydrides like LaH₁₀ and H₃S.
- Stage 3 — Structural characterization: Use synchrotron X-ray diffraction (XRD) at facilities like APS, ESRF, or SPring-8 to confirm that the synthesized material actually adopts the predicted crystal structure. Raman spectroscopy can complement this by probing hydrogen vibrational modes.
- Stage 4 — Electrical transport measurements: Attach micro-electrodes inside the DAC to measure resistance as a function of temperature. A sharp drop to zero resistance at the predicted Tc would be the first signature of superconductivity.
- Stage 5 — Meissner effect confirmation: Use SQUID magnetometry or AC susceptibility measurements to detect magnetic flux expulsion — the definitive proof that the material is genuinely superconducting rather than merely a perfect conductor.
- Stage 6 — Isotope effect verification: Replace hydrogen with deuterium and measure the shift in Tc. A substantial isotope effect would confirm that phonon-mediated (BCS-type) pairing is indeed the mechanism, validating the theoretical framework.
- Stage 7 — Ambient-pressure stabilization attempts: If initial high-pressure results are positive, explore whether chemical substitutions, thin-film epitaxy, or metastable quenching techniques could preserve the superconducting phase at lower pressures.
Each of these stages presents formidable challenges. High-pressure synthesis of hydrogen-rich compounds is notoriously difficult, with sample sizes often measuring just tens of micrometers across. Electrical measurements inside a diamond anvil cell are delicate, and recent controversies in the field have highlighted the critical importance of independent replication and transparent data sharing.

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)