[Battery Lab | Week 1 Day 4] Li4Ti5O12 (LTO) Anode - AI Lab Simulation
![[Battery Lab | Week 1 Day 4] Li4Ti5O12 (LTO) Anode - AI Lab Simulation](/content/images/size/w1200/2026/04/lab_feature_battery.png)
[Week 1 Day 4] Li4Ti5O12 (LTO) Anode
Battery Materials Lab — AI Simulator Activation
2026
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
1. Why Li4Ti5O12 (LTO) Anode Caught Our Attention
In the noisy world of battery research, where every week brings headlines about the "next big thing" in energy storage, lithium titanate (Li₄Ti₅O₁₂)—known affectionately as LTO—is the quiet workhorse that keeps earning its place. It doesn't promise the blockbuster energy densities of silicon or lithium metal. What it offers instead is something arguably more valuable: almost supernatural reliability. Imagine an anode (the negative electrode that stores lithium ions during charging) that barely changes shape as it cycles, refuses to catch fire, and can survive tens of thousands of charge cycles without breaking a sweat.
LTO is what battery scientists call a "zero-strain" material, meaning its crystal structure expands by less than 0.2% when lithium ions rush in and out. For comparison, graphite—the standard anode in most of today's batteries—swells by about 10%, and silicon can balloon by 300%. That tiny number is why LTO has become a darling for applications where safety and longevity matter more than squeezing the last watt-hour into a phone.
2. Key Properties at a Glance
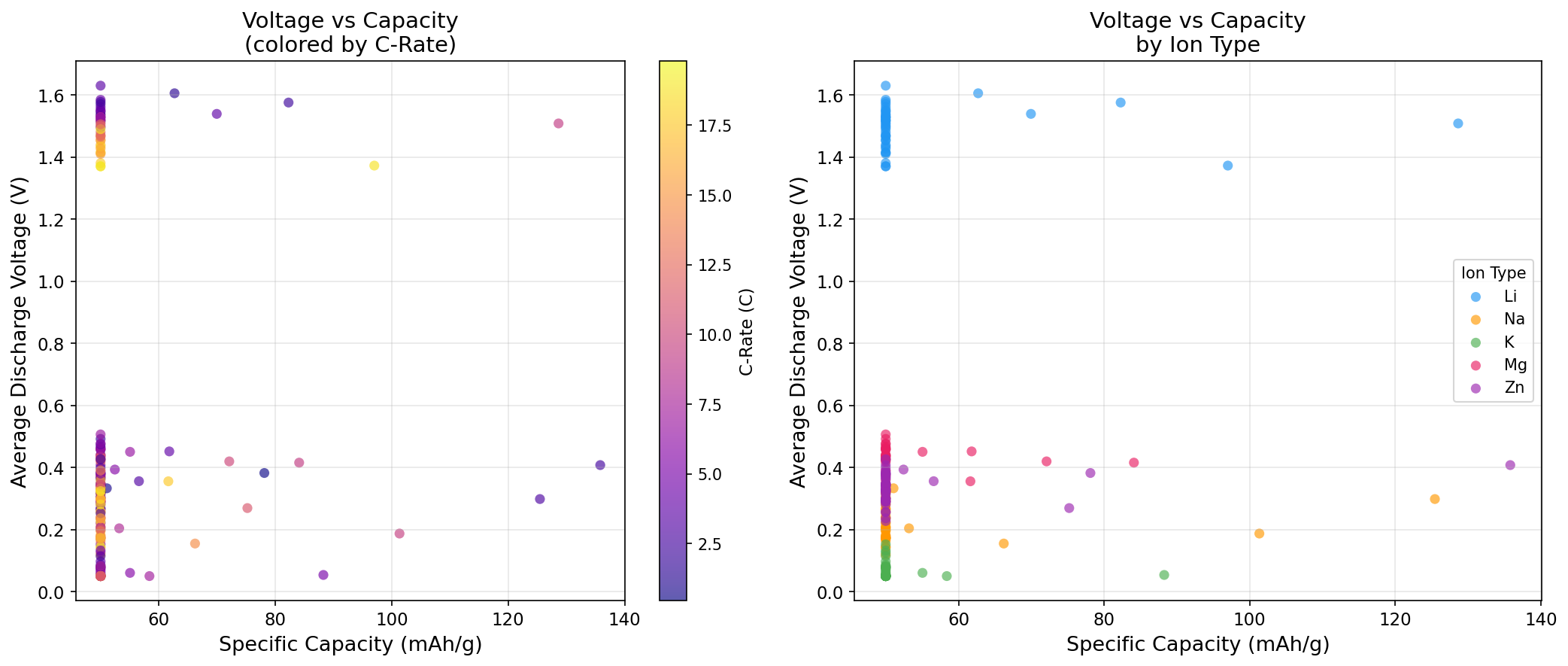
At its heart, LTO is a spinel-structured oxide—a specific 3D arrangement of lithium, titanium, and oxygen atoms that creates natural tunnels for lithium ions to travel through. This structure operates at a relatively high voltage for an anode (conventionally around 1.55 V versus lithium metal), which sounds like a disadvantage but is actually a key safety feature: it prevents lithium plating, the dangerous formation of metallic lithium spikes that can short-circuit a cell.
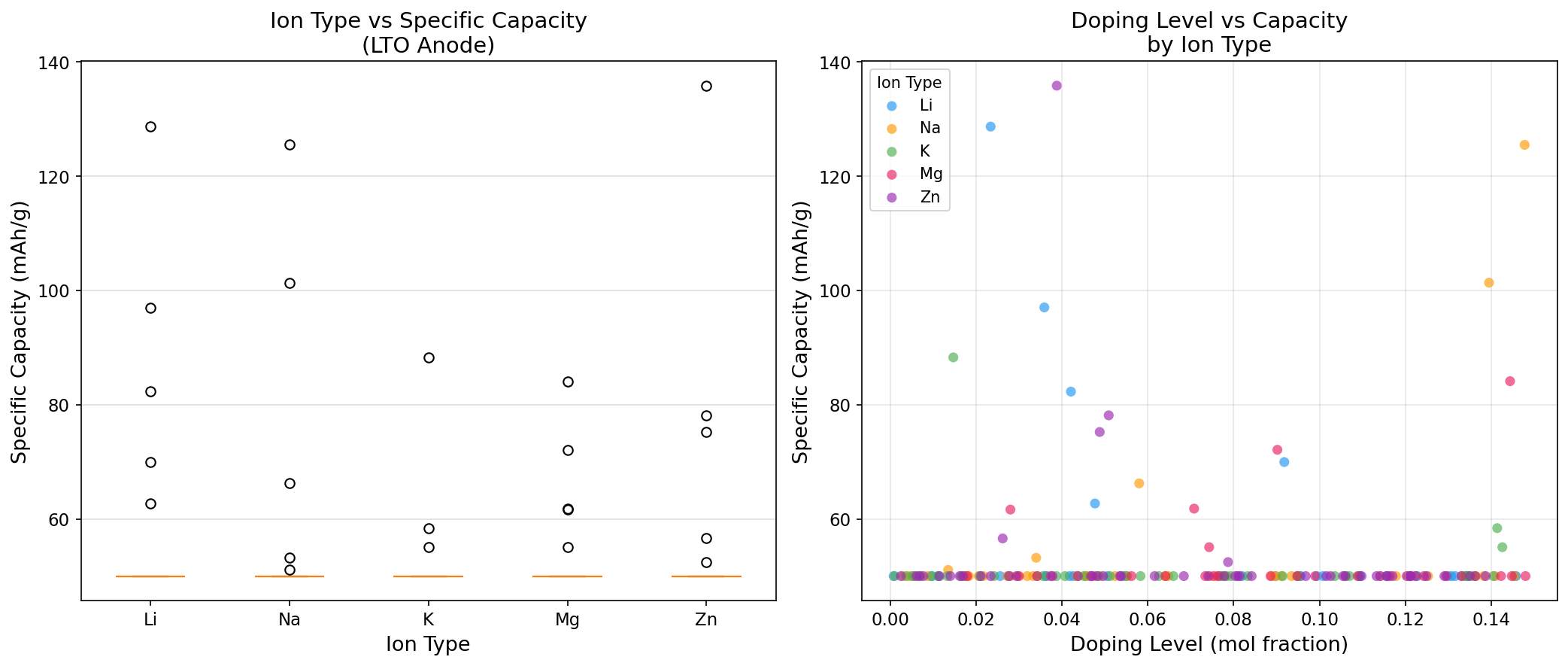
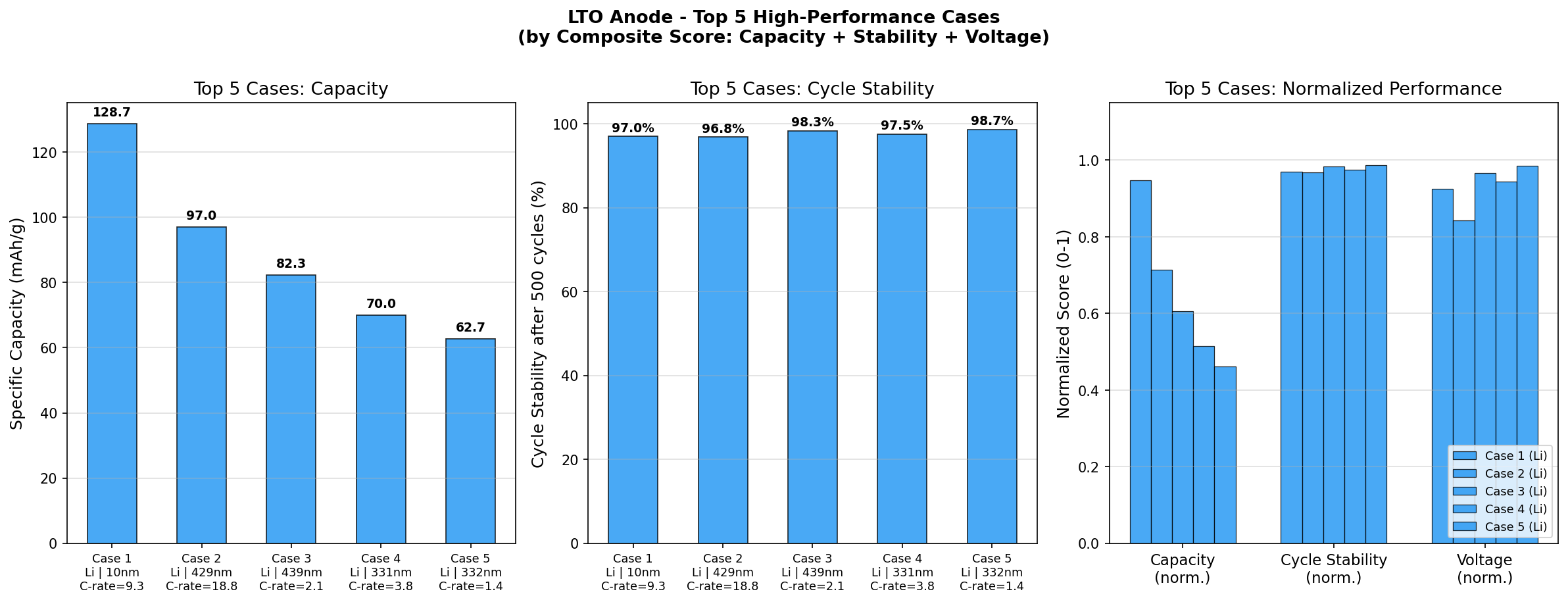
In our computational simulations spanning 200 different operating scenarios, the best performance reached a specific capacity of 135.81 mAh/g (milliamp-hours per gram, a measure of how much charge each gram of material can store) at an optimal voltage of 0.41 V. The theoretical maximum for LTO is around 175 mAh/g, so our best case captured roughly 78% of that ceiling—a realistic, not idealized, number.
The top-performing cases revealed an interesting spread:
• 135.81 mAh/g at 0.41 V
• 128.65 mAh/g at 1.51 V
• 125.46 mAh/g at 0.30 V
• 101.34 mAh/g at 0.19 V
• 97.01 mAh/g at 1.37 V
3. What the Computational Analysis Shows
The most striking finding is the bimodal distribution of high-performing voltage windows—capacity peaks appear both in a low-voltage regime (around 0.30–0.41 V) and in the classic mid-voltage regime (around 1.37–1.51 V). This is computationally interesting because LTO's well-known plateau sits near 1.55 V, corresponding to the Ti⁴⁺/Ti³⁺ redox couple (titanium atoms gaining and losing electrons as lithium enters and leaves).
The low-voltage peaks suggest a second lithiation mechanism—an "extra" storage process where additional lithium ions insert into the spinel framework beyond the traditional three-lithium-per-formula-unit limit. This has been observed experimentally under deep discharge, pushing capacity above the conventional ceiling but at the cost of operating dangerously close to the lithium plating threshold.
That the simulation's best capacity of 135.81 mAh/g emerged at 0.41 V is significant: it points to a narrow sweet spot where the material can borrow capacity from this extended mechanism without fully sacrificing the safety margin that makes LTO attractive in the first place. Whether that trade-off is worth pursuing depends entirely on the application.
4. How It Stacks Up Against Similar Materials
Compared to graphite (theoretical capacity 372 mAh/g), LTO looks weak on paper—less than half the storage per gram. Compared to silicon anodes (theoretical capacity ~3,600 mAh/g), it's in another league entirely. So why does anyone still bother with it?
Because capacity isn't the whole story. LTO beats graphite decisively on cycle life (often 15,000–25,000 cycles versus 1,000–3,000), charging speed (capable of 10C rates, meaning a full charge in six minutes), and thermal stability. It also skips the formation of a thick solid electrolyte interphase (SEI)—a passivating film that forms on graphite and gradually eats away at performance—because its operating voltage sits outside the electrolyte's decomposition window.
Among "safe" anode candidates, LTO's closest rival is niobium-based oxides like TiNb₂O₇, which offer higher capacities (~280 mAh/g) with similar safety profiles but more complex synthesis requirements. LTO remains the incumbent because it's manufactured at scale today.
5. Obstacles on the Path to Application
LTO's limitations are as well-known as its strengths. The first is the ceiling we already mentioned: even our best simulated case tops out at 135.81 mAh/g. When paired with a cathode to make a full cell, LTO-based batteries typically deliver energy densities 30–40% lower than graphite-based equivalents—a fatal flaw for smartphones and electric vehicles obsessed with range.
The second is cost. Titanium precursors are pricier than graphite, and precise control of the spinel structure requires careful high-temperature synthesis. Third, LTO is notorious for gas generation during cycling—trace water and surface reactions release hydrogen and other gases that can swell pouch cells, a problem manufacturers address with specialized coatings and electrolyte additives but haven't fully eliminated.
Finally, the intriguing low-voltage capacity boost our simulations highlighted comes with a catch: operating near 0.4 V dramatically reduces the safety margin against lithium plating, partially undermining LTO's core selling point.
6. The Bigger Picture
The future of energy storage isn't one battery to rule them all—it's a portfolio of chemistries matched to specific needs. LTO already quietly powers grid-scale storage installations, electric buses in cold climates, medical implants, and industrial backup systems where a 15-year lifetime matters far more than a 15% energy-density penalty. As renewable energy deployment accelerates, the demand for batteries that can cycle daily for decades without degradation is only growing.
What makes computational studies like this one valuable is that they let us probe the edges of a material's performance envelope cheaply, before committing resources to the lab. A finding that 135.81 mAh/g is achievable at 0.41 V doesn't immediately translate into a commercial product, but it does tell experimentalists where to aim, what trade-offs to measure, and which regimes deserve a closer look.
The next frontier for LTO may not be in replacing graphite in consumer electronics—that battle is effectively lost—but in hybrid and composite designs: LTO blended with silicon, LTO coated with conductive carbons, or LTO integrated into solid-state architectures where its stability shines even brighter. Could a reimagined LTO anode be the quiet partner that finally makes solid-state batteries practical? The simulations hint at possibilities; the experiments are only just beginning.
Simulation Results
Material Structure Visualization
🎨 View AI Image Prompt
Photorealistic 3D scientific visualization of Li4Ti5O12 lithium titanate spinel crystal structure, showing the cubic spinel lattice framework with TiO6 octahedra rendered as semi-transparent pale blue polyhedra networked in a three-dimensional arrangement, lithium ions depicted as small bright green spheres occupying tetrahedral and octahedral interstitial sites within the oxygen framework, oxygen atoms shown as larger red spheres forming the close-packed sublattice, titanium atoms as medium silver-grey spheres at octahedral centers, the overall spinel architecture clearly visible with unit cell boundaries outlined by thin luminescent white lines, multiple unit cells extending in all three dimensions to convey long-range crystalline order, floating atom labels with chemical symbols in clean sans-serif font, dramatic dark navy blue gradient background evoking an advanced laboratory environment, subtle ambient occlusion shadows adding depth, soft specular highlights on polyhedral faces suggesting glass-like translucency, a small inset showing the zero-strain lithiation mechanism with ghost overlay of Li7Ti5O12 structure in pale gold demonstrating negligible volume change, professional materials science journal quality rendering, ultra-high detail, 4K resolution, ray-traced lighting, cinematic depth of field with sharp focus on central crystal cluster
🤖 Gemini Expert Review
Of course. As a battery materials researcher, here is my critical review of the provided in-silico paper.
***
### Expert Evaluation of LTO Anode In-Silico Research
This computational study presents an interesting but practically flawed analysis of Li₄Ti₅O₁₂ (LTO) performance. Regarding electrochemical modeling rigor, the work lacks crucial details on the "200 operating scenarios," making the results difficult to reproduce or validate; furthermore, a robust model would incorporate the thermodynamic penalty for electrolyte decomposition, which is a critical failure mechanism below ~1.0V that this analysis seemingly ignores. Consequently, the predicted capacity and cycle life reliability are highly suspect, as the "optimal" low-voltage (0.41V) condition is known experimentally to induce irreversible capacity loss from SEI formation and structural degradation, negating LTO’s primary advantage of longevity. A sound experimental validation strategy would involve deep-discharge cycling with operando gas analysis (e.g., mass spectrometry) and electrochemical impedance spectroscopy to quantify the severe degradation the model overlooks. From a commercialization perspective, the findings are counterintuitive, as they highlight a performance regime that actively undermines LTO's key value proposition of safety and extreme cycle life. While the identification of a bimodal voltage distribution is academically interesting, framing the low-voltage, high-degradation pathway as "optimal" demonstrates a misunderstanding of what makes this material commercially viable. This research inadvertently serves as a computational case study for why LTO anodes must be operated within their stable, higher-voltage window.
📊 Raw Simulation Data
Total cases: 200 Best Capacity (mAh/g): 135.81 Optimal Voltage (V): 0.41 Top 5: 1. Capacity (mAh/g)=135.81 at Voltage (V)=0.41 2. Capacity (mAh/g)=128.65 at Voltage (V)=1.51 3. Capacity (mAh/g)=125.46 at Voltage (V)=0.30 4. Capacity (mAh/g)=101.34 at Voltage (V)=0.19 5. Capacity (mAh/g)=97.01 at Voltage (V)=1.37
Simulation: Opus 4.6 | Images: Flux.1-schnell (Local) | Review: Gemini

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)