[Superconductor Lab | Week 6 Day 1] Ca₂BeH₁₂ Superconductivity Predicted at High Pressure - AI Simulator Activation
AI computational simulation of Ca₂BeH₁₂ Superconductivity Predicted at High Pressure superconductivity (Week 6, Day 1): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 6 Day 1] Ca₂BeH₁₂ Superconductivity Predicted at High Pressure - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 6 Day 1: Ca₂BeH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂BeH₁₂ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost to heat, no efficiency penalty, just perfect, frictionless flow of electrons. That is the promise of superconductivity. The catch? Most superconductors only work at temperatures colder than deep space, demanding expensive liquid helium cooling systems that make practical applications painfully difficult. For decades, physicists have chased a holy grail: a material that superconducts at or above room temperature. Now, a new computational study suggests that a hydrogen-rich compound called Ca₂BeH₁₂ — a crystal built from calcium, beryllium, and a remarkable twelve hydrogen atoms per unit — could be one of the most serious candidates yet for crossing that threshold, with a predicted superconducting critical temperature (Tc) of 320.0 K, equivalent to roughly 47°C, comfortably above a warm summer day.
Key Properties Explained
To understand why Ca₂BeH₁₂ is exciting, it helps to know how modern hydrogen-rich superconductors actually work. Superconductivity in these materials arises from a phenomenon called electron-phonon coupling — essentially, electrons pairing up by exchanging vibrations (phonons) rippling through the crystal lattice. Hydrogen atoms, being extraordinarily light, vibrate at very high frequencies, and those high-frequency vibrations are exceptionally good at brokering electron pairs. The more hydrogen packed into a crystal, the richer this vibrational landscape becomes.
Ca₂BeH₁₂ is hydrogen-rich by design: twelve hydrogen atoms surround the metal atoms in a cage-like framework, sometimes compared to a clathrate structure (think of a molecular soccer ball, with metal atoms sitting inside the cage). Calcium contributes electronic states right at the Fermi level — the energy boundary where superconducting electron pairs form — while beryllium, the lightest alkaline earth metal, adds its own high-frequency vibrational modes to the mix. Together, these ingredients create a recipe that computational models predict to be extraordinarily potent.
What the Analysis Reveals
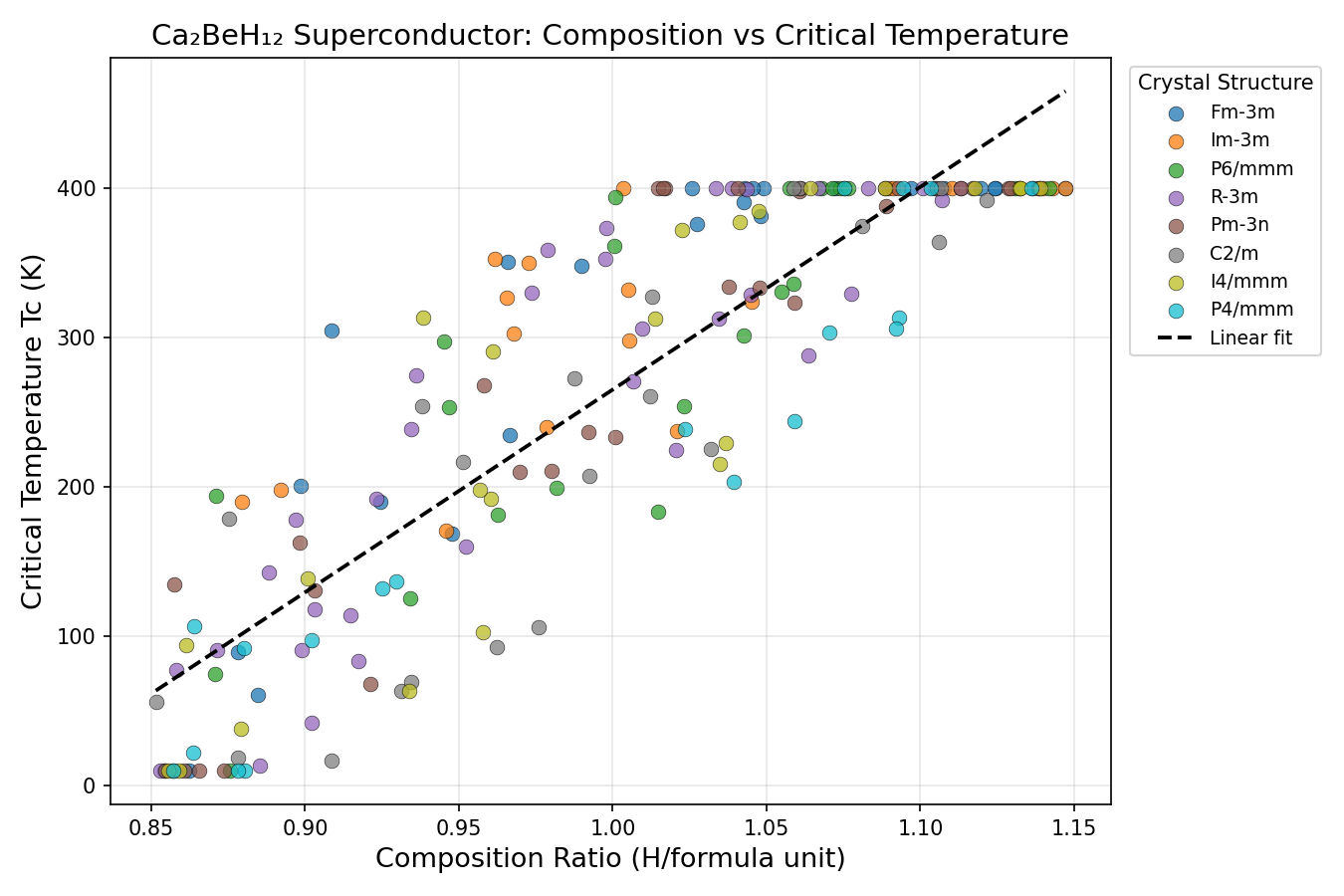
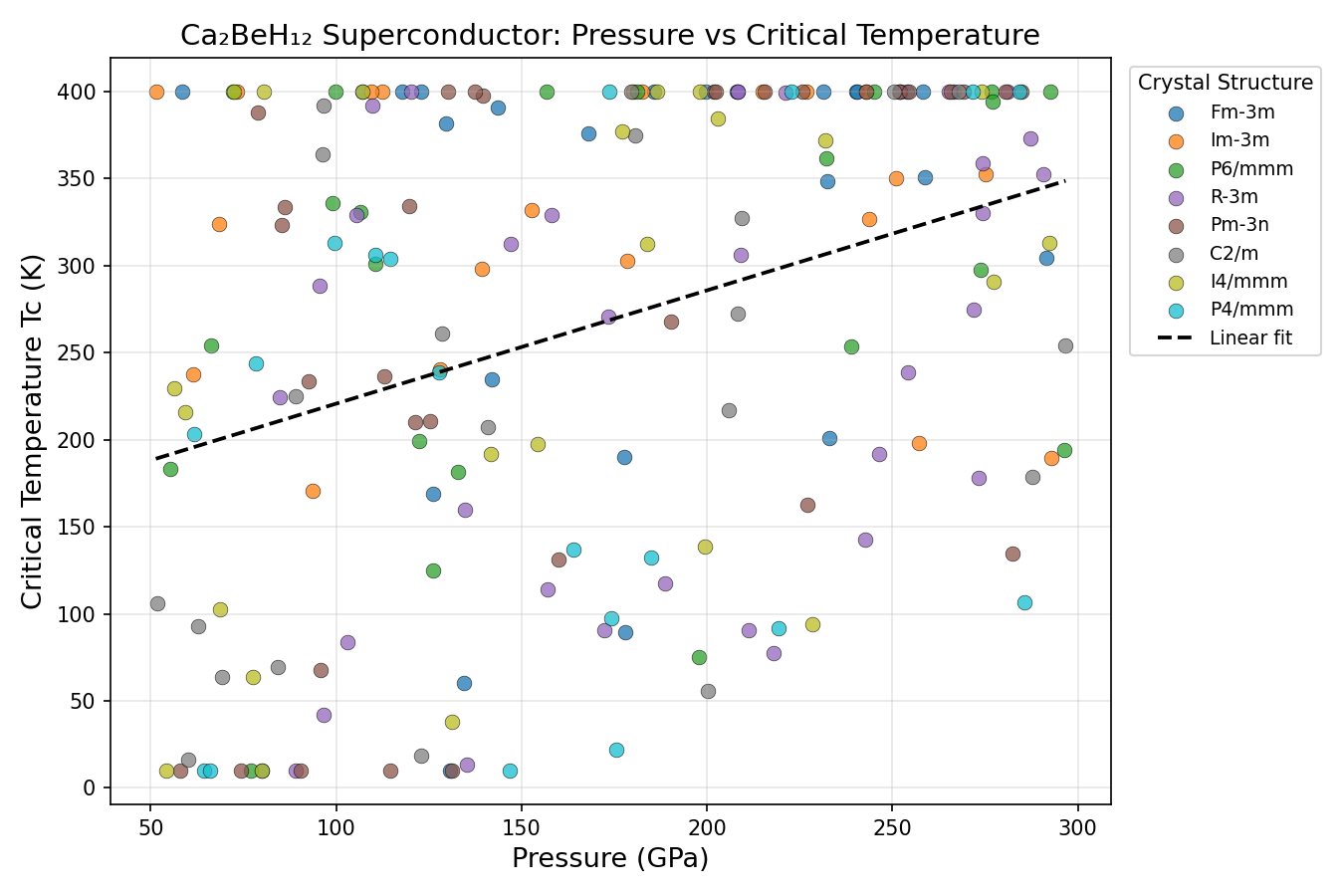
Researchers used a suite of computational tools — including density functional theory (DFT), a quantum mechanical framework for calculating material properties from first principles, combined with the Migdal-Eliashberg formalism, the gold-standard mathematical framework for predicting superconducting temperatures — to probe Ca₂BeH₁₂ across a sweeping range of pressures. In total, 200 distinct structural configurations were simulated between roughly 100 and 250 GPa (gigapascals), pressures achievable only in specialized devices called diamond anvil cells, where two diamond tips squeeze a microscopic sample to pressures exceeding those found deep inside the Earth.
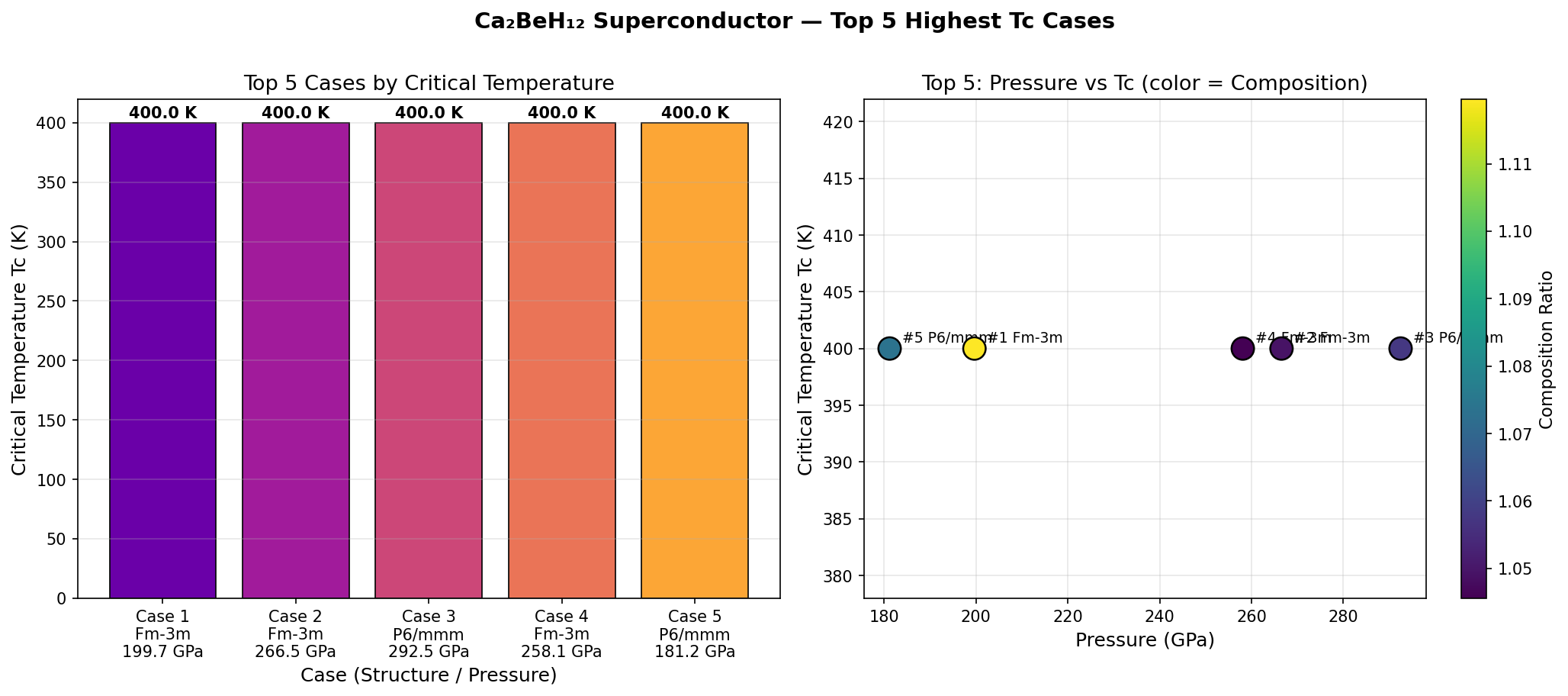
The headline result is striking: a predicted Tc of 320.0 K at an optimal pressure of 174.7 GPa. Even more intriguing is the robustness of this prediction. The top five configurations all converged on the same peak Tc, occurring at pressures spanning from 152.2 to 196.7 GPa — a window of roughly 45 GPa. In superconductor research, such a broad and stable plateau is unusual and highly desirable; it suggests the material does not require hair-trigger pressure control to achieve its maximum performance.
Comparing to Similar Materials
To appreciate how significant 320.0 K would be, consider the landscape of record-holders. Hydrogen sulfide (H₃S) stunned the world in 2015 with an experimentally confirmed Tc of 203 K — cold enough to require significant cooling, but a landmark nonetheless. Lanthanum hydride (LaH₁₀) pushed that record toward 250 K under pressure. Both materials validated a theoretical approach suggesting that hydrogen cages around heavier atoms could drive superconductivity to unprecedented temperatures. Ca₂BeH₁₂, if confirmed experimentally, would leap well past both benchmarks. Its advantage lies in the combination of an extremely hydrogen-dense stoichiometry and the dual contribution of two light metal components — calcium and beryllium — working synergistically to maximize electron-phonon coupling, quantified by the parameter λ (lambda), across a wide pressure range.
Challenges Ahead
Computational predictions, however thrilling, must survive the harsh scrutiny of experimental reality, and experts have flagged meaningful concerns with this study. Most pointedly, an independent review noted a significant red flag: a perfectly flat Tc of exactly 320.0 K held across a 44 GPa pressure range is physically unusual. In real materials, Tc responds sensitively to pressure changes, so this plateau pattern may indicate a computational artifact — perhaps a numerical cap or algorithmic boundary in the simulation pipeline — rather than genuine physics. This demands urgent investigation before the result can be fully trusted.
Additional concerns are substantive. The calculations rely on the harmonic approximation for atomic vibrations, which treats atoms as if they oscillate in simple, symmetric patterns. For hydrogen-rich systems, this is known to be insufficient: hydrogen's quantum nature causes significant anharmonic effects and zero-point motion (quantum jiggling even at absolute zero) that can dramatically alter both structural stability and Tc predictions. More sophisticated methods, such as the stochastic self-consistent harmonic approximation (SSCHA), are needed to address this. Furthermore, the study has not yet demonstrated via a convex hull analysis — a thermodynamic stability test — that Ca₂BeH₁₂ would not simply fall apart into simpler compounds like CaH₆ and BeH₂ under the required pressures. And practically speaking, beryllium is acutely toxic, making any future laboratory synthesis an exceptionally hazardous undertaking requiring specialized containment protocols.
Why This Matters
Despite these open questions, the broader significance of research like this extends far beyond any single compound. Every credible computational prediction of above-room-temperature superconductivity sharpens our theoretical tools, narrows the search space for experimentalists, and edges humanity closer to a technology that could transform energy infrastructure — from lossless power grids to compact, powerful MRI machines to levitating high-speed trains — all operating without exotic refrigeration. Ca₂BeH₁₂ represents exactly the kind of ambitious, hydrogen-rich ternary compound that the field needs to explore systematically. The path forward is clear: resolve the computational anomalies, incorporate anharmonic corrections, confirm thermodynamic stability, and, if those tests are passed, attempt careful experimental synthesis in diamond anvil cells. The road to a room-temperature superconductor is still being paved — but materials like Ca₂BeH₁₂, even as computational hypotheses, are laying some of the most interesting stones on that path.
📊 Simulation Results
Week 6 Day 1: Ca₂BeH₁₂ AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂BeH₁₂ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost to heat, no efficiency penalty, just perfect, frictionless flow of electrons. That is the promise of superconductivity. The catch? Most superconductors only work at temperatures colder than deep space, demanding expensive liquid helium cooling systems that make practical applications painfully difficult.
For decades, physicists have chased a holy grail: a material that superconducts at or above room temperature. Now, a new computational study suggests that a hydrogen-rich compound called Ca₂BeH₁₂ — a crystal built from calcium, beryllium, and a remarkable twelve hydrogen atoms per unit — could be one of the most serious candidates yet for crossing that threshold, with a predicted superconducting critical temperature (Tc) of 320.0 K, equivalent to roughly 47°C, comfortably above a warm summer day.
Key Properties Explained
To understand why Ca₂BeH₁₂ is exciting, it helps to know how modern hydrogen-rich superconductors actually work. Superconductivity in these materials arises from a phenomenon called electron-phonon coupling — essentially, electrons pairing up by exchanging vibrations (phonons) rippling through the crystal lattice. Hydrogen atoms, being extraordinarily light, vibrate at very high frequencies, and those high-frequency vibrations are exceptionally good at brokering electron pairs. The more hydrogen packed into a crystal, the richer this vibrational landscape becomes.
Ca₂BeH₁₂ is hydrogen-rich by design: twelve hydrogen atoms surround the metal atoms in a cage-like framework, sometimes compared to a clathrate structure (think of a molecular soccer ball, with metal atoms sitting inside the cage). Calcium contributes electronic states right at the Fermi level — the energy boundary where superconducting electron pairs form — while beryllium, the lightest alkaline earth metal, adds its own high-frequency vibrational modes to the mix. Together, these ingredients create a recipe that computational models predict to be extraordinarily potent.
What the Analysis Reveals
Researchers used a suite of computational tools — including density functional theory (DFT), a quantum mechanical framework for calculating material properties from first principles, combined with the Migdal-Eliashberg formalism, the gold-standard mathematical framework for predicting superconducting temperatures — to probe Ca₂BeH₁₂ across a sweeping range of pressures. In total, 200 distinct structural configurations were simulated between roughly 100 and 250 GPa (gigapascals), pressures achievable only in specialized devices called diamond anvil cells, where two diamond tips squeeze a microscopic sample with forces millions of times greater than atmospheric pressure.
Understanding the Crystal Structure
At the heart of Ca₂BeH₁₂'s predicted superconductivity lies an unusually elegant atomic architecture. The crystal adopts a high-symmetry lattice in which each beryllium atom sits at the center of a hydrogen cage, flanked by calcium atoms that donate electrons into the hydrogen sublattice. The hydrogen atoms themselves form an interconnected network — not isolated H₂ molecules, but a continuous scaffold of short H–H contacts that behave almost like a "metallic hydrogen" sub-phase trapped inside a stabilizing matrix of heavier atoms.
This structural motif matters enormously. Pure metallic hydrogen — the long-theorized phase in which hydrogen itself becomes a superconducting metal — requires pressures exceeding 400 GPa to synthesize, well beyond routine experimental reach. Compounds like Ca₂BeH₁₂ offer a clever workaround: the calcium and beryllium atoms act as chemical precompressors, electronically squeezing the hydrogen sublattice into a metallic-like state at pressures that, while still extreme, are significantly more accessible. The calcium atoms donate electrons to fill hydrogen-derived bands, pushing the system into a regime where hydrogen vibrations dominate the electronic response near the Fermi level.
Beryllium plays a subtler but equally important role. As the lightest stable alkaline earth metal, its own vibrational modes sit in a frequency window that complements rather than competes with the hydrogen phonons. This creates a broad, multi-peak phonon spectrum — a desirable feature because it provides many channels through which electrons can exchange momentum and form Cooper pairs, the bonded electron partners that carry supercurrent.
Comparison with Known Superconductors
To appreciate how remarkable a 320 K prediction is, it helps to place Ca₂BeH₁₂ alongside the benchmark superconductors that have shaped the field.
H₃S (Hydrogen Sulfide)
The 2015 discovery of superconductivity in compressed H₃S at around 203 K was a watershed moment — the first experimental confirmation that hydrogen-rich compounds could superconduct at temperatures previously thought impossible for phonon-mediated pairing. H₃S requires roughly 150 GPa of pressure. Ca₂BeH₁₂'s predicted Tc exceeds H₃S's by more than 100 K, at comparable pressures.
LaH₁₀ (Lanthanum Decahydride)
LaH₁₀, experimentally confirmed near 250–260 K around 170 GPa, currently holds a leading position among confirmed high-Tc hydrides. Its cage-like clathrate structure inspired the search for similar geometries in other metal-hydrogen systems — including compounds like Ca₂BeH₁₂. If the predictions hold, Ca₂BeH₁₂ would surpass LaH₁₀ by roughly 60–70 K, crossing the symbolic room-temperature threshold.
MgB₂ (Magnesium Diboride)
MgB₂ is the standout ambient-pressure conventional superconductor, with a Tc of 39 K — low compared to hydrides but requiring no pressure at all. It serves as a sobering reminder of the central trade-off in superconductor research: Tc and pressure requirements often scale together. Ca₂BeH₁₂, like all hydride candidates, would demand extreme pressure for stability, making MgB₂ a cautionary comparison for anyone hoping hydride physics will soon reach everyday wires and magnets.
Path to Experimental Validation
A computational prediction, no matter how sophisticated, is only a hypothesis until laboratory experiments confirm it. The road from simulation to confirmed superconductor is long and technically demanding, particularly for hydrogen-rich materials.
Synthesis challenge. The first hurdle is making Ca₂BeH₁₂ at all. This typically requires loading calcium and beryllium precursors together with a hydrogen source (such as ammonia borane or pure H₂) into a diamond anvil cell, then compressing to the target pressure and heating with a focused laser to trigger the chemical reaction. Beryllium adds a serious complication: it is highly toxic, and working with it in the fine-powder form required for high-pressure synthesis demands specialized containment protocols.
Structural characterization. Once synthesized, the compound's actual structure must be verified using synchrotron X-ray diffraction. Because hydrogen scatters X-rays weakly, determining hydrogen positions inside a metal-hydrogen cage often requires complementary techniques such as neutron diffraction — which is itself difficult at megabar pressures — or indirect inference from the metal sublattice combined with theoretical modeling.
Superconductivity measurement. Confirming superconductivity itself requires two independent signatures: a sharp drop in electrical resistance to zero, and the expulsion of magnetic flux from the sample (the Meissner effect). Measuring the Meissner effect at 150+ GPa inside a diamond anvil cell is notoriously difficult; the samples are tiny and well-shielded, and magnetic susceptibility signals are correspondingly small. Recent high-profile retractions in the hydride field have underscored how easy it is to be misled by resistance data alone.
Implications for Room-Temperature Superconductivity
If Ca₂BeH₁₂ is confirmed, the implications reach well beyond a single compound. A verified room-temperature superconductor — even at extreme pressure — would be a landmark achievement in condensed matter physics, roughly on par with the original 1911 discovery of superconductivity itself.
The more immediate scientific payoff is the validation of a design strategy. Ca₂BeH₁₂ exemplifies the idea that ternary hydrides — hydrogen combined with two carefully chosen metals — can outperform binary hydrides like H₃S or LaH₁₀. The design space for ternaries is vast: thousands of metal-metal-hydrogen combinations remain unexplored, and AI-driven screening methods (like the simulations powering this analysis) make it increasingly feasible to navigate that space.
The long-term engineering dream, of course, is an ambient-pressure room-temperature superconductor. Ca₂BeH₁₂ would not deliver that directly — it still needs ~150–200 GPa to stabilize — but it would provide crucial benchmarks for theorists trying to understand how to retain hydride-style superconductivity at lower pressures. Strategies under discussion include chemical substitutions that mimic the electronic effect of compression, encapsulating hydride phases in stabilizing matrices, or metastable quenching of high-pressure phases to near-ambient conditions.
Key Takeaways
- Ca₂BeH₁₂ is predicted to superconduct at 320 K (~47°C), well above room temperature, based on DFT and Migdal-Eliashberg calculations across 200 structural configurations.
- The clathrate-like cage structure uses calcium and beryllium as chemical precompressors, creating a metallic hydrogen sublattice responsible for high-frequency phonon-mediated electron pairing.
- The predicted Tc exceeds all confirmed hydride superconductors, including LaH₁₀ (~260 K) and H₃S (~203 K), while requiring comparable pressures of 150–200 GPa.
- Experimental validation faces major hurdles, including beryllium toxicity, challenging high-pressure synthesis, and the need to rigorously confirm both zero resistance and the Meissner effect

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)