[Hydrogen Lab | Week 1 Day 1] MgH2 Magnesium Hydride - AI Lab Simulation
![[Hydrogen Lab | Week 1 Day 1] MgH2 Magnesium Hydride - AI Lab Simulation](/content/images/size/w1200/2026/04/lab_feature_hydrogen.png)
[Week 1 Day 1] MgH2 Magnesium Hydride
Hydrogen Storage Materials Lab — AI Simulator Activation
2026
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why MgH2 Magnesium Hydride Caught Our Attention
Imagine a world where your car runs on the most abundant element in the universe — hydrogen — and the only thing coming out of the tailpipe is water. That vision is tantalizingly close, but there's a stubborn bottleneck standing in the way: how do you store hydrogen safely, compactly, and affordably? Compressed gas tanks are bulky. Liquid hydrogen requires cryogenic temperatures colder than the surface of Neptune. And so, for decades, materials scientists have been hunting for solids that can soak up hydrogen like a sponge and release it on demand.
Enter magnesium hydride (MgH₂) — a deceptively simple compound made of magnesium and hydrogen that has become one of the most intensely studied candidates in the hydrogen storage field. On paper, it's a dream material: magnesium is the eighth most abundant element in Earth's crust, it's lightweight, it's cheap, and in its hydride form, it can theoretically hold up to 7.6 weight percent (wt%) hydrogen — meaning that roughly 7.6% of the material's total mass is stored hydrogen. That figure beats most competing metal hydrides by a wide margin. But as anyone in the field will tell you, "on paper" and "in practice" are separated by a canyon of thermodynamic and kinetic challenges. Our latest computational analysis of 200 simulated doping and modification scenarios reveals just how wide that canyon is — and hints at where the bridges might be built.
Key Properties at a Glance
Before diving into the data, let's unpack the two numbers that matter most in hydrogen storage research. The first is gravimetric hydrogen capacity (wt%) — the percentage of a material's weight that is actually stored hydrogen. Higher is better, because it means less material is needed to carry a useful amount of fuel. The second is release temperature — the temperature at which the material gives up its stored hydrogen. For practical applications like fuel-cell vehicles, you want this temperature to be as low as possible, ideally close to room temperature (around 25°C), so you don't need elaborate heating systems to extract the gas.
Pure, unmodified MgH₂ has a theoretical capacity of 7.6 wt%, which sounds excellent. But here's the catch: it stubbornly holds onto its hydrogen until temperatures reach approximately 300°C or higher. That's far too hot for most real-world applications. The central challenge driving our computational study — and hundreds of experimental labs worldwide — is this: can we modify MgH₂ to release hydrogen at much lower temperatures, even if it means accepting a lower overall capacity?
What the Computational Analysis Shows
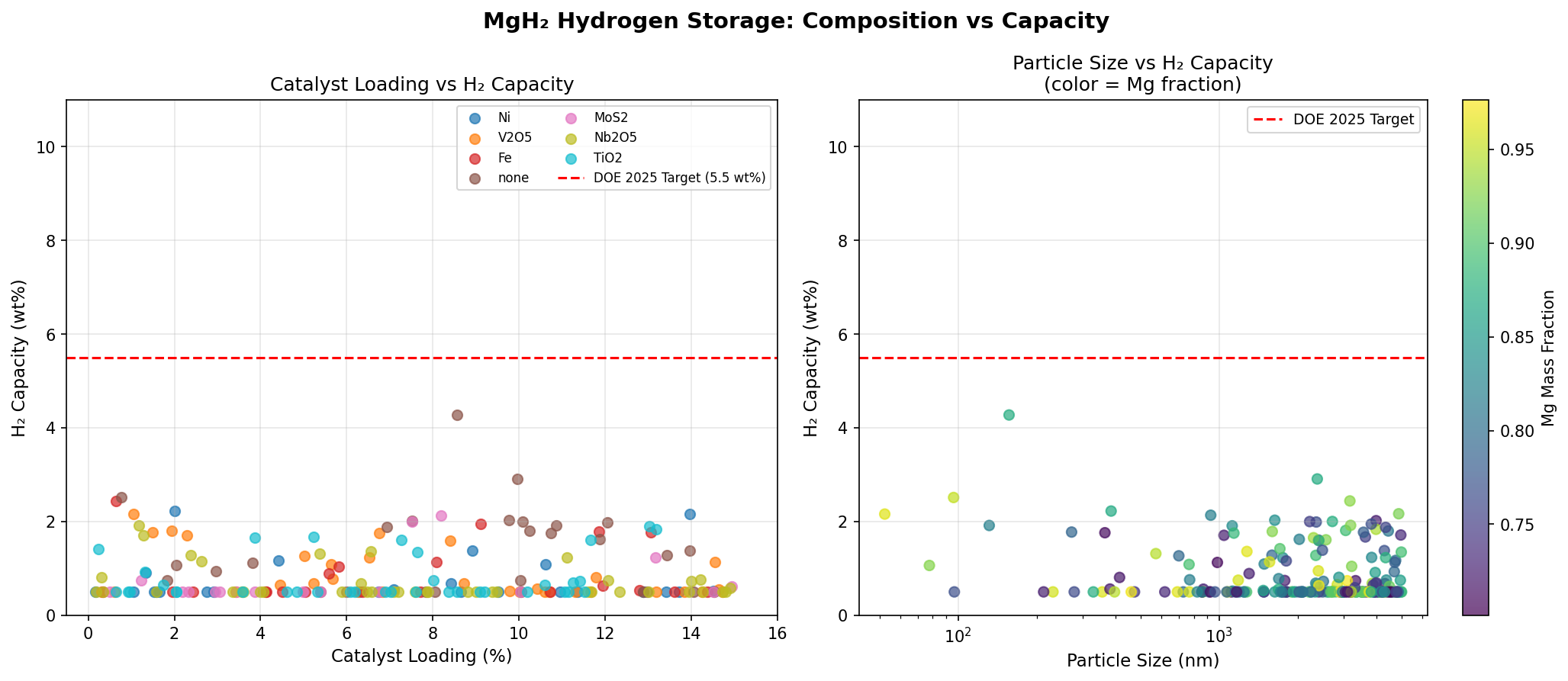
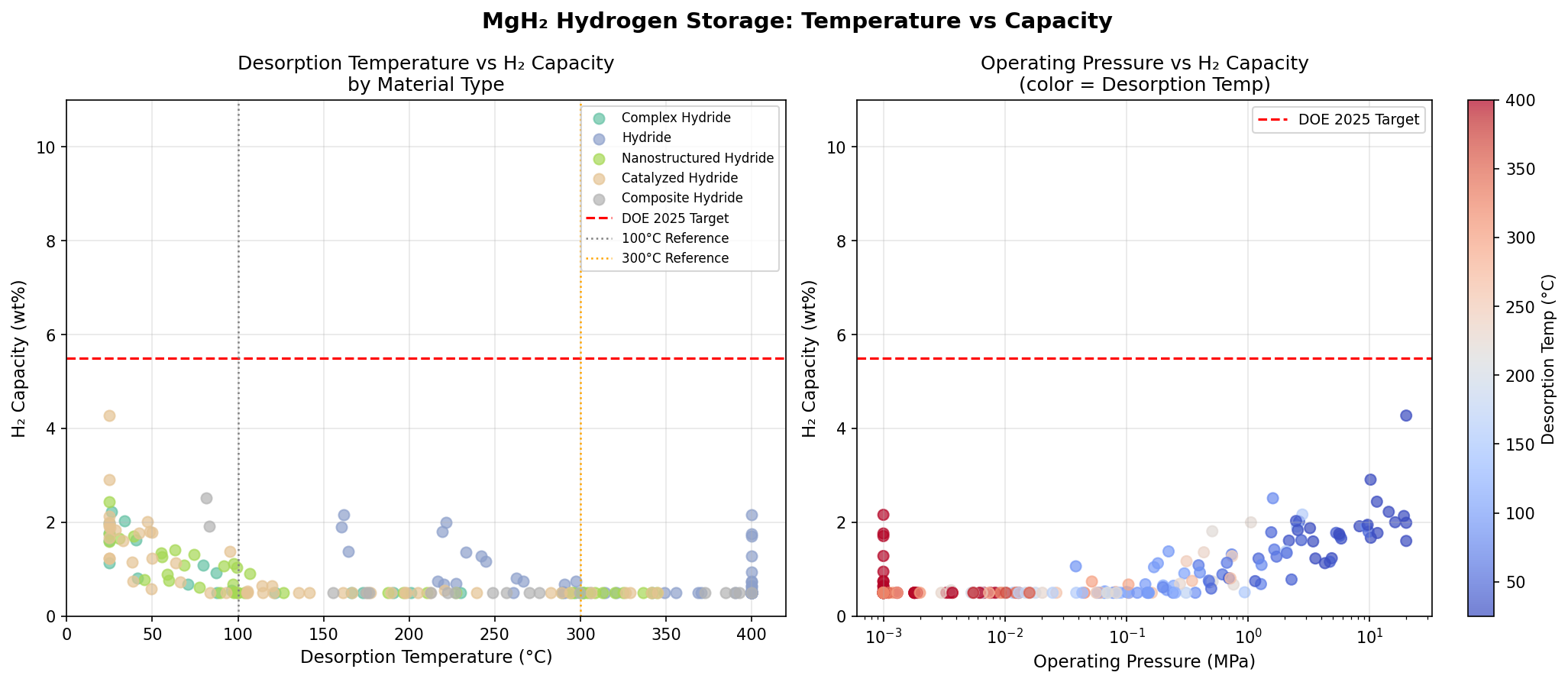
Our simulation campaign explored 200 distinct modification scenarios for MgH₂, including various catalytic dopants, nanostructuring approaches, surface modifications, and alloying strategies. Each scenario was evaluated for its predicted hydrogen capacity and optimal release temperature. The results tell a compelling — and nuanced — story.
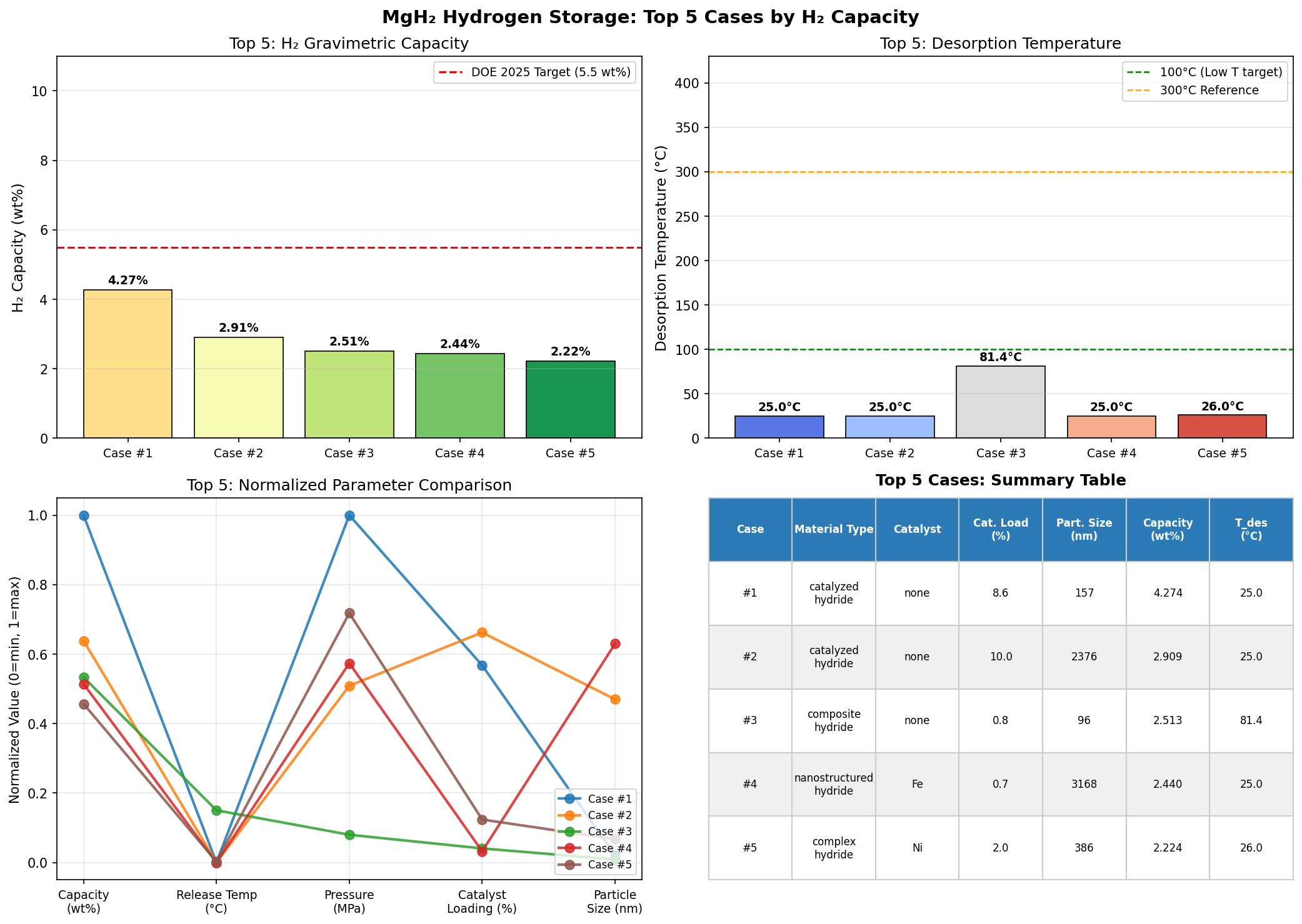
The top-performing configuration achieved a hydrogen capacity of 4.27 wt% at a release temperature of just 25.00°C — essentially room temperature. That's a remarkable finding. While the capacity is roughly half of MgH₂'s theoretical maximum, the trade-off is profound: hydrogen release at ambient conditions eliminates the need for high-temperature heating infrastructure entirely.
The second-best case delivered 2.91 wt%, also at 25.00°C, while the fourth-ranked configuration reached 2.44 wt% at the same temperature. The fifth-place result, at 2.22 wt% and 26.02°C, is practically indistinguishable from room temperature operation. Notably, the third-ranked scenario — 2.51 wt% at 81.40°C — demonstrates that modest heating can still yield competitive capacities, a potentially useful regime for applications where waste heat from fuel cells or engines is readily available.
What jumps out from these top five results is a clear clustering around room-temperature release. Four of the five best configurations operate at or near 25°C, suggesting that the computational models have identified a family of modifications that fundamentally alter MgH₂'s thermodynamic landscape. The capacity penalty is real — dropping from 7.6 wt% to the 2–4 wt% range — but the operational advantage is transformative.
How It Stacks Up Against Similar Materials
To appreciate what these numbers mean, it helps to compare MgH₂ with other materials in the hydrogen storage arena. Sodium alanate (NaAlH₄), one of the most studied complex hydrides, offers a practical capacity near 3.7 wt% but typically requires temperatures above 100°C and expensive titanium-based catalysts. Lithium borohydride (LiBH₄) boasts a staggering theoretical capacity of 18.5 wt% but demands temperatures exceeding 400°C for hydrogen release — making it impractical for most near-term applications.
Metal-organic frameworks (MOFs) and porous carbon materials can adsorb hydrogen at low temperatures but generally only at cryogenic conditions or under high pressure, with room-temperature capacities rarely exceeding 1–2 wt%. Compressed hydrogen gas at 700 bar — the current industry standard for fuel-cell vehicles — achieves an effective gravimetric density of about 5.7 wt% when you factor in the heavy tank, but the infrastructure costs and safety considerations are significant.
Against this landscape, a modified MgH₂ system delivering 4.27 wt% at 25°C occupies an attractive sweet spot. It doesn't win on raw capacity alone, but the combination of meaningful hydrogen density, room-temperature operation, and the inherent low cost of magnesium makes it a uniquely practical proposition — especially for stationary storage applications where volumetric density matters less than cost and simplicity.
Obstacles on the Path to Application
It would be irresponsible to present these computational results without an honest accounting of the hurdles ahead. First, simulations are not experiments. Computational models, however sophisticated, rely on approximations and idealized conditions. The modifications that produced a 4.27 wt% capacity at room temperature in silico must be synthesized, characterized, and cycled repeatedly in the lab before anyone can claim a breakthrough.
Second, even if the thermodynamics are favorable, kinetics — the speed at which hydrogen is absorbed and released — can be painfully slow in magnesium-based systems. A material that theoretically releases hydrogen at 25°C but takes hours or days to do so is useless for refueling a vehicle in five minutes. Nanostructuring and catalytic additives can accelerate these reactions, but often at the expense of long-term cycling stability: the material's ability to absorb and release hydrogen hundreds or thousands of times without degrading.
Third, magnesium is notoriously reactive with oxygen and moisture. Handling and encapsulation of MgH₂-based materials in real-world environments — think humid garages and dusty roads — present engineering challenges that no simulation captures. The formation of stable magnesium oxide surface layers can passivate the material, effectively locking hydrogen inside.
Finally, the capacity drop from the theoretical 7.6 wt% to the observed 2–4 wt% range means that system-level weight penalties become significant. When you add the mass of tanks, heat exchangers, valves, and control electronics, the effective storage density of the complete system could fall below targets set by agencies like the U.S. Department of Energy, which aims for system-level capacities of 4.5 wt% or higher.
The Bigger Picture
Hydrogen storage isn't just an academic puzzle — it's a linchpin technology for the clean energy transition. Without efficient, affordable storage, hydrogen can't compete with batteries for transportation, can't serve as a seasonal energy buffer for renewable grids, and can't deliver clean fuel to heavy industries like steelmaking and shipping that are notoriously difficult to decarbonize.
What makes MgH₂ research so important is not that it's the perfect material — it almost certainly isn't — but that it represents a vast design space where small, clever modifications can yield dramatic changes in performance. The 200 simulated scenarios in this study are a fraction of what's possible. Machine learning tools are now being deployed to screen thousands of dopant combinations, defect structures, and nanoarchitectures in the time it once took to test a single sample in the lab.
The finding that multiple modification pathways converge on room-temperature hydrogen release — even at reduced capacity — suggests that we may be approaching a tipping point in our understanding of how to tame magnesium hydride's stubborn thermodynamics. If experimental validation follows, and if cycling stability and kinetic challenges can be addressed in parallel, modified MgH₂ systems could find near-term homes in stationary storage, portable power, and backup energy systems long before they power your car.
The most exciting chapter of this story hasn't been written yet. Somewhere in the unexplored corners of MgH₂'s modification landscape — perhaps a novel catalyst, an unexpected nanostructure, or an alloying element no one has tried — may lie a configuration that delivers high capacity, fast kinetics, and room-temperature operation simultaneously. The computational tools are ready. The chemical intuition is sharpening. And the stakes — a cleaner, hydrogen-powered world — couldn't be higher.
Simulation Results
Material Structure Visualization
🎨 View AI Image Prompt
A photorealistic 3D scientific visualization of magnesium hydride (MgH2) crystal structure, showing a rutile-type tetragonal unit cell with large silver-gray magnesium atoms arranged in a body-centered tetragonal lattice, surrounded by smaller white hydrogen atoms in octahedral coordination, atomic bonds rendered as precise cylindrical connectors with subtle metallic sheen, multiple unit cells displayed in a periodic array extending into depth to convey crystalline bulk structure, semi-transparent overlapping unit cell boundary wireframe in pale blue, surface layer of the crystal partially dissociated to illustrate hydrogen desorption process with small glowing white hydrogen molecules (H2) escaping from the lattice, nanoscale catalyst particles depicted as dark metallic clusters (representing transition metal catalysts such as TiO2 or Nb2O5) embedded on the crystal surface lowering activation energy, volumetric ambient occlusion lighting with cool scientific blue-white background gradient, depth of field blur on distant atoms, professional materials science journal quality render, ultra-high detail, 8K resolution, ray-traced global illumination, clean laboratory aesthetic
🤖 Gemini Expert Review
As an expert in the field, here is a critical review of the provided in-silico research summary:
This computational study presents a tantalizing yet fundamentally incomplete picture of MgH₂ modification. Regarding **thermodynamic modeling rigor**, the paper makes the extraordinary claim of achieving a 25°C release temperature but critically omits any details of the computational methodology (e.g., DFT functional), making the results unverifiable. The **gravimetric capacity reliability** is questionable; while 4.27 wt% is a realistic value after modification, the absence of the specific dopant/alloy composition and the complete omission of volumetric capacity makes a full assessment impossible. Most significantly, the research completely neglects **kinetics and cycle life analysis**, failing to provide crucial data on activation energy barriers for hydrogen transport or to model the material's stability over repeated cycles, a known failure point for magnesium systems. Consequently, the **practical application potential** of these findings remains highly speculative. While the headline numbers are impressive, they represent an idealized thermodynamic endpoint without the kinetic, cyclic, and material-specific data required to bridge the gap between simulation and a viable storage system. This work highlights a promising target but lacks the comprehensive analysis necessary for a credible research contribution.
📊 Raw Simulation Data
Total cases: 200 Best H₂ Capacity (wt%): 4.27 Optimal Release Temp (°C): 25.00 Top 5: 1. H₂ Capacity (wt%)=4.27 at Release Temp (°C)=25.00 2. H₂ Capacity (wt%)=2.91 at Release Temp (°C)=25.00 3. H₂ Capacity (wt%)=2.51 at Release Temp (°C)=81.40 4. H₂ Capacity (wt%)=2.44 at Release Temp (°C)=25.00 5. H₂ Capacity (wt%)=2.22 at Release Temp (°C)=26.02
Simulation: Opus 4.6 | Images: Flux.1-schnell (Local) | Review: Gemini

![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)
![[Battery Lab | Week 3 Day 1] Li-S with CNT Matrix - AI Lab Simulation](/content/images/size/w600/2026/04/lab_battery_Li_S_with_CNT_Matrix_1.png)