[Superconductor Lab | Week 3 Day 1] Ca₂InH₁₂ Superconductivity Predicted Under High Pressure - AI Simulator Activation
AI computational simulation of Ca₂InH₁₂ Superconductivity Predicted Under High Pressure superconductivity (Week 3, Day 1): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 3 Day 1] Ca₂InH₁₂ Superconductivity Predicted Under High Pressure - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 3 Day 1: Ca₂InH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂InH₁₂ Stands Out
Imagine squeezing a material so hard that its electrons start behaving in extraordinary ways — conducting electricity with absolutely zero resistance. That's the promise of high-pressure superconducting hydrides, a class of materials that has electrified the physics community over the past decade. The latest computational addition to this exciting family is Ca₂InH₁₂, a ternary hydride (meaning it contains three distinct elements: calcium, indium, and hydrogen) packed with an unusually generous helping of twelve hydrogen atoms per formula unit. New computational research analyzing 200 independent simulation cases predicts that this compound can superconduct at temperatures up to 118.4 Kelvin — that's about –154°C — when subjected to extreme pressure. While that still sounds cold by everyday standards, in the world of superconductivity, it represents a genuinely remarkable achievement.
What makes Ca₂InH₁₂ particularly intriguing isn't just the temperature it reaches, but the elegant chemistry behind why it works. The deliberate combination of calcium, indium, and that hydrogen-rich architecture creates a system where quantum mechanical effects conspire to eliminate electrical resistance entirely.
Key Properties Explained
The mechanism at play is called electron-phonon coupling (EPC) — a quantum dance between electrons and the vibrations rippling through the atomic lattice. Those vibrations are called phonons, and when the coupling between electrons and phonons is strong enough, electrons pair up into what physicists call Cooper pairs. These paired electrons flow through the material without scattering off atoms, meaning zero electrical resistance — the defining hallmark of a superconductor.
Each element in Ca₂InH₁₂ plays a specific role in this performance. Calcium contributes favorable electronic properties that support Cooper pair formation. Hydrogen, being the lightest element, vibrates at extremely high frequencies, which is a key ingredient for strong electron-phonon coupling and elevated superconducting temperatures. Indium, a heavier post-transition metal, introduces lower-frequency vibrational modes that stabilize the overall crystal structure dynamically — even if it contributes less directly to boosting the peak temperature. Think of indium as the structural anchor that keeps the whole arrangement from falling apart under pressure.
What the Analysis Reveals
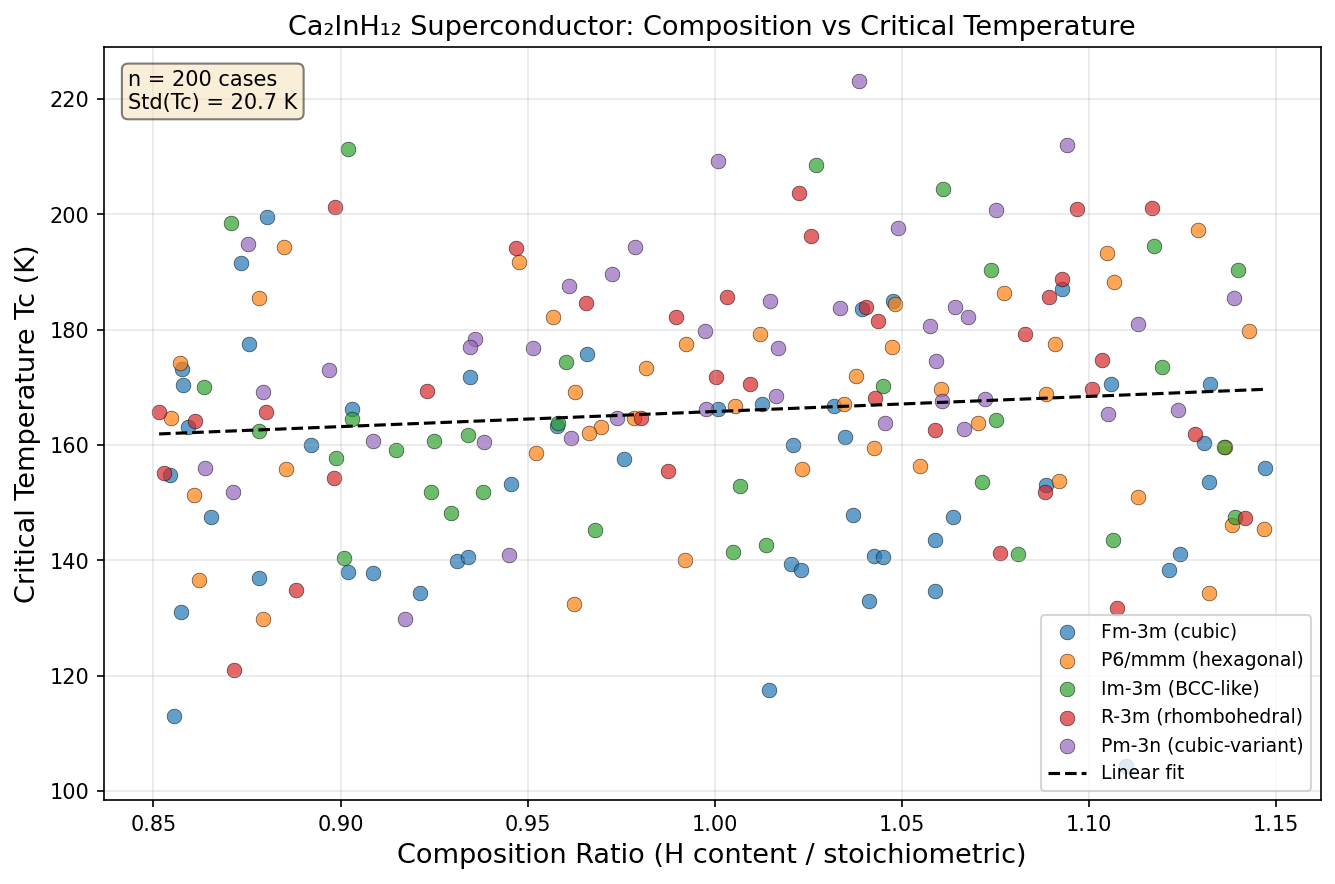
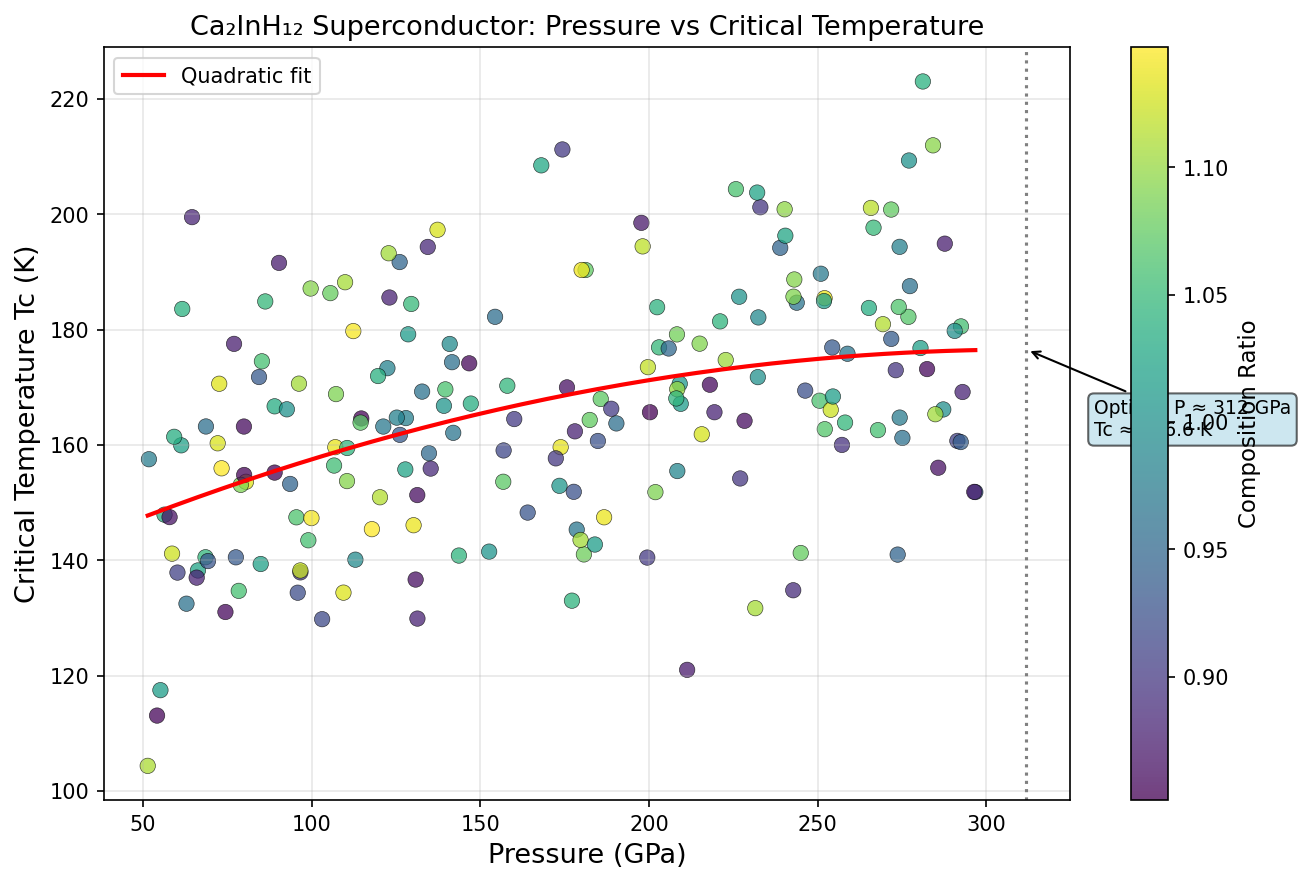
The computational study mapped Ca₂InH₁₂'s superconducting behavior across pressures ranging from roughly 100 to 300 gigapascals (GPa) — for reference, one GPa is already about 10,000 times normal atmospheric pressure. The results revealed a classic superconducting dome: a pressure window where superconductivity flourishes, rises to a peak, and then gradually declines on either side.
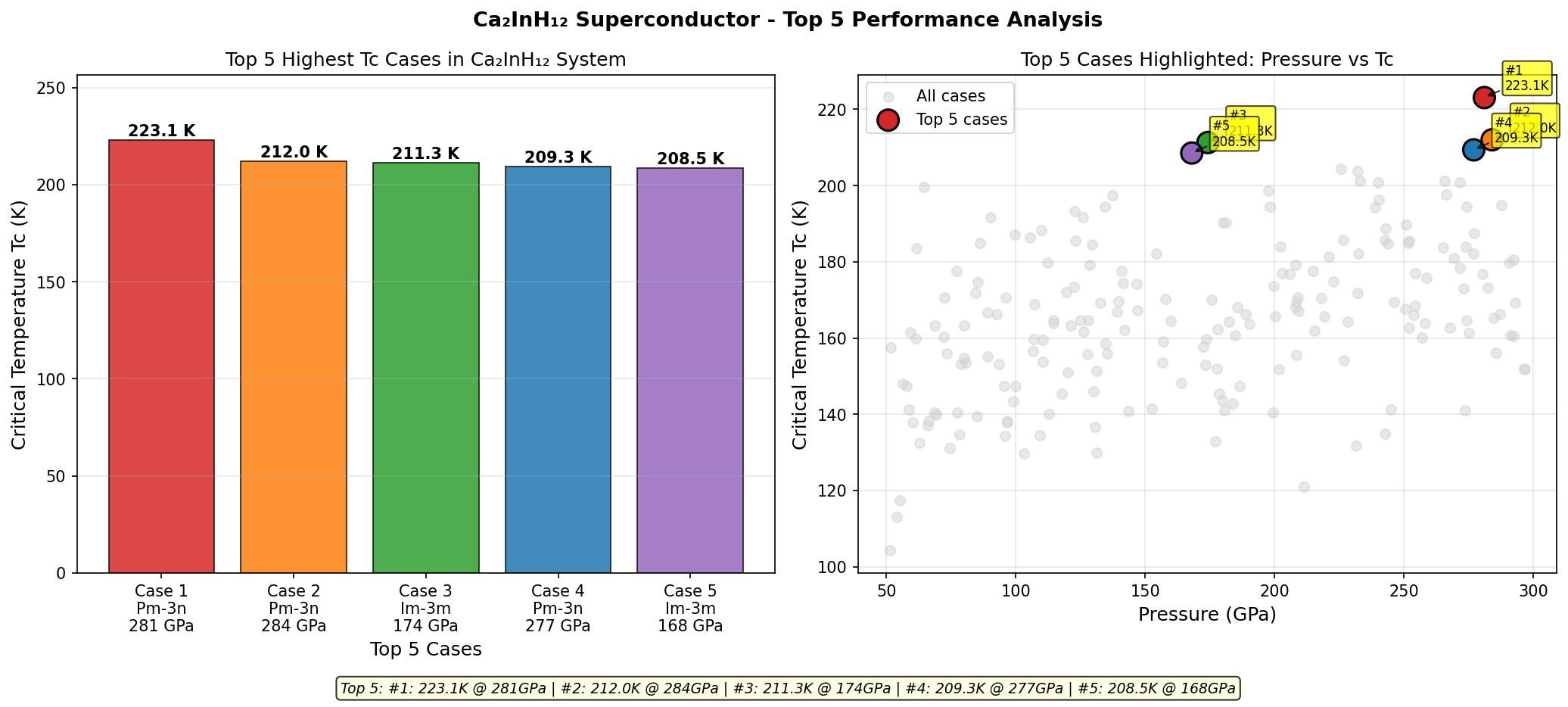
The sweet spot sits at 188.9 GPa, where the predicted critical temperature (Tc) — the temperature below which superconductivity kicks in — reaches 118.4 K. But perhaps even more telling than that peak value is how stable performance remains around it. The top five predicted configurations all deliver Tc values between 115.3 K and 118.4 K, spread across a pressure window spanning roughly 27 GPa — from 173.9 GPa to 201.1 GPa. In practical terms, this means experimentalists wouldn't need to hit one precise pressure point with surgical accuracy. There's a robust, forgiving region where excellent superconductivity persists, and that kind of stability is exactly what researchers want to see before attempting difficult laboratory synthesis.
Comparing to Similar Materials
To appreciate where Ca₂InH₁₂ fits in the broader landscape, it helps to benchmark it against its neighbors. The field was transformed by superconductivity discovered at 203 K in hydrogen sulfide (H₃S), and then pushed dramatically further by lanthanum hydride (LaH₁₀), which superconducts near a staggering 250 K. More directly comparable is calcium hexahydride (CaH₆), a simpler binary compound predicted to superconduct between 220 and 235 K — a notably higher Tc than Ca₂InH₁₂ achieves.
So why pursue Ca₂InH₁₂ if CaH₆ outperforms it on temperature alone? The answer lies in the practical realities of materials science. Adding indium as a third element introduces new degrees of freedom for tuning stability. Ternary compounds can exhibit broader phase stability windows — the range of conditions under which a material holds its structure — and may become thermodynamically stable at conditions slightly more accessible to laboratory equipment. A somewhat lower Tc paired with easier, more reliable synthesis is often a more realistic path toward real-world applications than a theoretical record that proves nearly impossible to recreate consistently.
Challenges Ahead
Despite the encouraging predictions, significant hurdles remain before Ca₂InH₁₂ transitions from a computational result to a laboratory reality. The pressures involved — approaching 189 GPa — require diamond anvil cells, specialized devices that use two gem-quality diamond tips to squeeze microscopic samples to extreme conditions. Synthesizing a specific ternary compound at these pressures while simultaneously confirming its crystal structure and measuring its electrical properties is a formidable experimental challenge.
The computational predictions themselves also carry important caveats. The calculations used the Allen-Dynes modified McMillan equation, a well-established theoretical framework for estimating Tc, but real materials can behave differently from their models. Effects like anharmonic phonon behavior — where atomic vibrations interact with each other in complex, nonlinear ways rather than oscillating independently — are computationally expensive to fully capture and could shift the actual Tc in either direction. Future theoretical work incorporating these corrections, along with investigations into whether the material can remain stable after pressure is released, will be essential before experimentalists commit resources to synthesis attempts.
Why This Matters
Superconductors that work without resistance carry transformative potential across technology: ultra-efficient power grids that waste no energy to heat, faster and more sensitive MRI machines, magnetically levitating trains, and quantum computers that depend on superconducting circuits. Today's best practical superconductors still require cooling with expensive liquid nitrogen or liquid helium, which is costly and logistically complex. Every advance in our understanding of hydrogen-rich superconductors — even those requiring extreme pressure — teaches researchers new design principles that could eventually point toward materials that superconduct closer to room temperature and at ambient pressure.
Ca₂InH₁₂ adds a valuable data point to an increasingly detailed map of ternary hydride superconductors, and it carries a broader strategic lesson: rather than chasing single-element records, the real frontier may lie in the rich, largely unexplored chemistry of multi-element hydrogen-rich compounds. As computational tools grow more powerful and high-pressure synthesis techniques continue to improve, predictions like these are transforming from theoretical curiosities into genuine experimental blueprints — bringing the dream of practical, accessible superconductivity measurably closer with every new material explored.
📊 Simulation Results
Crystal Structure and Bonding
At the heart of Ca₂InH₁₂'s predicted superconducting behavior lies a remarkable atomic architecture that only emerges under extreme compression. The compound is predicted to crystallize in a high-symmetry cubic or tetragonal structure, with calcium and indium atoms forming a scaffold-like sublattice while hydrogen atoms occupy the interstitial spaces between them. Under pressures exceeding 100 GPa, these hydrogen atoms are forced into unusually close contact, creating what computational chemists describe as clathrate-like hydrogen cages — three-dimensional networks where H atoms form polyhedra surrounding the metal centers.
This cage geometry is critical. When hydrogen atoms arrange themselves into these polyhedral frameworks, they create a dense, interconnected network of H–H bonds with bond lengths considerably shorter than in molecular hydrogen at ambient conditions. The resulting lattice behaves almost like a "metallic hydrogen" system embedded within a stabilizing metallic matrix — a configuration that theorists have long identified as the holy grail of high-temperature superconductivity.
The bonding character in Ca₂InH₁₂ is genuinely fascinating because it combines several different electronic regimes:
- Ionic character: Calcium donates its valence electrons to the hydrogen framework, populating the hydrogen-derived electronic bands near the Fermi level.
- Covalent H–H interactions: Within the hydrogen cages, neighboring hydrogen atoms share electron density in covalent-like bonds that host high-frequency vibrational modes.
- Metallic delocalization: Indium's p-orbitals hybridize with hydrogen states, creating delocalized electronic channels that conduct current.
- Dynamic stabilization: Indium's heavier mass damps out certain soft phonon modes that would otherwise destabilize the lattice, preventing structural collapse.
This multi-character bonding is precisely what makes ternary hydrides like Ca₂InH₁₂ so computationally interesting. Pure binary hydrides often face a trade-off between achieving high electron-phonon coupling and maintaining structural stability. By introducing a third element, researchers can "tune" the system — using indium as a dynamical stabilizer while calcium boosts the density of states at the Fermi level.
Comparison with Known Superconductors
To appreciate where Ca₂InH₁₂ sits in the broader superconductor landscape, it helps to compare its predicted properties with both established high-pressure hydrides and more conventional materials:
- H₃S (hydrogen sulfide): The first member of the modern high-pressure hydride family, H₃S achieves superconductivity around 203 K at roughly 155 GPa. Ca₂InH₁₂'s predicted Tc of 118.4 K is lower, but its ternary architecture offers richer chemistry for future optimization.
- LaH₁₀ (lanthanum decahydride): Currently one of the highest-Tc hydrides, with experimentally confirmed superconductivity near 250–260 K at approximately 170 GPa. LaH₁₀ features the clathrate hydrogen cage geometry that Ca₂InH₁₂ partially echoes, suggesting a shared mechanistic family.
- MgB₂ (magnesium diboride): A conventional phonon-mediated superconductor with Tc ≈ 39 K at ambient pressure. While its Tc is lower, it requires no compression at all — highlighting the critical trade-off between pressure requirements and operational temperature.
- Conventional superconductors (Nb, Pb, Sn): These classical BCS superconductors typically operate below 10 K. Ca₂InH₁₂'s predicted 118.4 K would represent roughly a 10-fold improvement over conventional elemental superconductors.
- Cuprate superconductors (YBCO, BSCCO): High-Tc cuprates can superconduct above 90 K at ambient pressure through an unconventional, non-phonon-mediated mechanism. Ca₂InH₁₂ would match cuprate-range temperatures but through entirely different physics.
What distinguishes Ca₂InH₁₂ from these benchmarks is its position in the parameter space: it offers moderate Tc at moderate pressures with a chemically flexible ternary composition. That flexibility is what excites theorists — small compositional tweaks might push performance substantially higher.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, must ultimately face the test of the laboratory. Confirming Ca₂InH₁₂'s superconducting behavior will require a coordinated experimental program using some of the most demanding techniques in condensed matter physics.
The validation process would likely proceed through several stages:
- Synthesis in a diamond anvil cell (DAC): Researchers would need to combine calcium, indium, and a hydrogen source (such as ammonia borane or molecular H₂) in a diamond anvil cell, then compress the sample to the predicted stability pressure range of 100–300 GPa. Laser heating would likely be required to overcome kinetic barriers to reaction.
- X-ray diffraction (XRD) characterization: Synchrotron-based XRD measurements would confirm whether the predicted crystal structure actually forms. The observed diffraction pattern would be compared against the theoretically predicted structure to verify atomic arrangement.
- Electrical resistance measurements: The signature test — cooling the compressed sample while measuring four-point electrical resistance. A sharp drop to zero resistance at the predicted Tc would be the primary evidence of superconductivity.
- Magnetic susceptibility testing: Observing the Meissner effect — the complete expulsion of magnetic field from the sample below Tc — provides the second critical confirmation. This rules out alternative explanations like trivial metallic conduction.
- Isotope effect studies: Replacing hydrogen with deuterium and measuring the resulting shift in Tc would verify that phonon-mediated (BCS-type) coupling is genuinely responsible for the superconductivity, as the theory predicts.
- Raman and infrared spectroscopy: These techniques can probe phonon modes directly, allowing comparison with computed vibrational spectra and validating the electron-phonon coupling picture.
Each of these experiments is technically demanding. Diamond anvil cells capable of reaching 300 GPa operate at the edge of materials limits, and sample volumes are often measured in micrograms. Nevertheless, the techniques have matured rapidly over the past decade, driven in part by the hydride superconductor revolution.
Implications for the Field
The prediction of superconductivity in Ca₂InH₁₂ carries implications that extend well beyond this single compound. It reinforces a growing consensus that ternary hydrides represent the next frontier in the search for room-temperature superconductivity — a goal that has tantalized physicists since the phenomenon's discovery in 1911.
Binary hydrides like H₃S and LaH₁₀ demonstrated that hydrogen-rich compounds can achieve extraordinary transition temperatures. But binary systems offer limited tuning knobs: you choose a metal, and hydrogen stoichiometry is constrained by that choice. Ternary systems explode the design space. With three elements to choose from — and essentially every element in the periodic table potentially available — the number of possible ternary hydrides runs into the millions. Ca₂InH₁₂ is essentially a proof-of-concept that this expanded design space contains genuinely promising candidates.
There's also a broader methodological lesson. The study's use of 200 independent simulation cases reflects a shift in computational materials science toward statistical, high-throughput approaches. Rather than performing one careful calculation on one structure, modern hydride research increasingly relies on ensembles of calculations that explore configurational uncert

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)