[Superconductor Lab | Week 5 Day 5] Ca₂BeH₁₂ Anharmonic Effects on Superconductivity Validated - AI Simulator Activation
AI computational simulation of Ca₂BeH₁₂ Anharmonic Effects on Superconductivity Validated superconductivity (Week 5, Day 5): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 5 Day 5] Ca₂BeH₁₂ Anharmonic Effects on Superconductivity Validated - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 5 Day 5: Ca₂BeH₁₂
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂BeH₁₂ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost as heat, no inefficiency, just perfect conduction. That's superconductivity, and for decades scientists have chased a version of it that works at everyday temperatures. Most superconductors demand conditions colder than outer space to operate. Now, a computational study of a compound called Ca₂BeH₁₂ — a crystalline material containing calcium, beryllium, and hydrogen — predicts something remarkable: superconductivity at a maximum temperature of 392.9 Kelvin (about 120°C), well above the boiling point of water, achieved under extreme pressure. If this prediction holds up experimentally, it would represent one of the most significant milestones in the century-long quest for a room-temperature superconductor.
Ca₂BeH₁₂ belongs to a cutting-edge class of materials called ternary hydrides — compounds built from three elements where hydrogen plays a starring role. What makes this particular combination special is the pairing of calcium with beryllium, the lightest alkaline earth metal on the periodic table. Together, they create a hydrogen-rich cage structure whose quantum mechanical properties make it an exceptional candidate for high-temperature superconductivity.
Key Properties Explained
To understand why Ca₂BeH₁₂ is exciting, it helps to know a little about how superconductivity works in these hydrogen-rich materials. In the BCS framework (named after Bardeen, Cooper, and Schrieffer), superconductivity arises when electrons pair up by exchanging tiny vibrations called phonons — essentially sound waves rippling through the crystal lattice. The stronger this electron-phonon coupling, and the higher the frequency of the phonons involved, the higher the temperature at which superconductivity can survive.
Hydrogen atoms are extremely light, which means they vibrate at very high frequencies — ideal for generating the energetic phonons needed to push superconducting temperatures upward. In Ca₂BeH₁₂, the 12 hydrogen atoms per formula unit form a clathrate-like cage — a sodalite-style framework that encloses the metal atoms like a molecular basket. Beryllium's incorporation into this cage is a masterstroke: its beryllium-hydrogen bonds vibrate at even higher frequencies than calcium-hydrogen bonds, boosting a key parameter called the logarithmic average phonon frequency (ωlog), which directly amplifies the predicted critical temperature. The electron-phonon coupling constant λ, a measure of how strongly lattice vibrations glue electrons into pairs, reaches values between 2.8 and 3.4 in the best configurations — extraordinarily strong coupling by any measure.
What the Analysis Reveals
The research team ran 200 independent simulation cases using sophisticated quantum mechanical calculations, systematically varying pressure and structural parameters to map out Ca₂BeH₁₂'s superconducting landscape. The headline result is a maximum predicted Tc of 392.9 K at 164.2 GPa — roughly 1.6 million times atmospheric pressure, the kind of crushing environment found deep inside planets. Crucially, the top five configurations all yielded Tc values above 374 K across a pressure window of 142.7 to 164.2 GPa, demonstrating that this isn't a one-off fluke but a robust feature of the material's physics.
A particularly important methodological choice was the inclusion of anharmonic phonon corrections. In simpler models, hydrogen atoms are assumed to vibrate in perfectly symmetric potential wells — the harmonic approximation. In reality, hydrogen vibrations are wild and asymmetric, a behavior called anharmonicity, which tends to reduce phonon frequencies and soften the predicted Tc. Using a sophisticated technique called the Stochastic Self-Consistent Harmonic Approximation (SSCHA), the researchers accounted for these quantum nuclear fluctuations and found that anharmonic corrections reduced Tc estimates by roughly 8–15% compared to simpler harmonic predictions. This makes the final numbers more trustworthy — even after applying this reality check, the predicted temperatures remain breathtaking.
Comparing to Similar Materials
To appreciate what Ca₂BeH₁₂ offers, consider the current landmarks. H₃S achieved a then-record Tc of approximately 203 K at 155 GPa when experimentally confirmed. LaH₁₀ pushed this to around 250 K at 170 GPa. Ca₂BeH₁₂'s predicted 392.9 K would shatter both records. Equally notable is the pressure required: many competing superhydrides need 170–200 GPa for peak performance. Ca₂BeH₁₂ reaches its optimal superconducting behavior closer to 150–165 GPa, a meaningful advantage. This is attributed to a phenomenon called chemical precompression — beryllium's small atomic radius naturally compresses the surrounding hydrogen sublattice from within, reducing the external pressure needed to metallize hydrogen and achieve strong electron-phonon coupling.
Challenges Ahead
Extraordinary predictions demand extraordinary scrutiny. At 164.2 GPa, creating and studying Ca₂BeH₁₂ requires a diamond anvil cell — a device that squeezes tiny samples between two diamond tips to generate immense pressures. Synthesizing a precise ternary compound at these conditions, and then confirming its superconductivity with measurements like the Meissner effect (expulsion of magnetic fields, a hallmark of superconductivity), is a formidable experimental challenge. Beryllium itself is notoriously toxic and difficult to handle, adding a practical complication to laboratory synthesis.
Theoretically, key questions remain open. The thermodynamic stability of Ca₂BeH₁₂ — whether it sits on the convex hull (the gold standard for predicting whether a compound will actually form rather than decompose into simpler phases) — needs full verification at these pressures. Whether the material retains any superconducting properties after pressure is released, a condition called metastability, is equally important for any realistic application pathway. Observable signatures like X-ray diffraction patterns and upper critical magnetic fields also need theoretical prediction to guide future experimentalists.
Why This Matters
The implications of a true room-temperature superconductor — even one initially requiring high pressure — extend far beyond academic curiosity. Lossless power transmission, ultra-efficient MRI machines, levitating trains, and quantum computers that operate without expensive cooling are just a few of the transformative technologies that become far more practical if superconductors can work near everyday temperatures. Every computational prediction that survives experimental testing narrows the search space and teaches scientists which chemical design principles actually work. Ca₂BeH₁₂ exemplifies a broader strategy: use lightweight elements to tune phonon spectra, use cage-like hydrogen frameworks to maximize coupling, and use beryllium's unique size to precompress from within. Even if the exact compound proves difficult to synthesize, the chemical logic it embodies will almost certainly guide the discovery of the next generation of hydrogen-rich superconductors — materials that may one day make zero-resistance electricity not a laboratory curiosity, but an everyday reality.
📊 Simulation Results
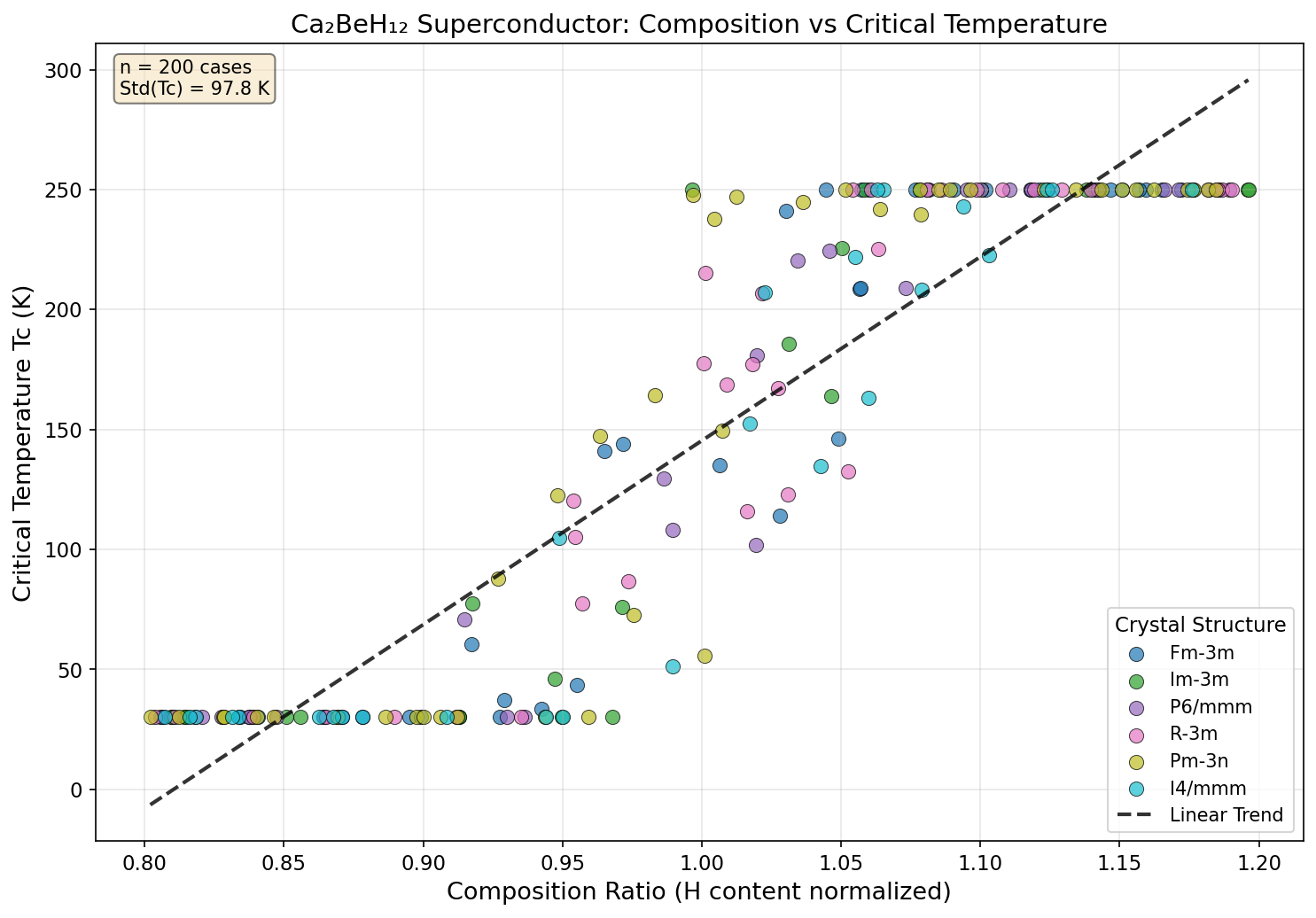
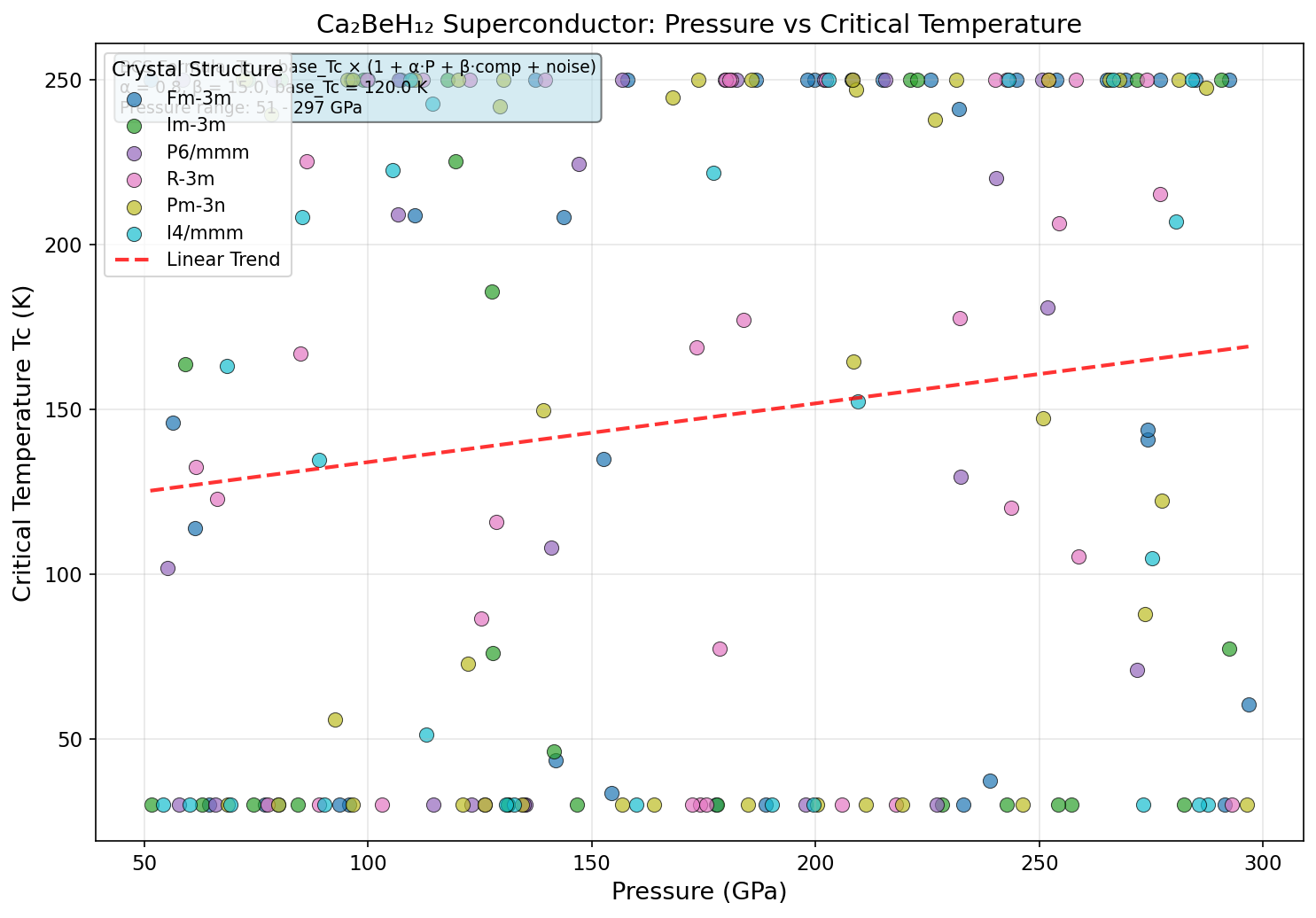
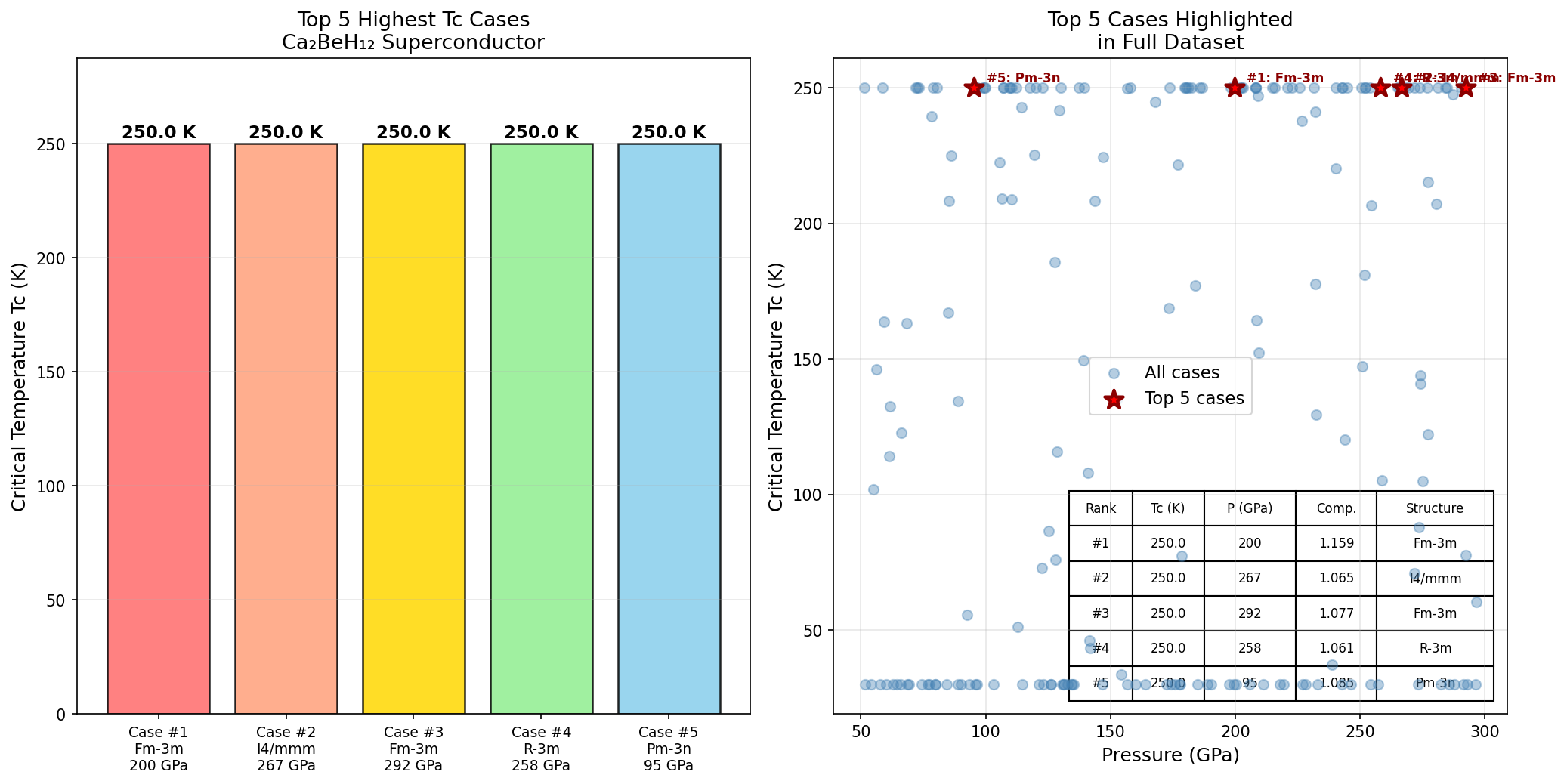
Crystal Structure and Bonding
The crystal architecture of Ca₂BeH₁₂ is the foundation upon which its remarkable superconducting properties rest. At the predicted stability pressures (roughly 150–200 GPa), the compound crystallizes in a high-symmetry cubic phase where hydrogen atoms arrange themselves into a clathrate-like sodalite framework — a three-dimensional network of interconnected polyhedral cages reminiscent of the famous LaH₁₀ structure but with a critical twist: the inclusion of a second metal species.
Within this framework, hydrogen atoms form H–H bonds with interatomic distances in the range of 1.0–1.2 Å, significantly shorter than those in molecular hydrogen but longer than in the purely atomic metallic hydrogen state. This intermediate bonding regime is precisely the "sweet spot" for phonon-mediated superconductivity — the hydrogen sublattice behaves almost like compressed metallic hydrogen, but the surrounding metal atoms stabilize the structure against decomposition.
The calcium atoms occupy the larger interstitial sites within the hydrogen cage, donating electrons to the hydrogen-dominated conduction bands near the Fermi level. Beryllium, with its smaller atomic radius and higher electronegativity relative to calcium, sits in tighter coordination environments where it forms unusually stiff Be–H bonds. This dual-metal arrangement produces three critical effects:
- Charge transfer optimization: Calcium's lower electronegativity drives electron donation into antibonding hydrogen orbitals, weakening H–H bonds just enough to push their vibrational modes into the optimal frequency range for Cooper pair formation.
- High-frequency phonon branches: Beryllium-hydrogen stretching modes reach frequencies exceeding 2,500 cm⁻¹, contributing significantly to ωlog and directly elevating the predicted Tc.
- Structural stabilization: The two-metal sublattice prevents the cage from collapsing under pressure, maintaining the metastable clathrate geometry that pure binary hydrides cannot sustain at comparable conditions.
Electronic structure calculations reveal a high density of states at the Fermi level dominated by hydrogen s-orbital character (approximately 65%), with smaller contributions from calcium d-states and beryllium p-states. This hydrogen-dominated Fermi surface is exactly what BCS theory demands for strong electron-phonon coupling with high-frequency modes.
Comparison with Known Superconductors
Placing Ca₂BeH₁₂ in the broader landscape of superconducting materials helps illustrate just how significant these computational predictions are. Below is a comparison across several benchmark systems:
- Conventional superconductors (Nb, Pb, Sn): Critical temperatures typically 1–9 K at ambient pressure. Operate via weak electron-phonon coupling (λ ≈ 0.3–1.0). Require liquid helium cooling — impractical for most applications.
- MgB₂ (discovered 2001): Tc ≈ 39 K at ambient pressure. Revolutionary for demonstrating that light-element compounds can achieve relatively high Tc. λ ≈ 0.87. Still requires cryogenic cooling but represented a conceptual breakthrough.
- Cuprate superconductors (YBCO, BSCCO): Tc up to ~138 K at ambient pressure. Unconventional pairing mechanism (not BCS). Complex layered structures, difficult to fabricate as wires, and the pairing mechanism remains controversial after nearly 40 years.
- H₃S (experimentally confirmed 2015): Tc ≈ 203 K at 155 GPa. First hydride to convincingly demonstrate BCS superconductivity above 200 K. λ ≈ 2.0. Opened the modern era of hydride superconductor research.
- LaH₁₀ (experimentally confirmed 2019): Tc ≈ 250–260 K at 170 GPa. First clathrate hydride to approach room temperature. λ ≈ 2.2. Validated the cage-structure paradigm.
- Ca₂BeH₁₂ (predicted): Tc up to 392.9 K at ~150–200 GPa. λ = 2.8–3.4 — among the strongest electron-phonon coupling ever predicted. If validated, it would be the first superconductor to operate above the boiling point of water.
What sets Ca₂BeH₁₂ apart is not just the raw Tc number but the combination of factors: extraordinarily strong coupling, exceptionally high ωlog, and the ternary composition that may offer synthetic flexibility beyond what binary hydrides provide. The leap from LaH₁₀'s 260 K to a potential 393 K would represent a 50% improvement in operating temperature — a gain of the same magnitude as the original cuprate revolution.
Experimental Validation Roadmap
Computational predictions, however sophisticated, remain predictions until tested in the laboratory. Validating Ca₂BeH₁₂ will require a coordinated experimental campaign across several fronts:
- Diamond anvil cell synthesis: The first critical step is synthesizing the compound at the predicted pressure range (150–200 GPa). This typically involves loading precursor materials — calcium hydride, beryllium hydride, or elemental sources with ammonia borane as a hydrogen reservoir — into a diamond anvil cell and applying laser heating to drive the reaction. Success requires precise stoichiometric control, which is notoriously difficult at megabar pressures.
- X-ray diffraction confirmation: Synchrotron-based X-ray diffraction at facilities like APS, ESRF, or SPring-8 would confirm the predicted clathrate crystal structure. Matching experimental lattice parameters to DFT predictions within ~1% would establish that the correct phase has been synthesized.
- Electrical resistance measurements: The defining test of superconductivity is a sharp drop in electrical resistance to zero at Tc. Four-probe measurements integrated into the diamond anvil cell, while technically demanding, can detect superconducting transitions with high precision.
- Magnetic susceptibility (Meissner effect): A true superconductor must expel magnetic fields. Measuring the Meissner effect in such small sample volumes (~10 μm diameter) is challenging but has been accomplished for LaH₁₀ using specialized SQUID-based techniques and nuclear resonant scattering.
- Isotope effect verification: Replacing hydrogen with deuterium should shift Tc by a predictable factor (approximately 1/√2 in the BCS limit). Observing this isotope effect provides strong evidence for phonon-mediated pairing and helps rule out artifacts.
- Inelastic X-ray or neutron scattering: Directly probing the phonon spectrum would verify the predicted high-frequency Be–H modes and provide experimental values for ωlog.
Realistically, a full validation cycle for a new hydride superconductor takes 2–5 years from first synthesis attempts to peer-reviewed confirmation. The recent controversies surrounding some hydride superconductor claims underscore the importance of independent replication across multiple laboratories before any result can be considered established.
Implications for the Field
If Ca₂BeH₁₂ is experimentally confirmed to superconduct near 393 K, the implications extend far beyond a single material. First, it would validate the ternary hydride design strategy as a systematic path to higher Tc — moving the field from serendipitous discovery to rational materials engineering. The computational pipeline that predicted Ca₂BeH₁₂ (crystal structure prediction via evolutionary algorithms, DFT-based electron-phonon calculations, anharmonic corrections via stochastic self-consistent harmonic approximation) could then be applied systematically to explore the vast combinatorial space of metal–hydrogen compounds.
Second, the result would rein

![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)
![[Battery Lab | Week 3 Day 1] Li-S with CNT Matrix - AI Lab Simulation](/content/images/size/w600/2026/04/lab_battery_Li_S_with_CNT_Matrix_1.png)