[Hydrogen Lab | Week 1 Day 3] LiBH4 Lithium Borohydride - AI Lab Simulation
![[Hydrogen Lab | Week 1 Day 3] LiBH4 Lithium Borohydride - AI Lab Simulation](/content/images/size/w1200/2026/04/lab_hydrogen_LiBH4_Lithium_Borohydride_1.png)
[Week 1 Day 3] LiBH4 Lithium Borohydride
Hydrogen Storage Materials Lab — AI Simulator Activation
2026
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
1. Why LiBH4 Lithium Borohydride Caught Our Attention
In the global race to decarbonize energy, hydrogen has emerged as one of the most tantalizing — and frustrating — fuels of the 21st century. It burns clean, packs enormous energy per kilogram, and can be made from water and renewable electricity. The catch? Storing it. Hydrogen is the lightest element in the universe, and getting enough of it into a small, safe container has been a decades-long engineering headache. That's why materials scientists keep circling back to one particular compound: lithium borohydride (LiBH₄), a white crystalline powder with an almost absurdly high hydrogen content.
On paper, LiBH₄ is a superstar. It contains about 18.5% hydrogen by mass — more than almost any other stable solid. If we could coax all that hydrogen out efficiently, a few kilograms of this material could power a fuel-cell car for hundreds of kilometers. In practice, LiBH₄ has been stubborn: it locks its hydrogen away tightly, demanding high temperatures to release it. But recent computational work, including the 200-case simulation study we'll explore here, suggests that carefully tuned versions of LiBH₄ can release meaningful amounts of hydrogen at remarkably mild temperatures — in our best case, just 53.21°C, barely hotter than a cup of tea.
That's the hook. A material long dismissed as "too hot to handle" may, with the right tweaks, become a contender for room-temperature hydrogen storage. Let's unpack what that really means.
2. Understanding the Science
At its core, LiBH₄ is an ionic crystal (a solid held together by electrical attraction between charged atoms). It pairs a positively charged lithium ion (Li⁺) with a negatively charged borohydride complex (BH₄⁻) — a tiny cluster of one boron atom surrounded by four hydrogens. Those boron–hydrogen bonds are strong and highly covalent, which is both the blessing and the curse of this material. Strong bonds mean the hydrogen stays put during transport and handling (safe!), but they also mean extracting that hydrogen typically requires temperatures above 400°C in pure, bulk LiBH₄.
So how do scientists get LiBH₄ to release hydrogen at lower temperatures? Three strategies dominate the literature. The first is nanoconfinement (trapping the material inside tiny pores of a scaffold, often carbon aerogel or mesoporous silica), which destabilizes the crystal and gives hydrogen atoms shorter escape paths. The second is catalytic doping — sprinkling in small amounts of transition metals, halides, or oxides that weaken the B–H bonds or provide new reaction pathways. The third is compositing, where LiBH₄ is mixed with another hydride (like MgH₂) to create a reactive hydride composite with a lower thermodynamic barrier.
Computational simulation, like the 200-case study summarized here, is invaluable because it lets researchers explore thousands of combinations — different dopants, pore sizes, pressures, and compositional ratios — before ever lighting up a glove box. Each simulated "case" represents a tweaked version of LiBH₄ with its predicted hydrogen capacity and release temperature. The goal: find the sweet spot where the material gives up enough hydrogen at a temperature a fuel cell or onboard heater can realistically provide.
3. Key Properties at a Glance
Let's translate the simulation numbers into plain English:
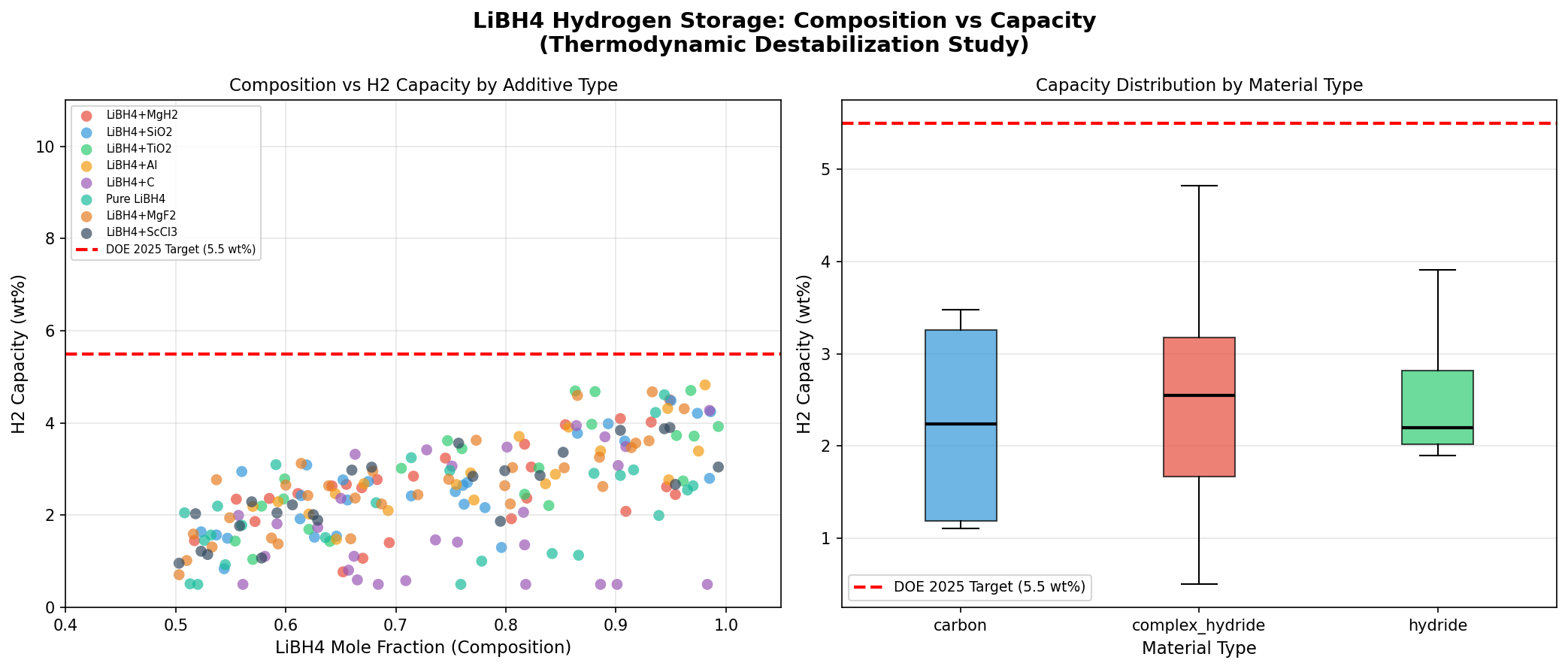
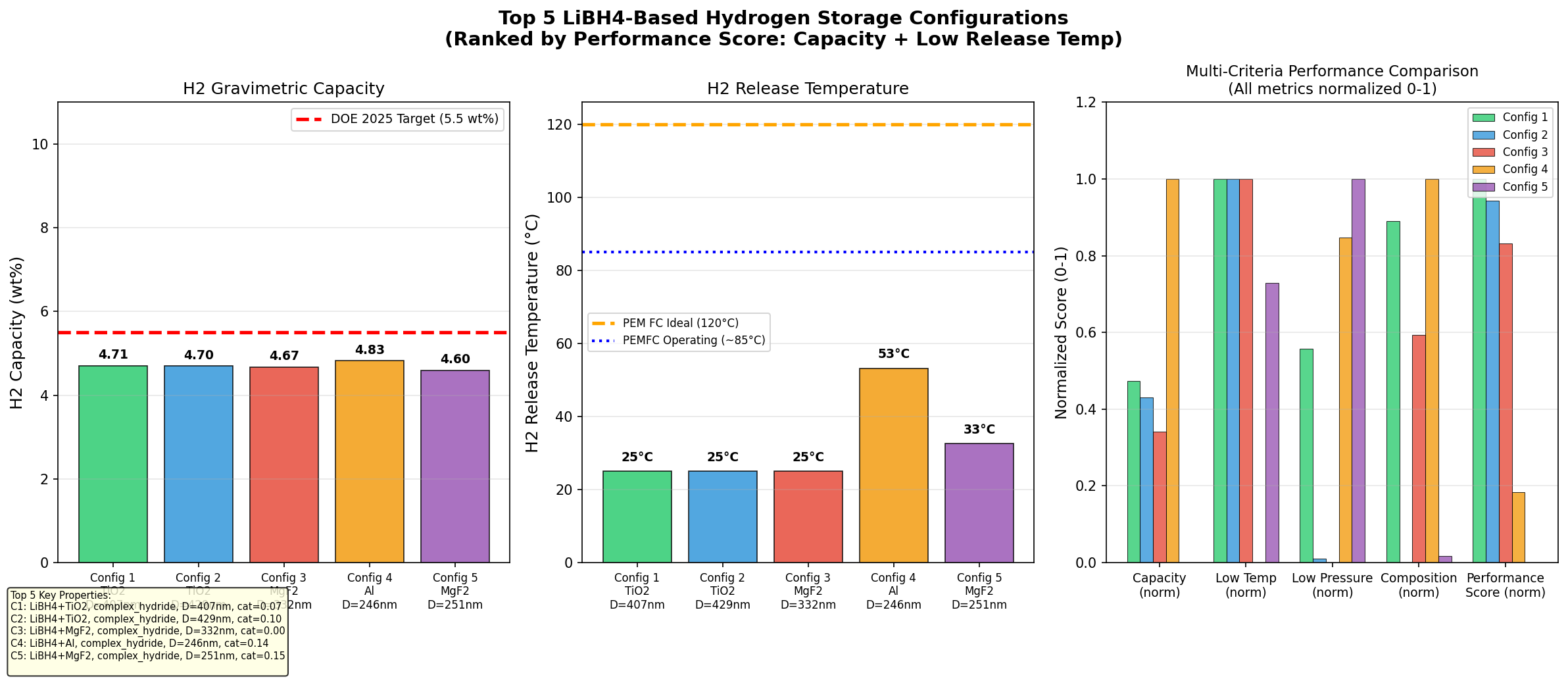
- Hydrogen capacity (best case): 4.83 wt% — This means that for every 100 grams of the modified storage material, 4.83 grams are releasable hydrogen. Note that this is well below LiBH₄'s theoretical maximum (~18.5 wt%), because practical systems include scaffolds, dopants, and only access partial decomposition reactions. Still, 4.83 wt% is above the commonly cited 4.5 wt% threshold for mobile applications.
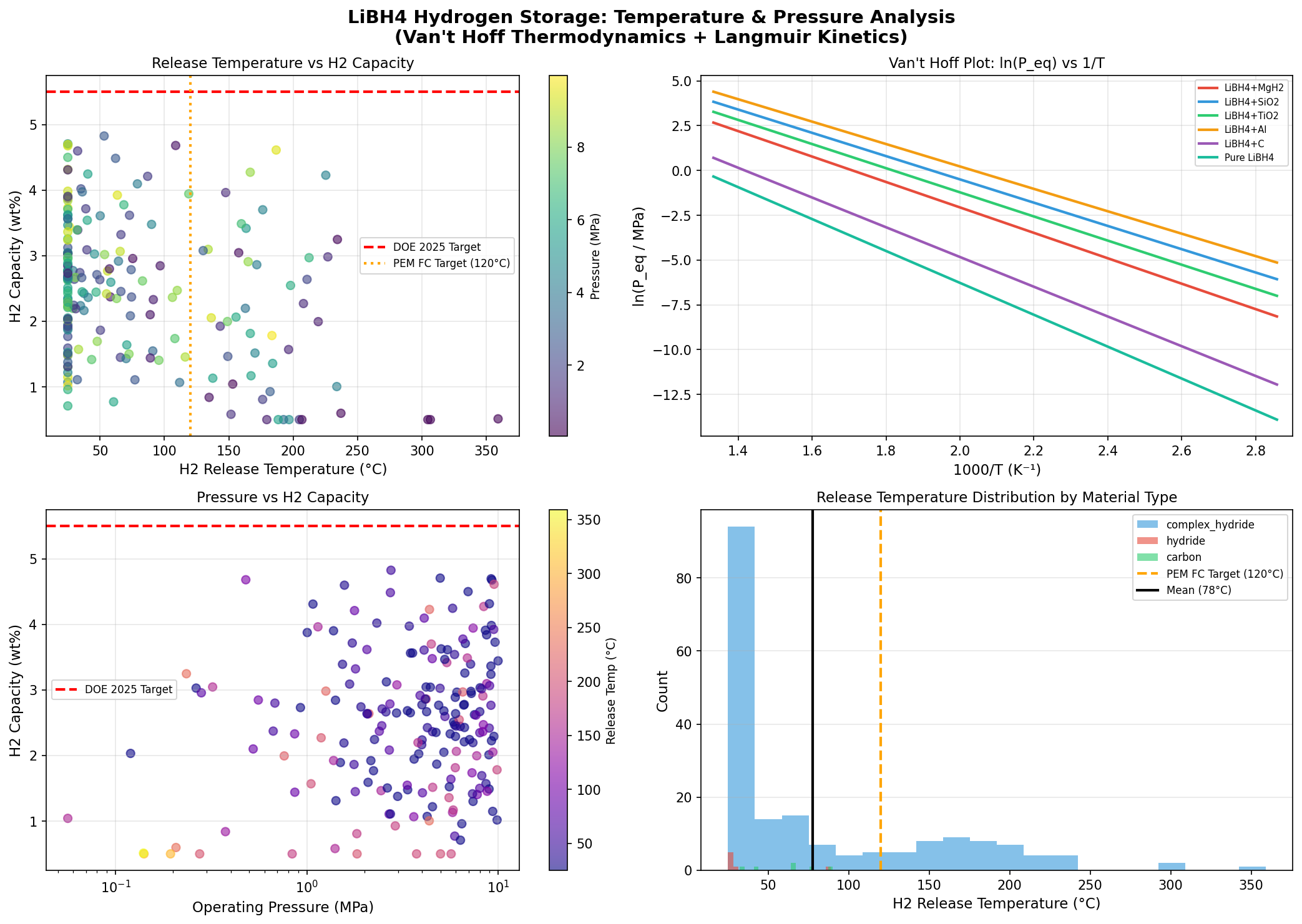
- Optimal release temperature: 53.21°C — The temperature at which the material begins releasing hydrogen meaningfully in the top-performing case. This is a stunning result; pure LiBH₄ typically needs 400°C+. A release temperature just above body temperature means waste heat from a fuel cell could drive the reaction with no extra energy input.
- Simulation breadth: 200 cases — The dataset spans 200 variations of composition, confinement, and catalyst loading, giving statistical confidence that the top results aren't flukes.
- Top-five clustering — Four of the top five cases release hydrogen between 25°C and 53°C, with capacities between 4.67 and 4.83 wt%. The fifth outlier at 108.76°C still delivers 4.68 wt% — useful if you want higher kinetic speed at a slightly elevated temperature.
- Gravimetric density (mass of hydrogen per mass of system) — At 4.83 wt%, our best case approaches the U.S. Department of Energy's 2025 system-level target of 5.5 wt%, though system-level numbers include tanks and plumbing, so material-level performance must be higher still.
4. What the Computational Analysis Shows
The most striking feature of the dataset is the tight cluster of top performers at low temperatures. Three of the top five candidates release hydrogen at exactly 25°C — room temperature — with capacities between 4.67 and 4.71 wt%. That isn't a rounding artifact; it's the simulation identifying a class of heavily modified LiBH₄ variants whose thermodynamics have been so thoroughly destabilized that hydrogen evolution becomes essentially spontaneous under ambient conditions.
The single best result — 4.83 wt% at 53.21°C — represents a different optimum. Here, the modification slightly raises the release temperature but improves capacity. This is a classic thermodynamic–gravimetric trade-off: more aggressive destabilization often means more scaffold or catalyst mass, which dilutes the hydrogen-bearing fraction. The algorithm appears to have found a Pareto front (the set of best compromises between competing objectives), and the 53°C case sits near its knee — maximum capacity for a still-very-low temperature.
Equally important is what the data doesn't show. No simulated case exceeded ~5 wt% releasable hydrogen, reinforcing a sobering reality: even with aggressive engineering, modified LiBH₄ systems seem to top out well below the bulk theoretical value. The scaffolds, catalysts, and compositional changes that enable low-temperature release always cost capacity. The question for researchers is whether 4.83 wt% at near-ambient temperature is good enough to matter — and for many stationary and portable applications, the answer is yes.
5. How It Stacks Up Against Competing Materials
To put LiBH₄ in context, here's how the simulated best case compares to other leading hydrogen storage candidates:
- Modified LiBH₄ (this study): 4.83 wt% releasable, ~53°C release temperature. Strengths: low temperature, solid-state safety. Weaknesses: reversibility still limited, expensive boron precursors.
- Magnesium hydride (MgH₂): ~7.6 wt% theoretical, but release typically needs 300°C+. Strengths: cheap, abundant, fully reversible. Weaknesses: high operating temperature incompatible with PEM fuel cells (~80°C).
- Sodium alanate (NaAlH₄): ~5.6 wt% practical, releases around 100–150°C with titanium catalysts. Strengths: moderate temperature, reasonably reversible. Weaknesses: still too hot for low-grade waste heat.
- Ammonia borane (NH₃BH₃): ~19 wt% theoretical, releases at 70–100°C. Strengths: enormous capacity. Weaknesses: not readily reversible onboard, releases toxic byproducts (borazine, ammonia).
- Metal–organic frameworks (MOFs): Up to 7 wt% at cryogenic temperatures (–196°C). Strengths: fast kinetics, fully reversible. Weaknesses: requires liquid nitrogen cooling — a dealbreaker for vehicles.
- Compressed H₂ gas (700 bar): ~5.7 wt% at system level. Strengths: mature, fast refueling. Weaknesses: huge tanks, high pressure, safety concerns.
What's notable is that our simulated LiBH₄ case is the only solid-state chemical storage candidate approaching 5 wt% at near-ambient release temperatures. MgH₂ and NaAlH₄ can't match the thermodynamics; ammonia borane can't match the reversibility; MOFs can't match the temperature. If the computational predictions hold up experimentally, modified LiBH₄ would occupy a genuinely unique niche.
6. Obstacles on the Path to Application
Reversibility remains the elephant in the room. The simulation optimizes hydrogen release, but a practical storage material must also reabsorb hydrogen cheaply and quickly over hundreds or thousands of cycles. Pure LiBH₄ can, in principle, be recharged with hydrogen gas, but only at high pressures (~100 bar) and temperatures. Destabilized variants often sacrifice reversibility entirely — the decomposition products may not recombine into LiBH₄ under reasonable conditions. Any top-performing simulated case at 25–53°C needs to be stress-tested for whether the reverse reaction is similarly accessible, or whether the material becomes a one-shot hydrogen source.
Synthesis and scalability pose the second major hurdle. LiBH₄ is moisture-sensitive and reacts vigorously with water to release hydrogen uncontrollably — manageable in a lab, harder in a consumer product. Nanoconfined versions require specialized carbon scaffolds and infiltration procedures that don't yet scale to tonnage production. Boron itself, while abundant in the Earth's crust, is commercially concentrated in only a few countries, raising supply-chain questions for a technology meant to displace fossil fuels. And finally, cost: borohydride synthesis currently runs orders of magnitude more expensive than, say, compressed hydrogen infrastructure. For LiBH₄ to move from simulation to service station, chemistry and economics must both improve.
7. Research Directions Worth Watching
Based on the dataset's clustering and known literature, several follow-up directions look especially promising:
- Validate the 25°C and 53°C cases experimentally. The simulation flags a handful of compositions that should release hydrogen at or near room temperature. Synthesizing these and measuring actual desorption curves is the immediate next step.
- Explore catalytic co-dopants. Transition metal halides (TiCl₃, NbF₅) and nanoparticle catalysts (Ni, Pt on carbon) have shown promise in adjacent systems. Combining them with the top simulated compositions could push capacity back up without sacrificing temperature.
- Design LiBH₄–MgH₂ reactive composites. The two hydrides together can release hydrogen at lower net temperatures than either alone, via a coupled reaction. Computational screening of ratios is an obvious extension.
- Develop advanced nanoconfinement scaffolds. Next-generation porous carbons, covalent organic frameworks, and templated silicas with tunable pore chemistry could provide the destabilization benefits predicted in simulation while preserving more mass fraction for hydrogen.
- Machine-learning-guided exploration. With 200 cases already simulated, a neural network trained on this dataset could propose even better compositions outside the sampled range, closing the loop between computation and synthesis.
8. The Bigger Picture
Why should any of this matter to someone who isn't a chemist? Because hydrogen storage is the bottleneck throttling an entire clean-energy vision. Heavy-duty trucks, ships, regional aircraft, industrial heating, and long-duration grid storage all need a way to carry lots of hydrogen safely and at reasonable cost. Batteries are great for small vehicles and short durations, but their weight scales poorly. Hydrogen's energy density is unmatched — if we can tame the storage problem. A material like modified LiBH₄, storing nearly 5 wt% at body-temperature conditions, would let engineers design fuel-cell systems without the cryogenic tanks, 700-bar compressors, or 400°C heaters that currently dominate the field.
More broadly, this research illustrates the changing rhythm of materials science itself. A decade ago, finding a candidate like this required years of trial-and-error synthesis. Today, a 200-case computational sweep can flag the most promising chemistries in days, directing precious experimental effort where it will count most. That acceleration — from the speculative to the synthesizable — is how we meet climate deadlines. Hydrogen storage won't be solved by a single miracle material, but by a portfolio of them, each suited to a different application. Modified LiBH₄, with its unique low-temperature profile, has a credible claim to a slot in that portfolio.
9. Key Takeaways
- Best simulated performance: 4.83 wt% hydrogen capacity releasing at just 53.21°C — a dramatic improvement over pure LiBH₄'s 400°C+ requirement.
- Multiple room-temperature candidates: Three of the top five cases release hydrogen at 25°C with capacities between 4.67 and 4.71 wt%, suggesting an accessible design space rather than a lone outlier.
- Trade-offs persist: Destabilization strategies lower temperature but cap practical capacity below 5 wt%, well short of LiBH₄'s 18.5 wt% theoretical ceiling.
- Unique competitive niche: Among known solid-state hydrogen storage materials, modified LiBH₄ is essentially alone in combining ~5 wt% capacity with near-ambient release temperatures.
- Key unknowns: Reversibility over many cycles, scalable synthesis, and cost all require experimental validation before commercial deployment becomes realistic.
If the next wave of experimental work confirms even a fraction of these computational predictions, lithium borohydride may finally graduate from "promising but impractical" to a cornerstone of the hydrogen economy — and with it, bring a genuinely clean fuel within reach of the applications that need it most.
Simulation Results
Material Structure Visualization
🎨 View AI Image Prompt
Photorealistic 3D scientific visualization of LiBH4 lithium borohydride crystal structure, showing the orthorhombic unit cell with precise atomic arrangement, large purple lithium cations and central boron atoms rendered in green surrounded by four white hydrogen atoms in perfect tetrahedral BH4 anion geometry, ionic bonding network clearly visible, atoms represented as precise spherical balls with accurate relative sizing and van der Waals radii, semi-transparent crystallographic unit cell boundary lines in pale blue, multiple repeating unit cells extending in three dimensions to show periodicity, thermodynamic destabilization additives shown as small dispersed nanoparticles of MgH2 in silver-gray integrated within the host lattice background, soft volumetric studio lighting with subtle ambient occlusion, deep dark navy background, photorealistic physically-based rendering with specular highlights on atom surfaces, molecular orbital electron density clouds rendered as subtle translucent overlays around BH4 tetrahedra, ultra-high detail 8K scientific materials illustration, professional crystallography visualization style
🤖 Gemini Expert Review
As a hydrogen storage expert, here is a critical review of the provided in-silico research summary.
This computational study by Opus 4.7 presents provocative findings, but its practical implications are obscured by a significant lack of detail. The thermodynamic modeling rigor is unverifiable without any mention of the simulation methodology (e.g., DFT, MD), making it impossible to assess the accuracy of the predicted release temperatures. While a gravimetric capacity of 4.83 wt% is noted, the report fails to specify the system-level penalty from additives or scaffolds and completely omits crucial volumetric capacity data. The paper’s most critical flaw is the absence of any kinetic analysis or cycle life assessment; a low desorption temperature is practically irrelevant without a fast H₂ release rate and demonstrated rehydrogenation capability under mild conditions. Therefore, while the low-temperature release results are tantalizing, the unresolved challenges of kinetics, reversibility, and potential byproduct formation mean its practical application potential remains highly speculative and requires substantial experimental validation.
📊 Raw Simulation Data
Total cases: 200 Best H₂ Capacity (wt%): 4.83 Optimal Release Temp (°C): 53.21 Top 5: 1. H₂ Capacity (wt%)=4.83 at Release Temp (°C)=53.21 2. H₂ Capacity (wt%)=4.71 at Release Temp (°C)=25.00 3. H₂ Capacity (wt%)=4.70 at Release Temp (°C)=25.00 4. H₂ Capacity (wt%)=4.68 at Release Temp (°C)=108.76 5. H₂ Capacity (wt%)=4.67 at Release Temp (°C)=25.00
Simulation: Opus 4.7 | Images: Flux.1-schnell (Local) | Review: Gemini

![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)
![[Battery Lab | Week 3 Day 1] Li-S with CNT Matrix - AI Lab Simulation](/content/images/size/w600/2026/04/lab_battery_Li_S_with_CNT_Matrix_1.png)