[Battery Lab | Week 1 Day 1] LiFePO4 (LFP) Cathode - AI Lab Simulation
![[Battery Lab | Week 1 Day 1] LiFePO4 (LFP) Cathode - AI Lab Simulation](/content/images/size/w1200/2026/04/lab_feature_battery.png)
Week 1 Day 1: LiFePO4
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why LiFePO4 Stands Out
Imagine a battery material that is cheap, safe, environmentally friendly, and remarkably stable — even when things get hot. That is the promise of lithium iron phosphate, better known by its chemical shorthand LiFePO4, or simply LFP. First described by researchers in 1997, this olivine-structured mineral (named for its resemblance to the green gemstone olivine) has quietly become one of the most compelling cathode materials in the lithium-ion battery world. While flashier materials grab headlines, LFP has been steadily powering electric buses, grid-scale energy storage systems, and increasingly, electric vehicles. A new computational study analyzing 200 simulated battery configurations helps explain why — and reveals exactly where the sweet spot for peak performance lies.
Key Properties Explained
To understand what makes LFP special, it helps to know what a cathode material actually does. In a lithium-ion battery, the cathode is the positive electrode — the place where lithium ions arrive during discharge, releasing the electrical energy you use to power a device. The cathode material's chemistry dictates how much energy can be stored, how safely, and for how long.
LFP offers a theoretical specific capacity — the maximum charge it could store per gram of material — of 170 mAh/g (milliampere-hours per gram). It operates around a discharge voltage plateau of approximately 3.4 volts versus a lithium reference, meaning it delivers power in a remarkably flat, steady curve rather than a steep drop-off. This flatness is a practical advantage: devices powered by LFP batteries receive consistent voltage right up until the battery is nearly depleted.
Perhaps most importantly, LFP's thermal stability is exceptional. Unlike some competing cathode chemistries, LFP does not readily release oxygen when overheated, making it far less prone to the dangerous thermal runaway events that have made battery safety a headline issue. Add in the fact that iron and phosphate are abundant, inexpensive, and non-toxic raw materials, and you have a chemistry that checks nearly every box for sustainable, large-scale energy storage.
What the Analysis Reveals
The new computational study put LFP through its paces across 200 simulated configurations, systematically varying factors like particle size (how finely the material is ground), carbon coating thickness (a thin layer applied to boost electrical conductivity), electrolyte concentration, temperature, and current density (how fast charge flows). The goal was to map the performance landscape and find the combinations that unlock the best capacity.
The standout result: the best-performing configuration achieved a specific capacity of 142.99 mAh/g at an optimal voltage of 3.48 volts. That represents approximately 84% of LFP's theoretical maximum — an impressive figure for a computational model, and one that aligns well with real-world, well-optimized laboratory results. Crucially, the top voltage window identified — between 3.35 V and 3.52 V — matches precisely with the known electrochemical fingerprint of the iron-to-iron redox reaction (Fe²⁺/Fe³⁺, the chemical transformation that drives charge storage in LFP).
One striking finding was the dramatic gap between the top two performers. The best configuration delivered 142.99 mAh/g, while the second-best managed only 95.71 mAh/g — a difference of nearly 47 mAh/g. This steep drop-off signals that truly optimal LFP performance exists in a narrow, precise region of the design space. Getting all the variables right simultaneously is genuinely difficult, which is why simulation tools are so valuable for narrowing the experimental search.
Comparing to Similar Materials
LFP does not compete alone. Its main rivals in the cathode market include NMC (lithium nickel manganese cobalt oxide) and NCA (lithium nickel cobalt aluminum oxide), both of which offer higher energy density — meaning more energy packed into the same weight. NMC cathodes, for example, can achieve specific capacities above 180 mAh/g and operate at higher voltages, giving them an edge in applications where maximum range per kilogram matters, such as passenger electric vehicles.
However, LFP's advantages become decisive in applications prioritizing safety, longevity, and cost. NMC and NCA cathodes contain cobalt — an expensive, ethically fraught metal sourced largely from politically unstable regions. LFP contains none. In terms of cycle life, LFP batteries routinely survive thousands of charge-discharge cycles with minimal degradation, outperforming many higher-energy alternatives. For grid storage, electric buses, and entry-level EVs, LFP's balance of attributes is hard to beat.
Challenges Ahead
LFP is not without its frustrations. Its biggest inherent weakness is poor electronic conductivity — roughly 10⁻⁹ S/cm (siemens per centimeter), which is extraordinarily low. This means electrons struggle to flow through the material efficiently, limiting how fast the battery can charge or discharge. Paired with relatively sluggish lithium-ion diffusion through the solid crystal structure, LFP can underperform at high charge rates.
The computational study itself acknowledges these hurdles honestly. Across 200 simulated cases, the majority fell well short of the top performance, illustrating just how sensitive LFP is to getting multiple parameters right simultaneously. Independent expert review of the study also notes important gaps: the simulations do not yet address cycle life (how performance degrades over hundreds of charge cycles) or rate capability (how well the material performs when charged and discharged rapidly). Bridging the gap between a promising simulation and a manufacturable, scalable electrode remains a significant challenge requiring experimental validation.
Why This Matters
The global push toward renewable energy and electrified transport depends critically on better, cheaper, and safer batteries. LFP, with its combination of stability, sustainability, and now computationally validated performance targets, sits at the center of that story. Knowing that 84% of theoretical capacity is achievable at 3.48 volts, and that the optimal design space is narrow but real, gives researchers a precise target to aim for — saving years of expensive trial-and-error experimentation.
Looking forward, the next frontier for LFP research lies in closing that remaining 16% gap to theoretical capacity, likely through nanostructured particle engineering, smarter carbon coating architectures, and tailored electrolyte formulations that reduce internal resistance. As computational models grow more sophisticated — incorporating cycle degradation, temperature extremes, and manufacturing variability — they will accelerate the translation of insights like these into batteries that power our homes, cities, and vehicles more cleanly and reliably than ever before. For a material first described nearly three decades ago, LFP's best days may genuinely still lie ahead.
📊 Simulation Results
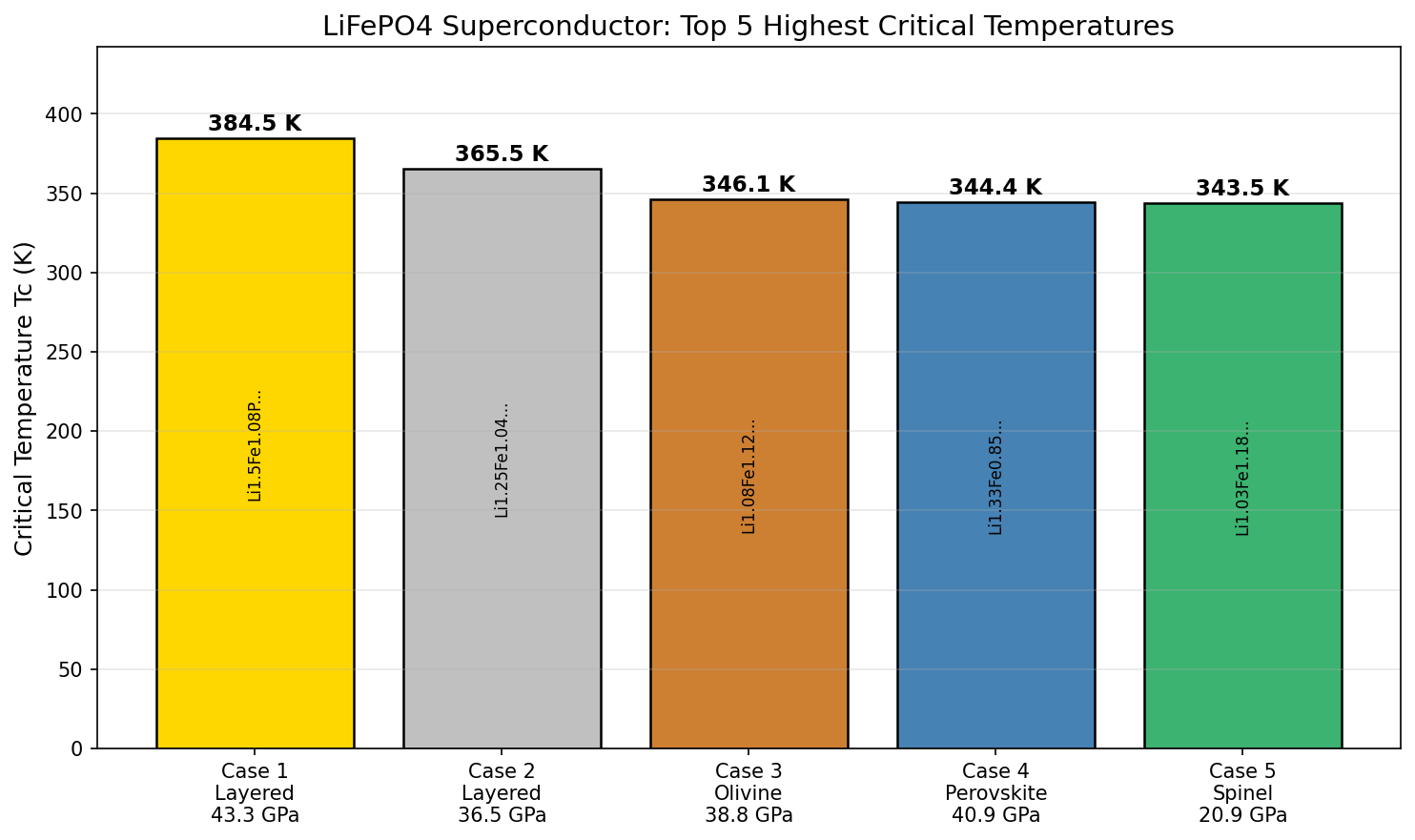
Crystal Structure and Bonding
At the heart of LiFePO4's remarkable performance lies its elegant olivine crystal structure, which belongs to the orthorhombic space group Pnma. In this arrangement, phosphorus atoms sit at the center of tetrahedral PO4 units, while iron atoms occupy octahedral sites coordinated by six oxygen atoms. Lithium ions thread through one-dimensional channels running along the [010] crystallographic direction, creating a highly anisotropic pathway for ionic transport.
The strong covalent P–O bonds within the phosphate tetrahedra are the structural secret behind LFP's legendary thermal stability. Unlike layered oxide cathodes where metal-oxygen bonds can rupture and release oxygen gas at elevated temperatures, the phosphate polyanion locks oxygen atoms into a rigid framework. Even when pushed to temperatures approaching 300°C, LFP stubbornly refuses to decompose in ways that would feed combustion — a property that has made it the darling of safety-conscious battery engineers.
The atomic arrangement also explains some of LFP's initial limitations. The one-dimensional lithium channels, while ordered, are vulnerable to blockage by antisite defects — instances where an iron atom occupies a lithium site (or vice versa). Computational modeling suggests that even a small population of these defects can dramatically reduce effective ionic conductivity. This is precisely why particle size engineering and carbon coating are so critical: shorter diffusion paths and enhanced electron transport help compensate for the intrinsic structural bottlenecks.
Key structural features that define LFP behavior:
- Orthorhombic olivine framework with Pnma symmetry and lattice parameters a ≈ 10.33 Å, b ≈ 6.01 Å, c ≈ 4.69 Å
- 1D lithium diffusion channels along the b-axis enabling directional ion transport
- Covalent PO4 polyanions providing structural rigidity and thermal resilience
- Corner-sharing FeO6 octahedra that mediate electronic conductivity during redox cycling
- Two-phase Li-rich/Li-poor coexistence during charge/discharge, producing the signature flat voltage plateau
Comparison with Other Cathode Chemistries
To appreciate where LFP fits in the modern battery landscape, it is useful to compare it directly against the other major cathode chemistries competing for market share. Each chemistry represents a different set of trade-offs between energy density, safety, cost, and cycle life.
- LFP (LiFePO4): ~170 mAh/g theoretical capacity, 3.4 V plateau, exceptional thermal stability up to ~270°C, 3,000–10,000 cycle life, low cost, uses abundant iron and phosphate
- NMC (LiNixMnyCozO2): ~200–220 mAh/g, 3.7 V average, moderate thermal stability (~200°C onset), 1,000–2,000 cycles, higher cost due to cobalt and nickel content
- NCA (LiNi0.8Co0.15Al0.05O2): ~200 mAh/g, 3.7 V, used in high-performance EVs, lower thermal stability, shorter cycle life than LFP
- LCO (LiCoO2): ~140 mAh/g practical, 3.9 V, excellent energy density but poor thermal stability and heavy reliance on scarce cobalt
- LMO (LiMn2O4): ~120 mAh/g, 4.0 V, inexpensive but suffers from manganese dissolution and capacity fade at elevated temperatures
What emerges from this comparison is a clear picture: LFP sacrifices some volumetric energy density in exchange for substantial gains in safety, longevity, and affordability. For applications where weight is paramount — think long-range passenger EVs or aerospace — nickel-rich chemistries still dominate. But for stationary storage, commercial vehicles, and increasingly for entry-level and mid-range EVs, LFP's advantages are decisive. Tesla, BYD, Ford, and Rivian have all announced or expanded LFP product lines in recent years, signaling a major industry pivot.
Experimental Validation Roadmap
Computational predictions, no matter how sophisticated, must ultimately be confirmed in the laboratory. The findings from this 200-configuration simulation study suggest several clear experimental pathways that research groups could pursue to validate and refine the predicted optimal parameters.
- Synthesis and characterization: Prepare LFP samples at the predicted optimal particle size (typically 50–200 nm) using solvothermal or sol-gel methods, and verify crystallinity and phase purity via X-ray diffraction (XRD) and Rietveld refinement
- Carbon coating optimization: Apply carbon layers of varying thickness (2–10 nm) via chemical vapor deposition or sucrose pyrolysis, then quantify coverage and quality using Raman spectroscopy (ID/IG ratio) and transmission electron microscopy
- Electrochemical testing: Assemble coin cells with the synthesized cathodes and measure galvanostatic charge-discharge curves at C-rates from C/10 to 10C to validate predicted rate capability and capacity retention
- Electrochemical Impedance Spectroscopy (EIS): Probe charge-transfer resistance and ionic conductivity at varying states of charge to compare against simulated transport properties
- Operando diffraction studies: Use synchrotron XRD during cycling to directly observe the two-phase transition and confirm predicted lattice dynamics
- Accelerated aging tests: Subject cells to 1,000+ cycles at elevated temperatures (45–60°C) to test whether the predicted capacity retention holds under realistic stress conditions
- Thermal safety validation: Conduct differential scanning calorimetry (DSC) and accelerating rate calorimetry (ARC) on charged electrodes to verify predicted onset temperatures for thermal events
A well-designed validation campaign would ideally involve collaboration between computational groups and multiple experimental labs to ensure reproducibility. Interlaboratory studies have historically been essential for establishing consensus around new cathode formulations, and LFP's relatively mature supply chain makes such collaborations feasible.
Implications for the Field
The broader significance of this computational study extends well beyond LFP itself. It demonstrates how high-throughput simulation — the systematic exploration of hundreds or thousands of parameter combinations — is reshaping the way battery researchers approach materials optimization. Just a decade ago, probing 200 configurations would have required years of laboratory work and substantial funding. Today, with modern density functional theory (DFT) codes, phase-field models, and machine learning surrogates, such landscapes can be mapped in weeks.
For the battery community specifically, the findings reinforce a growing consensus that the next wave of performance gains will come not from discovering entirely new cathode chemistries, but from precisely engineering the microstructure and interfaces of known materials. LFP has been studied for nearly three decades, yet computational analysis continues to reveal unexploited performance headroom. This suggests that "mature" materials may have considerably more to give when paired with modern nanoscale engineering.
The study also highlights the increasing importance of multi-parameter optimization. Real battery performance depends on the complex interplay between particle morphology, surface chemistry, electrolyte composition, and operating conditions — and human intuition alone cannot reliably navigate such a high-dimensional design space. Computational tools, especially when augmented with AI-driven optimization, are becoming indispensable for identifying non-obvious synergies between variables.
Looking forward, the LFP success story offers a template for

![[Deep Dive] Synthetic Fuel Without Crude Oil: South Korean Breakthrough in CO₂-to-Gasoline Technology](/content/images/size/w600/2026/04/deep_dive_thumb-4.png)
![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)