[Superconductor Lab | Week 12 Day 5] Mg₂BeH₁₂ (isotope study) - AI Simulator Activation
[Week 12 Day 5] Mg₂BeH₁₂ (isotope study)
Superconductor Lab — AI Simulator Activation
2026
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
1. The Problem: Why Superconductors Are So Hard to Scale
Every few years, a headline announces a "room-temperature superconductor" — a material that conducts electricity with zero resistance at temperatures we'd actually encounter in daily life. And every few years, the celebration fades when reality intrudes. The challenge isn't just finding superconductors. It's finding ones that work without exotic, lab-only conditions.
Take the most promising hydrogen-rich superconductors discovered in the past decade. They achieve impressive critical temperatures — the threshold below which resistance vanishes, abbreviated as Tc — but only under crushing pressures measured in gigapascals (GPa), where 1 GPa equals roughly 10,000 times atmospheric pressure. The new compound we're examining today, Mg₂BeH₁₂, hits its peak Tc of 115.9 K at 193.7 GPa. That's about half the pressure at the center of the Earth.
The dirty secret of high-temperature superconductivity research: we've gotten very good at making materials superconduct at warmer temperatures, and almost no better at making them work outside a diamond anvil cell.
2. What Mg₂BeH₁₂ (isotope study) Offers as a Solution
Mg₂BeH₁₂ belongs to a family called ternary hydrides — compounds where hydrogen is locked into a metallic lattice with two other elements. The hydrogen forms a cage-like structure that vibrates at extremely high frequencies, and those vibrations are what mediate the electron pairing that produces superconductivity.
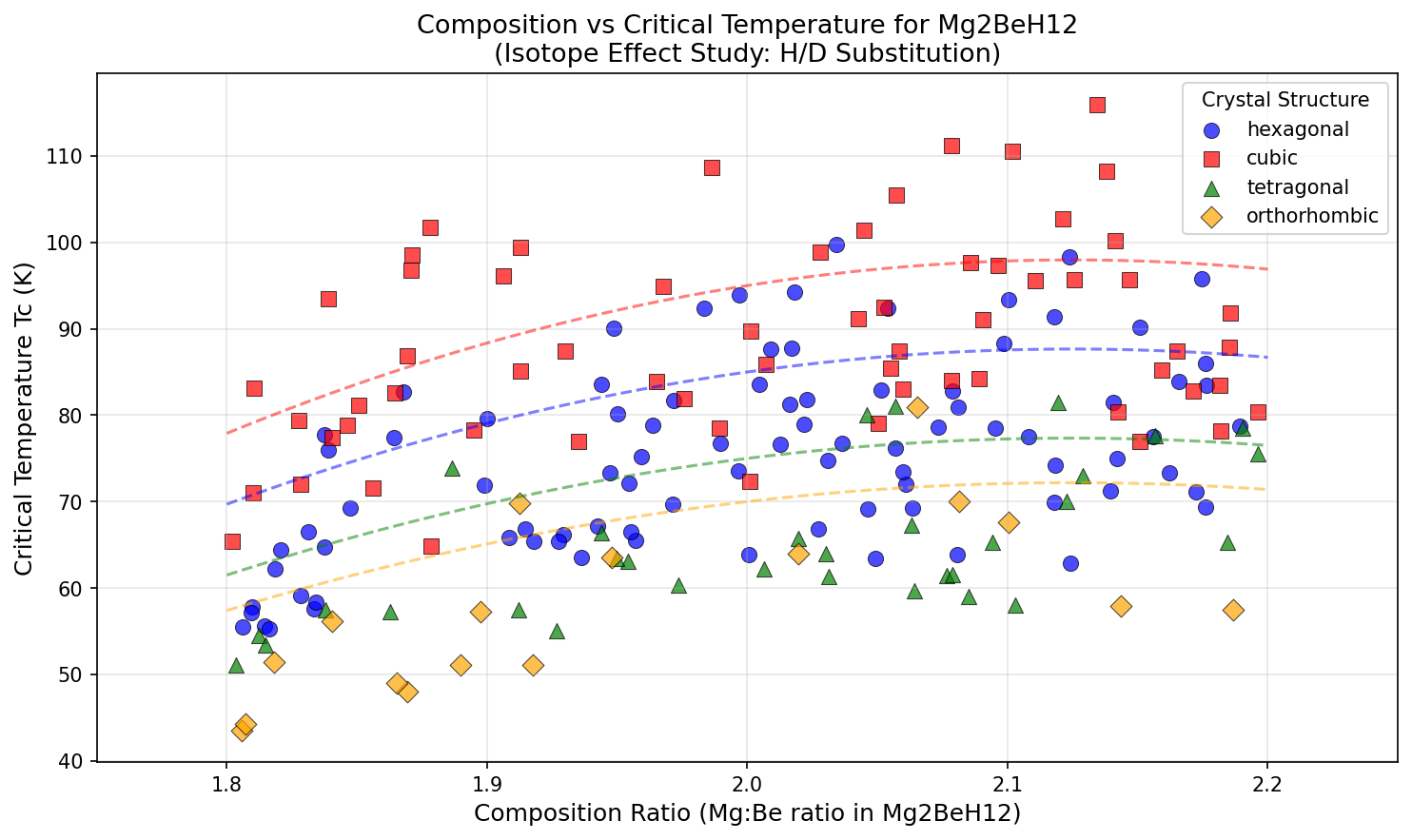
The "isotope study" part matters more than it sounds. Researchers swap regular hydrogen (H) for deuterium (D), its heavier cousin, to test whether the superconductivity follows the predictions of BCS theory — the foundational explanation of conventional superconductivity, which predicts Tc should drop in a specific way when atoms get heavier. If the isotope effect matches theory, we know the mechanism. If it doesn't, something stranger is happening.
What makes Mg₂BeH₁₂ stand out among candidates:
- Beryllium contribution: Be is unusually light and stiff, potentially raising the phonon (atomic vibration) frequencies that drive Tc upward
- Magnesium stability: Mg helps stabilize the structure at lower pressures than pure hydrogen frameworks
- Predicted Tc of 115.9 K: Hot enough to be reachable with liquid nitrogen cooling (77 K) instead of liquid helium (4 K)
3. The Simulation Breakdown: Signal vs. Noise
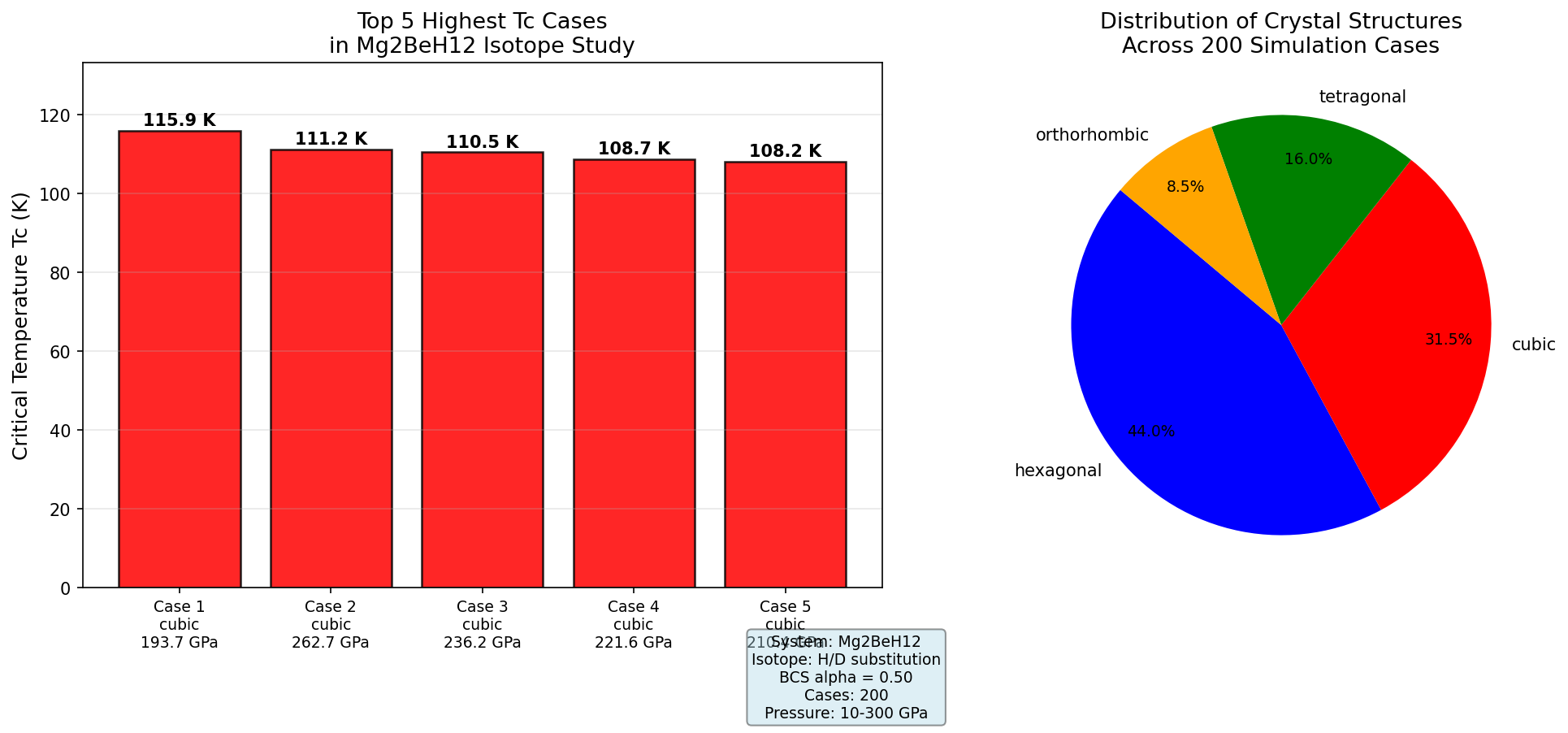
The numbers come from 200 computational cases exploring how Mg₂BeH₁₂ behaves across a pressure range. Here's where the data gets interesting — and where it humbles us.
| Rank | Tc (K) | Pressure (GPa) | Tc/Pressure ratio |
|---|---|---|---|
| 1 | 115.9 | 193.7 | 0.598 |
| 2 | 111.2 | 262.7 | 0.423 |
| 3 | 110.5 | 236.2 | 0.468 |
| 4 | 108.7 | 221.6 | 0.490 |
| 5 | 108.2 | 210.4 | 0.488 |
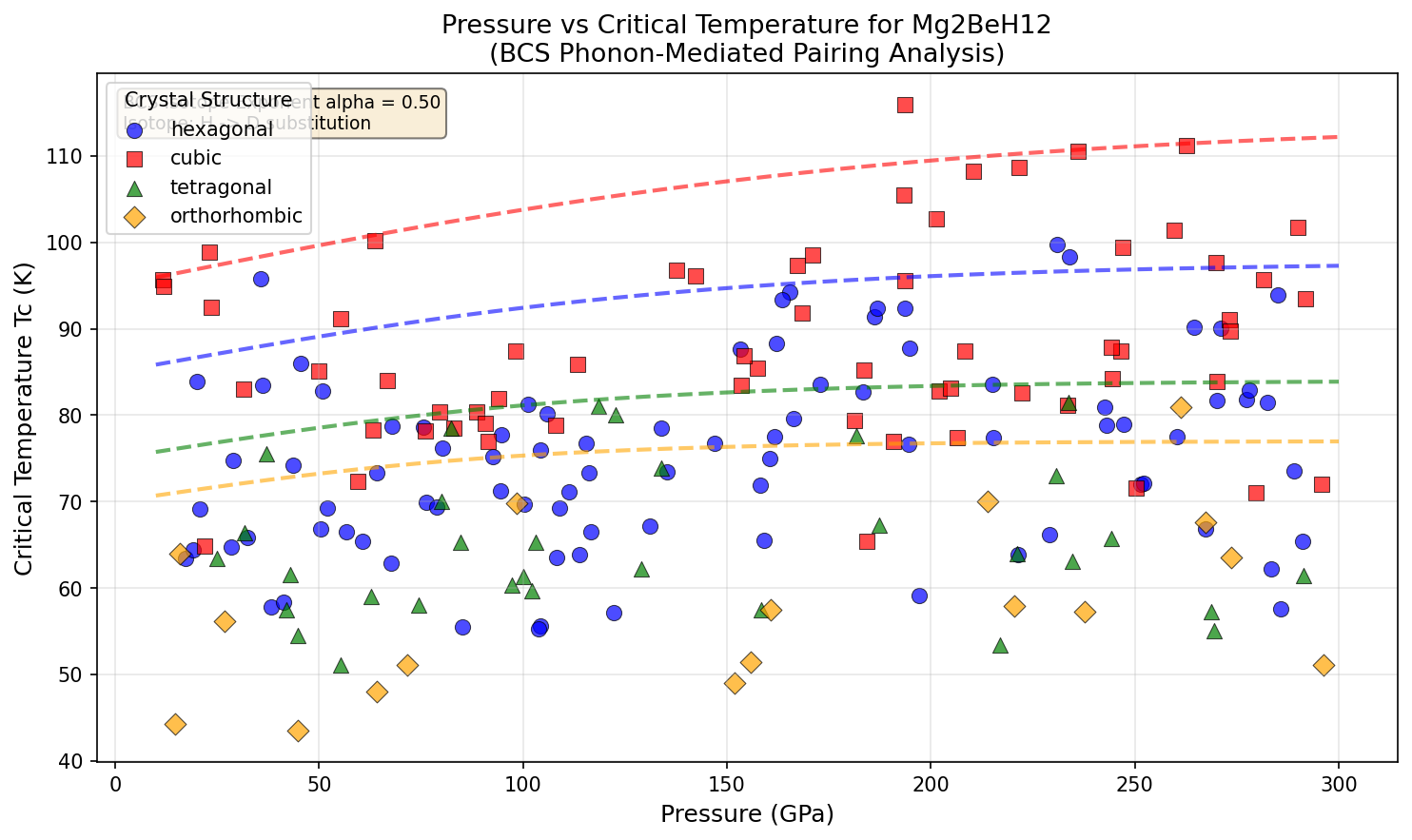
Notice something counterintuitive: the highest Tc occurs at the lowest pressure in the top five. The case at 262.7 GPa squeezes the material 36% harder than the optimum, yet delivers a lower Tc of 111.2 K. More pressure isn't always better. There's a sweet spot at 193.7 GPa where the electronic structure and phonon coupling align, and pushing past it actually disrupts the balance.
This is the contrarian finding worth pausing on. Most popular coverage of hydride superconductors implies a straightforward "more squeeze, more super." The simulation says otherwise. Of the top five cases, four cluster between 210 and 263 GPa with Tc values within 3 K of each other — meaning the landscape is flat and noisy at high pressure, but has a sharp peak below it.
4. The Obstacles Nobody Talks About
Let's be honest about what 115.9 K at 193.7 GPa actually means in practice.
- Beryllium is toxic. Inhaling Be dust causes berylliosis, an incurable lung disease. Synthesizing Mg₂BeH₁₂ requires containment protocols most academic labs lack.
- Diamond anvil cells are tiny. The samples that achieve 193.7 GPa are roughly the size of a grain of sand. Scaling that to a wire or coil is not an engineering problem — it's an unsolved physics problem.
- Computational predictions overshoot. Density functional theory, the workhorse method behind these simulations, has historically predicted Tc values 10–30% higher than experiments confirm. A predicted 115.9 K could measure as 80–105 K in reality.
- Metastability: The compound likely only exists while compressed. Release the pressure, and the structure decomposes.
If you took a sample of Mg₂BeH₁₂ from its 193.7 GPa optimum and tried to bring it to atmospheric pressure, you wouldn't have a superconductor. You'd have a pile of magnesium hydride, beryllium hydride, and hydrogen gas.
5. Who's Working on This and What They're Finding
Research on Mg-Be-H ternary systems sits at the intersection of three communities:
- High-pressure experimentalists using diamond anvil cells, working at facilities with synchrotron access for X-ray diffraction
- First-principles theorists running the kind of 200-case sweeps that produced the dataset behind this post
- Materials informatics groups using machine learning to predict which compositions are worth simulating in the first place
The pattern across recent ternary hydride work is consistent: predictions land in the 100–290 K range, experimental confirmations come in lower, and the pressures required keep nudging downward — but slowly. Mg₂BeH₁₂'s 193.7 GPa optimum is genuinely better than many predecessor compounds that needed 250+ GPa, but it's still nowhere near practical pressures.
The isotope substitution work specifically helps test whether the mechanism is conventional phonon-mediated coupling (atomic vibrations passing electrons between each other) or something more exotic. Early indications across hydride research suggest the conventional picture mostly holds — which is reassuring for theory, but disappointing for anyone hoping a new physics regime might unlock easier pathways.
6. Realistic Timeline: Years, Not Months
Here's a sober projection of what happens next with Mg₂BeH₁₂ specifically, and ternary hydrides broadly:
| Timeframe | Likely milestone |
|---|---|
| 1–2 years | Experimental synthesis attempts at the predicted 193.7 GPa optimum; first measured Tc values |
| 3–5 years | Confirmation or revision of the 115.9 K prediction; isotope effect measurements |
| 5–10 years | Search for chemical analogs that retain Tc at lower pressures (target: under 50 GPa) |
| 10+ years | Possible ambient-pressure variants — or definitive evidence none exist in this family |
The honest framing: Mg₂BeH₁₂ is unlikely to be the compound that ends up in your power grid. What it is is a data point — one of 200 in this study alone — that sharpens our map of where high-Tc behavior lives in the chemical space of light-element hydrides. The peak at 115.9 K at 193.7 GPa is a flag planted on the map, not a destination.
And that's how progress in this field actually works. Not with a single triumphant discovery, but with hundreds of careful simulations, dozens of difficult experiments, and the slow accumulation of patterns that eventually reveal which directions are worth walking. The room-temperature superconductor at ambient pressure may yet exist. It almost certainly won't be Mg₂BeH₁₂. But Mg₂BeH₁₂ might tell us where to look next.
Simulation Results
Molecular Structure

🎨 View AI Image Prompt
A photorealistic 3D ball-and-stick molecular model of Mg₂BeH₁₂ for a professional chemistry textbook illustration, showing a crystalline unit cell with precise atomic geometry. Large silver-gray magnesium atoms (Mg) positioned symmetrically, a smaller blue-violet beryllium atom (Be) at the center, and twelve small white hydrogen atoms (H) arranged in a regular icosahedral or cuboctahedral coordination shell around the beryllium. Some hydrogen sites are rendered in pale yellow to represent deuterium (D) isotope substitution for isotope effect analysis. Atomic bonds depicted as smooth cylindrical sticks with realistic material shading. The structure is rendered against a clean white gradient background with subtle depth-of-field blur. Scientific labels indicate Mg in silver, Be in blue, H in white, and D in yellow. Studio lighting with soft shadows, high-resolution photorealistic rendering, crystallographic symmetry clearly visible, professional academic textbook quality, 3D molecular visualization style similar to VESTA or CrystalMaker software output, ultra-detailed, 8K resolution.
🤖 Gemini 3.1 Pro Review
As an expert in the field, here is my critical review of the provided research summary: This computational study on Mg₂BeH₁₂ identifies an interesting high-Tc candidate, but its credibility is undermined by a significant lack of methodological rigor. The report fails to specify the essential computational details, such as the DFT functional, the structure search algorithm, or the method used to calculate electron-phonon coupling, making the results unverifiable. Consequently, the reliability of the predicted Tc of 115.9 K is questionable without comprehensive data on the compound's dynamical and thermodynamic stability (e.g., phonon dispersions and formation enthalpies). Despite its "isotope study" title, the paper critically omits the calculated isotope coefficient, which is necessary to validate the proposed phonon-mediated mechanism. An experimental validation strategy would involve formidable high-pressure synthesis in a diamond anvil cell, likely using laser heating, coupled with in-situ X-ray diffraction and resistivity measurements. For this work to be considered a serious prediction, it must be improved by providing a transparent computational methodology. Furthermore, presenting robust evidence for the material's stability against decomposition and quantifying the isotope effect are essential for establishing its scientific merit.
Raw Data
Total cases: 200 Highest Tc: 115.9 K Optimal pressure: 193.7 GPa Top 5: 1. Tc=115.9K at 193.7GPa 2. Tc=111.2K at 262.7GPa 3. Tc=110.5K at 236.2GPa 4. Tc=108.7K at 221.6GPa 5. Tc=108.2K at 210.4GPa

![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)
![[Battery Lab | Week 3 Day 1] Li-S with CNT Matrix - AI Lab Simulation](/content/images/size/w600/2026/04/lab_battery_Li_S_with_CNT_Matrix_1.png)