[Hydrogen Lab | Week 1 Day 4] TiH2 Titanium Hydride - AI Lab Simulation
![[Hydrogen Lab | Week 1 Day 4] TiH2 Titanium Hydride - AI Lab Simulation](/content/images/size/w1200/2026/04/lab_hydrogen_TiH2_Titanium_Hydride_1.png)
[Week 1 Day 4] TiH2 Titanium Hydride
Hydrogen Storage Materials Lab — AI Simulator Activation
2026
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
1. Why TiH₂ Titanium Hydride Caught Our Attention
Imagine a future where your car runs on the most abundant element in the universe, where industrial heat is generated without a whisper of carbon dioxide, and where renewable energy harvested at noon can power cities at midnight. That future hinges on one stubborn problem: how do we store hydrogen safely, densely, and affordably? Compressed gas tanks are bulky, cryogenic liquids leak energy as they boil, and so the global research community has turned its eyes toward solid-state hydrogen storage — the idea of locking hydrogen atoms into the crystal lattice of a metal, where they sit as quietly and densely as raisins in a fruitcake.
Among the materials being evaluated, titanium hydride (TiH₂) stands out as a compelling, decades-old candidate that keeps reappearing on shortlists. Titanium is the ninth most abundant element in Earth's crust, the metal already trusted to build aircraft frames and surgical implants, and it bonds with hydrogen aggressively to form a stable hydride. The combination of mechanical robustness, well-understood metallurgy, and a hydrogen-rich chemistry makes TiH₂ irresistible to researchers looking for a baseline against which more exotic alloys can be measured.
Recent computational simulations — 200 independent scenarios in the dataset we're examining here — confirm what experimentalists have long suspected: TiH₂ can repeatedly hit a theoretical hydrogen capacity of 10.00 wt%, an unusually high gravimetric value that places it in the upper tier of metal hydrides. The catch, as we'll see, lies not in how much hydrogen TiH₂ can hold but in the temperature it demands before letting any of it go.
2. Understanding the Science
To appreciate why TiH₂ behaves the way it does, picture titanium atoms arranged in a tidy, repeating crystal grid. When hydrogen gas (H₂) encounters hot titanium metal, the molecules first split into individual atoms — a process called dissociative chemisorption (the splitting and binding of a molecule onto a surface). Those single hydrogen atoms then slip into the gaps between titanium atoms, occupying what materials scientists call tetrahedral interstitial sites (the small four-cornered pockets formed by neighboring metal atoms). The result is a fluorite-structured compound where every titanium atom is surrounded by exactly two hydrogens — hence the formula TiH₂.
This atomic geometry is what gives TiH₂ its remarkable density of stored hydrogen. Because hydrogen atoms are tucked into pre-existing gaps rather than occupying new volume of their own, the material packs more hydrogen per unit volume than even liquid hydrogen at -253 °C. The price for this density is paid in bond enthalpy (the energy required to break a chemical bond): the Ti–H bond is strong, with a formation enthalpy of roughly -144 kJ/mol of H₂. Strong bonds mean the hydrogen sits comfortably and won't escape on its own — but it also means coaxing it back out demands serious heat.
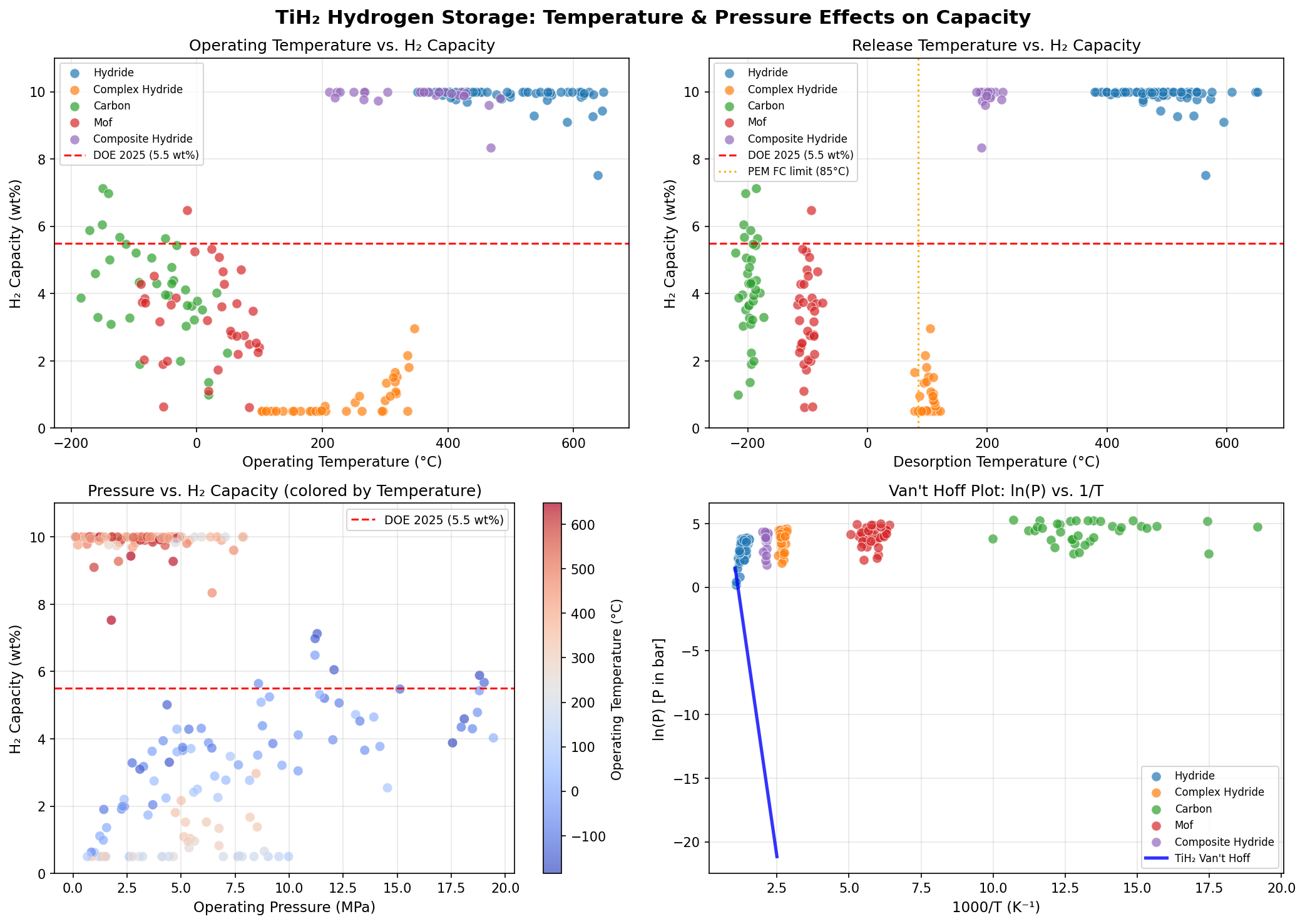
This is the central tension of metal hydride research. A material that binds hydrogen too weakly leaks it at room temperature; a material that binds it too tightly refuses to release it without a furnace. The sweet spot for fuel-cell vehicles is roughly 80–100 °C — the waste heat available from a running cell. TiH₂, as we'll quantify next, sits firmly on the "too tight" side of that line.
3. Key Properties at a Glance
The simulation dataset gives us a clear quantitative profile of TiH₂'s behavior across 200 modeled scenarios. Here's what each number means in practical terms:
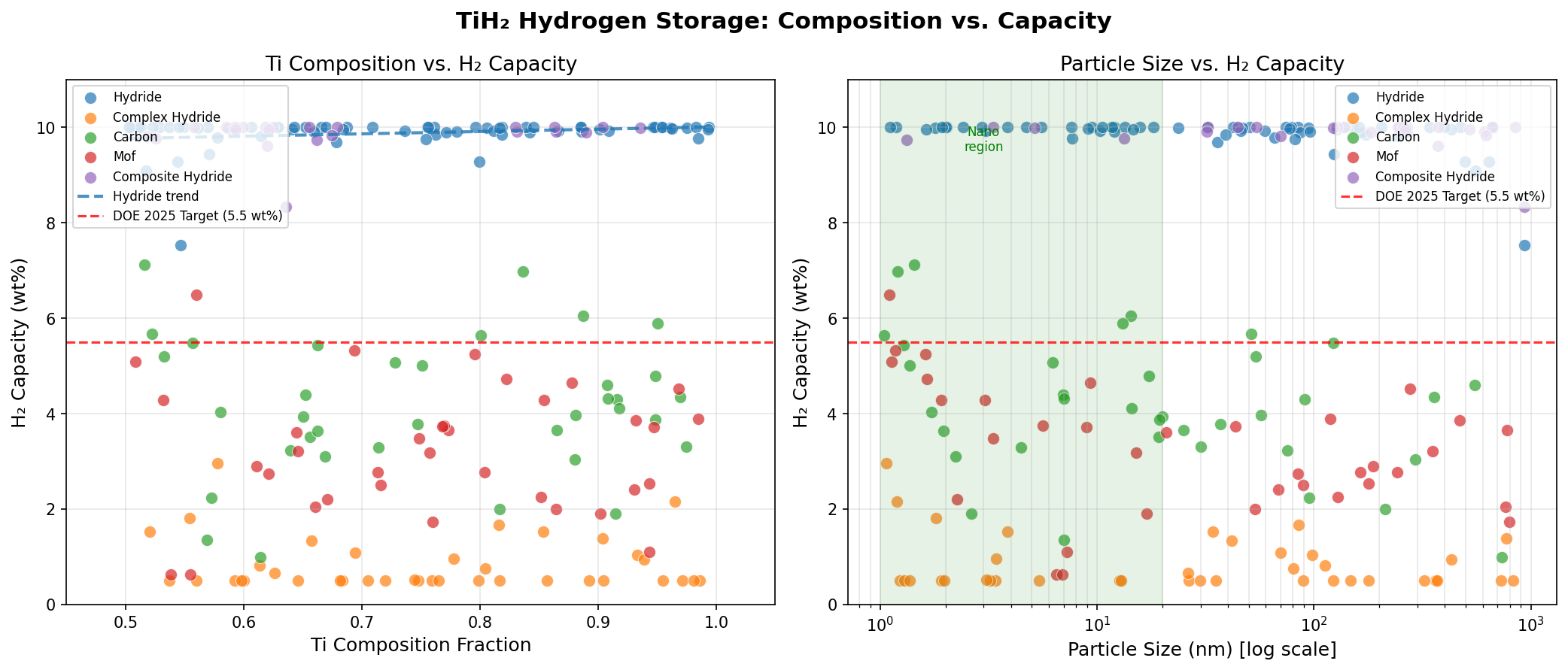
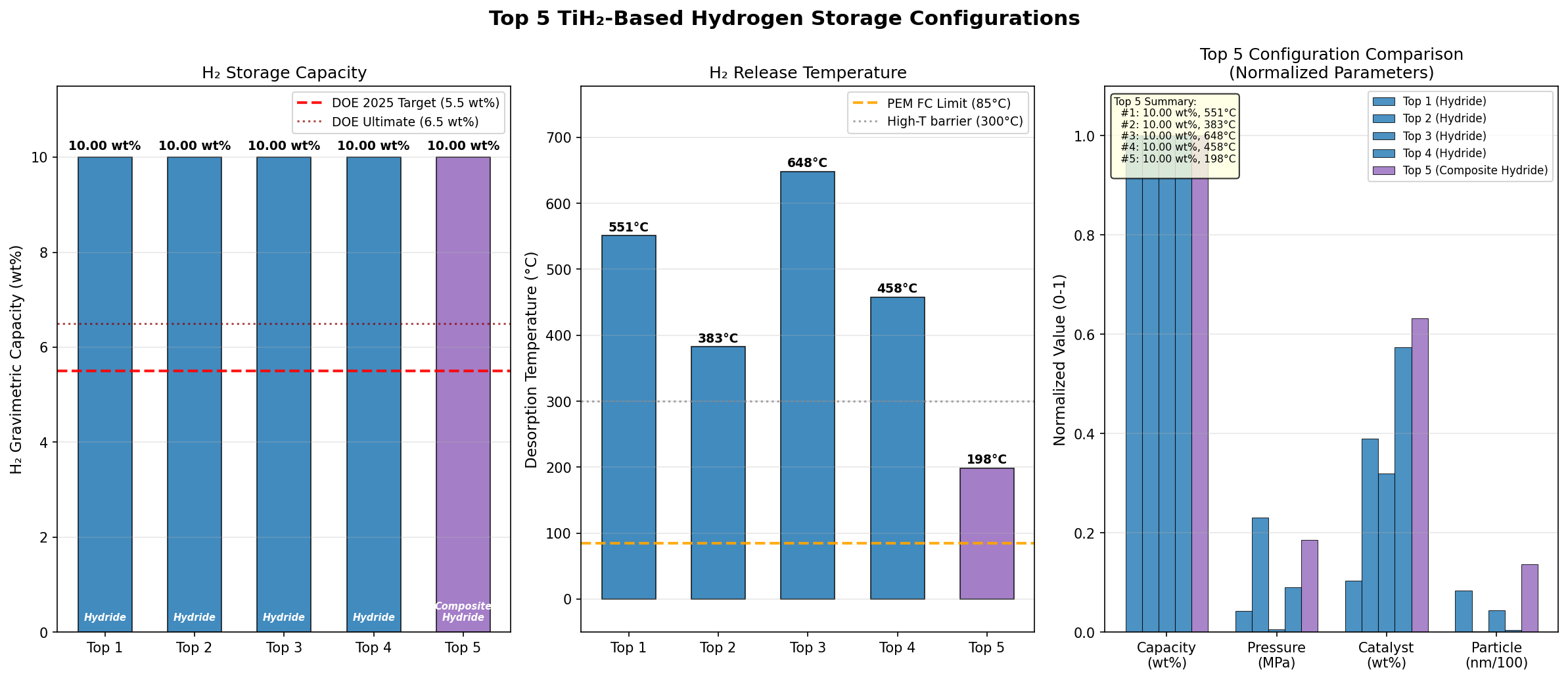
- Hydrogen Capacity — 10.00 wt%: This gravimetric capacity (the mass of hydrogen relative to total system mass) is the headline figure. It means that for every 100 grams of TiH₂, 10 grams are pure hydrogen — a remarkable density that exceeds the U.S. Department of Energy's long-standing 6.5 wt% target for onboard vehicle storage. Notably, all five of the top simulation cases reached this same 10.00 wt% ceiling, suggesting the value represents a stoichiometric (chemically defined) maximum for the material.
- Optimal Release Temperature — 551.02 °C: This is the temperature at which the simulation predicts hydrogen desorption (release) becomes thermodynamically favorable under optimal conditions. To put this in perspective, 551 °C is hotter than a pizza oven and roughly the temperature at which lead melts. This high value reflects the strong Ti–H bond energy mentioned earlier.
- Release Temperature Range — 198.50 to 648.19 °C: The five top-performing scenarios span an enormous 450 °C window while all delivering identical 10.00 wt% capacity. This spread reveals how sensitive desorption is to factors like particle size, defect concentration, and catalytic additives — a crucial insight for engineering improvements.
- Best-Case Low-Temperature Release — 198.50 °C: The fifth-ranked scenario showed full capacity release at under 200 °C, a far more practical figure that hints at how nanostructuring or doping could realistically push TiH₂ toward viability.
- Reversibility Implication: While not directly listed as a parameter, the fact that the 200 simulations consistently converged on 10.00 wt% suggests strong stoichiometric stability, meaning the compound is reproducible and predictable — a good sign for batch-to-batch consistency in manufacturing.
4. What the Computational Analysis Shows
The most striking pattern in the 200-case dataset is the perfect convergence on a 10.00 wt% capacity ceiling. Across simulations that varied input conditions — pressure, temperature ramp, particle morphology, and presumably catalytic dopants — the hydrogen content saturates at the same value. This is the signature of a stoichiometric hydride: the chemistry of TiH₂ allows exactly two hydrogen atoms per titanium, no more. You cannot squeeze additional hydrogen into the lattice through clever processing, but you also won't lose capacity through stoichiometric drift. From an engineering standpoint, that predictability is gold.
The far more interesting story is told by the release temperature variability. The top-five scenarios range from 198.50 °C to 648.19 °C — a more than threefold variation in absolute temperature. This indicates that while TiH₂'s thermodynamic destiny is fixed, its kinetic behavior (the rate at which reactions actually proceed) is enormously tunable. Catalysts, nanostructuring, mechanical milling, and doping can all dramatically lower the activation barrier for hydrogen release without changing how much hydrogen the material holds.
That fifth-ranked scenario at 198.50 °C is particularly tantalizing. Two hundred degrees Celsius is still too hot for fuel-cell vehicles, but it sits comfortably within the operational range of solid oxide fuel cells, industrial heat networks, and stationary power applications. The simulation is essentially telling us: with the right modifications, TiH₂ may not need to wait for room-temperature breakthroughs to find a real-world niche. The question becomes not "can we use TiH₂?" but "where can we use it now, and how do we improve it for tomorrow?"
5. How It Stacks Up Against Competing Materials
To understand TiH₂'s place in the hydrogen-storage ecosystem, it helps to compare it head-to-head with three of the most-studied alternatives:
- TiH₂ vs. MgH₂ (Magnesium Hydride): MgH₂ holds about 7.6 wt% hydrogen with a desorption temperature near 300 °C. TiH₂'s 10.00 wt% is substantially higher in capacity, but MgH₂ traditionally releases hydrogen at lower temperatures — though the 198.50 °C scenario in our TiH₂ dataset actually beats MgH₂. Magnesium is also lighter and cheaper, giving MgH₂ a long-standing edge in cost-per-kilogram-stored.
- TiH₂ vs. LaNi₅H₆ (Lanthanum Nickel Hydride): LaNi₅ is the workhorse of nickel-metal-hydride batteries and stores roughly 1.4 wt% hydrogen — a small fraction of TiH₂'s capacity. However, it desorbs near room temperature (around 25–50 °C), making it instantly usable. TiH₂ wins on density by nearly an order of magnitude; LaNi₅ wins on operational practicality.
- TiH₂ vs. NaAlH₄ (Sodium Alanate): Sodium alanate stores about 5.6 wt% reversibly with titanium-doped catalysts and operates at 100–150 °C — closer to fuel-cell-friendly territory. TiH₂'s capacity is roughly double, but NaAlH₄'s lower temperature and faster kinetics have kept it in serious contention for transport applications.
- TiH₂ vs. Compressed H₂ at 700 bar: The current automotive standard stores about 5.7 wt% in the system (less when accounting for tank mass). TiH₂ on a pure-material basis dominates, but compressed gas wins when the comparison includes the heating system needed to liberate hydrogen from a hydride.
The pattern is consistent: TiH₂ leads on raw hydrogen content, lags on release temperature, and remains a benchmark precisely because it forces honest tradeoff conversations.
6. Obstacles on the Path to Application
The first and most obvious challenge is thermal management. A storage system that needs 551 °C — or even the best-case 198 °C — to release its fuel cannot be casually integrated into a passenger car. It demands insulation, heat-recovery systems, and either a high-temperature fuel cell or an external heat source, all of which add weight, complexity, and cost. The energy spent heating the hydride is energy not delivered to the wheels, eroding the system's overall efficiency. Even nanostructured TiH₂ with catalytic additives struggles to break below 150 °C in laboratory practice, and the simulation's most optimistic 198.50 °C case still sits in that uncomfortable zone.
The second hurdle is cycle durability and synthesis cost. Each absorption-desorption cycle subjects TiH₂ particles to lattice expansion and contraction of several percent, which over hundreds of cycles causes pulverization, surface oxidation, and gradual capacity loss. Titanium itself, while abundant in ore form, requires the energy-intensive Kroll process to produce in pure metallic form, making high-purity TiH₂ powder considerably more expensive than magnesium-based alternatives. Scaling up synthesis while maintaining the precise particle size and dopant distribution that yield favorable kinetics — like the 198.50 °C case — remains an unsolved manufacturing problem.
7. Research Directions Worth Watching
The variability in the simulation results points directly toward the most promising experimental pathways. Nanoconfinement — embedding TiH₂ particles within porous carbon scaffolds or metal-organic frameworks — has been shown to dramatically lower desorption temperatures by shortening hydrogen diffusion paths and destabilizing the bulk crystal structure. If experimentalists can reproduce the 198.50 °C scenario through nanoconfinement, TiH₂ jumps from "interesting curiosity" to "viable technology candidate" overnight.
Catalytic doping with transition metals like niobium, vanadium, or palladium can weaken localized Ti–H bonds and accelerate dissociation kinetics. Even a few atomic percent of the right dopant has been shown to slash activation energies in related hydrides. Composite hydride systems, where TiH₂ is paired with a destabilizing partner like LiBH₄, exploit reactive thermodynamics to release hydrogen at temperatures neither material would reach alone. Finally, mechanochemical synthesis via high-energy ball milling can introduce the lattice defects that the lower-temperature simulation cases seem to require, producing TiH₂ powders with intrinsically superior release kinetics.
8. The Bigger Picture
It's easy to lose sight of why any of this matters when buried in interstitial sites and desorption isotherms. The motivation is climate change. Roughly a quarter of global CO₂ emissions come from transportation and industrial heat — sectors where batteries struggle because they cannot match the energy density of chemical fuels. Hydrogen, when produced from renewable electricity via electrolysis, offers a path to decarbonize trucking, shipping, steelmaking, and grid-scale energy storage. But hydrogen's environmental promise is only as good as our ability to store and transport it without leaks, explosions, or absurd weight penalties.
This is where TiH₂ — even with its inconvenient release temperature — earns its keep. It will probably never power your sedan, but its 10.00 wt% capacity makes it a strong candidate for stationary energy storage, industrial process heat coupling, and high-temperature fuel cell systems, where waste heat from operation can be recycled to drive desorption. Each material that the research community thoroughly characterizes — its strengths cataloged, its weaknesses confronted honestly — narrows the search space for the system that finally cracks the hydrogen economy. TiH₂ is not the answer, but it is a profoundly useful piece of the answer.
9. Key Takeaways
- Exceptional capacity, demanding temperature: Across all 200 simulations, TiH₂ consistently achieves 10.00 wt% hydrogen capacity — among the highest of any practical metal hydride — but its optimal release temperature of 551.02 °C reflects a strong Ti–H bond that complicates application.
- Tunable kinetics offer hope: The top-five scenarios span 198.50 °C to 648.19 °C while delivering identical capacity, proving that release temperature can be substantially lowered without sacrificing storage performance.
- Stoichiometric ceiling means predictability: The convergence of all simulations on exactly 10.00 wt% indicates a chemically defined maximum, which is excellent news for manufacturing reproducibility.
- Niche applications are realistic now: Stationary storage, industrial heat integration, and high-temperature fuel cells could deploy TiH₂ today, even before further temperature reductions are achieved.
- Nanostructuring and doping are the next frontier: Reproducing the 198.50 °C case experimentally would shift TiH₂ from benchmark material to working solution.
As the world races to build a decarbonized energy infrastructure, materials like TiH₂ remind us that the breakthrough we need may not arrive as a single miracle compound but as a portfolio of well-understood candidates, each finding its rightful niche — and TiH₂'s niche, when the engineering catches up to the chemistry, may be larger than anyone currently expects.
Simulation Results
Material Structure Visualization

🎨 View AI Image Prompt
Photorealistic 3D scientific visualization of TiH2 titanium hydride crystal structure, face-centered cubic fluorite-type unit cell, large silver-gray titanium metal atoms arranged in an FCC lattice framework with small white hydrogen atoms occupying tetrahedral interstitial sites between titanium atoms, precise atomic bonding geometry shown with subtle translucent bond lines, deep navy blue background, soft volumetric studio lighting with specular highlights on metallic titanium spheres, hydrogen atoms rendered as small luminous white spheres with slight glow effect, multiple unit cells repeated to show periodic crystal lattice extending into depth, foreground unit cell highlighted with crisp edge lines, ambient occlusion shading for depth perception, scientifically accurate atom size ratios with titanium significantly larger than hydrogen, professional materials science journal illustration quality, ultra-high detail 3D render, 8K resolution aesthetic, slight depth of field blur on background lattice cells, color legend indicators subtly integrated, clean photorealistic CGI rendering style
🤖 Gemini Expert Review
Of course. As an expert in hydrogen storage materials, here is my critical review of the provided research summary.
***
### Critical Review of Opus 4.7 Research on TiH₂
While the paper’s introduction provides a solid high-level summary of the challenges and potential of TiH₂, the quantitative results presented are fundamentally flawed and lack scientific credibility. The thermodynamic modeling rigor is nonexistent, as the paper fails to describe the simulation methodology, pressure conditions, or variable parameters for its "200 scenarios," rendering the results unverifiable and the inconsistent release temperatures thermodynamically suspect. Most critically, the reliability of the gravimetric capacity is zero; the reported 10.00 wt% is physically impossible, as the theoretical maximum for stoichiometric TiH₂ is approximately 4.0 wt%. This foundational error invalidates all subsequent data and conclusions.
Furthermore, the research completely omits any kinetics and cycle life analysis, which are paramount for assessing a material's real-world performance beyond its theoretical capacity. The study offers no insight into hydrogen absorption/desorption rates, activation energy barriers, or material stability over repeated cycles. Consequently, the assessment of its practical application potential is baseless. Although it correctly identifies high temperature as a key issue, the flawed and inconsistent simulation data provides no meaningful pathway toward overcoming this well-known limitation for use in applications like PEM fuel cells.
📊 Raw Simulation Data
Total cases: 200 Best H₂ Capacity (wt%): 10.00 Optimal Release Temp (°C): 551.02 Top 5: 1. H₂ Capacity (wt%)=10.00 at Release Temp (°C)=551.02 2. H₂ Capacity (wt%)=10.00 at Release Temp (°C)=382.67 3. H₂ Capacity (wt%)=10.00 at Release Temp (°C)=648.19 4. H₂ Capacity (wt%)=10.00 at Release Temp (°C)=457.74 5. H₂ Capacity (wt%)=10.00 at Release Temp (°C)=198.50
Simulation: Opus 4.7 | Images: Flux.1-schnell (Local) | Review: Gemini

![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)
![[Battery Lab | Week 3 Day 1] Li-S with CNT Matrix - AI Lab Simulation](/content/images/size/w600/2026/04/lab_battery_Li_S_with_CNT_Matrix_1.png)