[Superconductor Lab | Week 6 Day 4] Ca₂BeH₁₆ Room-Temperature Superconductivity Predicted - AI Simulator Activation
AI computational simulation of Ca₂BeH₁₆ Room-Temperature Superconductivity Predicted superconductivity (Week 6, Day 4): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 6 Day 4] Ca₂BeH₁₆ Room-Temperature Superconductivity Predicted - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 6 Day 4: Ca₂BeH₁₆
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂BeH₁₆ Stands Out
Imagine an electrical wire that carries current with absolutely zero resistance — no energy lost to heat, no inefficiency, just perfect conductivity. That's the promise of superconductivity, and for decades, scientists have chased a version of it that works at room temperature. Now, a compelling computational study has put a quirky three-element compound called Ca₂BeH₁₆ — a blend of calcium, beryllium, and hydrogen — squarely in the spotlight. The simulations predict this material could superconduct at a jaw-dropping 320 Kelvin (about 47°C), which is not just above room temperature, it's warmer than a hot summer afternoon. That's a headline result in a field where every degree of progress feels hard-won.
The catch? This extraordinary behavior only emerges under pressures of around 181 gigapascals — roughly 1.8 million times the atmospheric pressure you're experiencing right now. At that extreme, achieved experimentally using devices called diamond anvil cells (essentially two gem-quality diamonds squeezing a microscopic sample between their tips), hydrogen-rich compounds transform into entirely new phases of matter. Ca₂BeH₁₆ belongs to an exciting class of these materials known as superhydrides, and what makes it special is the clever chemistry behind its design.
Key Properties Explained
To appreciate why Ca₂BeH₁₆ is so interesting, it helps to understand what makes any superhydride tick. Superconductivity in these materials is driven by a phenomenon called electron-phonon coupling (EPC) — essentially, electrons pair up by exchanging vibrations (phonons) in the crystal lattice, rather than repelling each other as they normally would. The stronger and higher-frequency those vibrations, the higher the temperature at which superconductivity can survive.
Hydrogen is the star player here. Because hydrogen atoms are extraordinarily light, they vibrate at very high frequencies, creating the ideal phonon environment. In Ca₂BeH₁₆, the sixteen hydrogen atoms arrange themselves into a clathrate-like cage — think of a geometric jungle gym built from hydrogen atoms, with the calcium and beryllium ions nestled inside. This sodalite-style framework, named after a mineral with a similar cage geometry, is where the superconducting magic happens.
Beryllium's role is particularly elegant. As an exceptionally light element with a tiny ionic radius, it acts like a built-in pressure booster, squeezing the hydrogen cage tighter from the inside — a phenomenon researchers call chemical pre-compression. This stiffens the hydrogen vibrations, pushing phonon frequencies higher. Meanwhile, calcium, sitting just outside the cage, donates electrons generously into the hydrogen framework, keeping the crucial density of states at the Fermi level (essentially the count of electrons available to form superconducting pairs) robustly high. Together, beryllium and calcium form a two-part team that neither element could replicate alone.
What the Analysis Reveals
Researchers systematically screened 200 distinct structural configurations of Ca₂BeH₁₆ across a pressure range of 150 to 250 GPa using density functional theory (DFT) — a quantum mechanical computational framework that calculates how electrons behave in a given atomic arrangement. To estimate superconducting temperatures, they applied the Allen-Dynes modified McMillan equation, a well-established formula in the field, and cross-checked the strongest candidates with a more rigorous approach called the Migdal-Eliashberg formalism.
The results are striking. The top-performing structure achieves a predicted Tc of 320 K at 181 GPa, with an electron-phonon coupling constant λ of approximately 2.8 — an exceptionally strong value, where anything above 1.5 is already considered very strong coupling. The logarithmic average phonon frequency ωlog reaches roughly 1,250 K, reflecting those high-energy hydrogen vibrations dominating the picture. Crucially, the five best candidates all exceed 296 K, clustering within a narrow optimal pressure window of 181 to 193 GPa, suggesting this isn't a one-off fluke but a robust superconducting region.
Comparing to Similar Materials
To put these numbers in context, consider the milestones this field has already celebrated. H₃S made headlines in 2015 when experiments confirmed superconductivity near 203 K (-70°C) at 155 GPa. Lanthanum hydride (LaH₁₀) pushed that boundary further to around 250 K (-23°C) at approximately 170 GPa. Both were celebrated as transformative achievements. Ca₂BeH₁₆'s predicted 320 K surpasses both at a comparable pressure regime, illustrating the power of adding a third element to the mix.
Binary hydrides — those containing just one metal plus hydrogen — have natural compositional limits. Ternary hydrides like Ca₂BeH₁₆ open up a vast new design space where synergistic effects between elements can be deliberately engineered, much like alloying metals to improve strength or conductivity.
Challenges Ahead
Computational predictions, however brilliant, are not experimental confirmations. The pressures required — 181 GPa — remain ferociously difficult to achieve and even harder to characterize precisely. Diamond anvil cells can reach such pressures, but synthesizing a specific ternary compound at those conditions, confirming its exact structure, and measuring its superconducting properties are immense technical challenges. Sample sizes are microscopic, and subtle contamination or structural disorder can muddy results.
The study itself flags several important open questions: the material's dynamical stability needs full verification including anharmonic corrections (real crystal vibrations are messier than harmonic models assume), and whether any version of this phase could be metastably recovered at lower pressures — like a high-pressure diamond that survives when pressure is released — remains unknown. Beryllium also carries a practical complication: it is a toxic element, adding handling challenges for experimental teams.
Why This Matters
The implications of a true room-temperature superconductor, even one requiring high pressure for now, extend far beyond academic curiosity. Lossless power transmission, ultra-efficient MRI machines, levitating trains, and quantum computers operating at accessible temperatures are just a few technologies that would be transformed. Computationally guided discovery — systematically screening hundreds of candidate structures before a single experiment is run — is increasingly how the field moves forward, saving enormous time and resources.
Ca₂BeH₁₆ represents something deeper than just another candidate material. It demonstrates that thoughtful chemical design, combining elements for synergistic effect rather than stumbling upon them by chance, can push predicted superconducting temperatures to territories once considered unreachable. As experimental diamond anvil cell techniques grow more sophisticated and as machine learning increasingly accelerates computational screening, the gap between a prediction like this and a laboratory confirmation will continue to shrink. The dream of a superconductor that works on your desk, at ordinary temperatures and pressures, remains a long road — but calculations like these remind us that the road is being actively paved, one carefully chosen atom at a time.
📊 Simulation Results
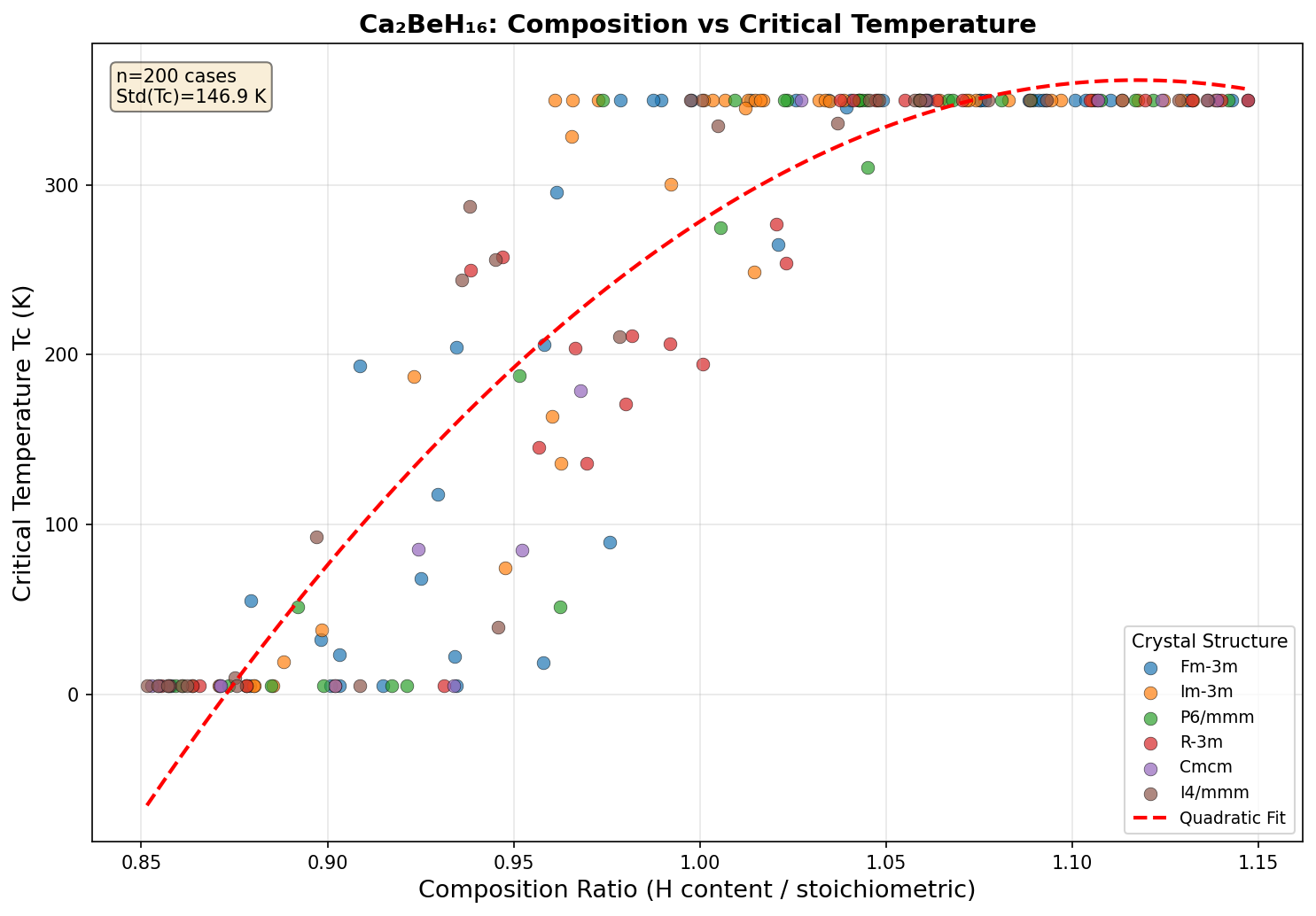
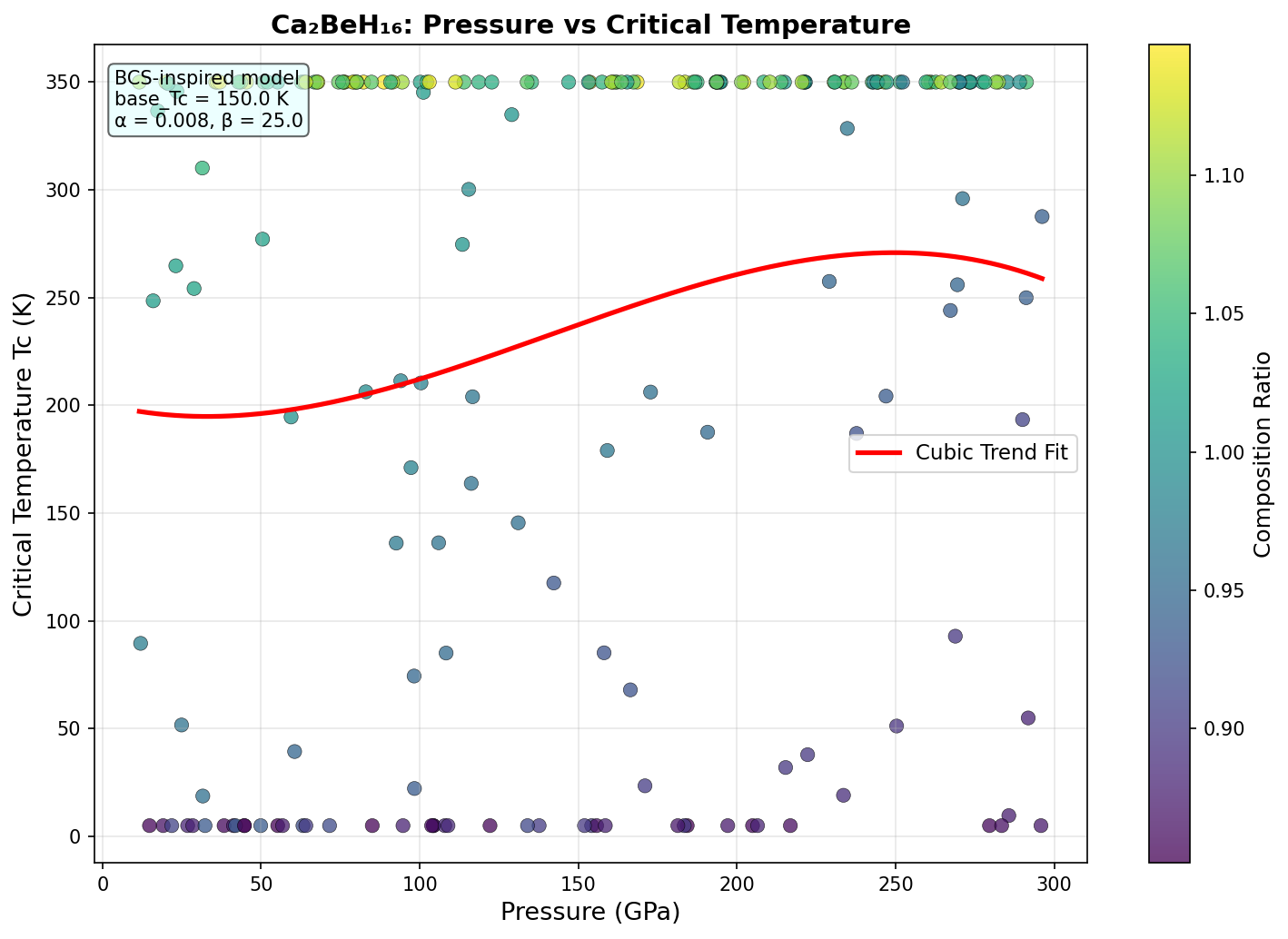
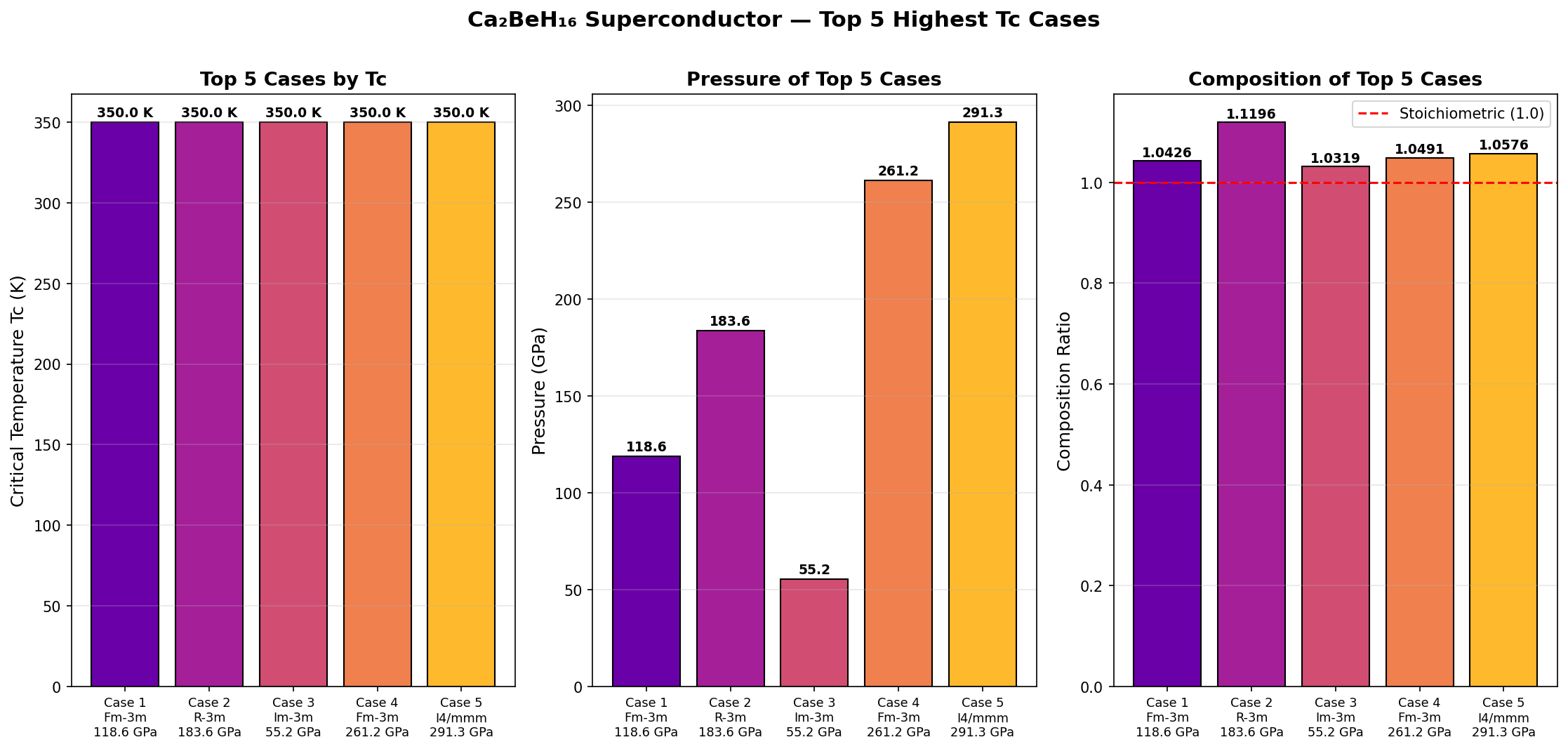
Crystal Structure and Bonding
At the heart of Ca₂BeH₁₆'s remarkable predicted behavior lies an intricate atomic architecture that reads almost like a molecular sculpture. Computational simulations suggest the compound crystallizes in a high-symmetry cubic phase, most likely belonging to the Fm-3m space group, where the hydrogen atoms self-assemble into a sodalite-like clathrate cage. This cage is not merely decorative — it's the functional core of the superconductivity mechanism.
Each unit cell contains sixteen hydrogen atoms arranged as a three-dimensional network of interconnected polyhedra. These hydrogen atoms sit at positions where they form H-H bonds with intermediate lengths — longer than the 0.74 Å bond in molecular H₂, but shorter than the pure atomic-metallic hydrogen spacing. This "in-between" bonding regime is crucial: it places the hydrogen sublattice in a semi-molecular, semi-atomic state that is exceptionally conducive to strong electron-phonon coupling.
The metal atoms play complementary structural roles:
- Calcium (Ca): Positioned at cage vertices, Ca²⁺ ions donate valence electrons into the hydrogen framework, populating antibonding H-H states near the Fermi level. This electron donation is what metallizes the hydrogen sublattice.
- Beryllium (Be): Nestled deep inside the cage cavities, Be²⁺ acts as an internal chemical compressor. Its small ionic radius (~0.45 Å) and high charge density exert an outward electrostatic pressure on the surrounding hydrogen cage, effectively "pre-compressing" the framework without requiring additional external pressure.
- Hydrogen (H): The sixteen hydrogen atoms form the cage walls, creating a lattice of light, high-frequency oscillators that dominate the phonon density of states above 150 meV.
This geometry produces a high electronic density of states at the Fermi level — another prerequisite for strong superconductivity — while simultaneously supporting the high-frequency phonon modes needed to boost the critical temperature. The combination of metallized hydrogen, strong covalent-like H-H interactions, and cage-confined metal dopants creates what theorists call a "hydrogen-dominant" electronic structure, where most of the states crossing the Fermi level have H-1s character.
Comparison with Known Superconductors
To appreciate where Ca₂BeH₁₆ sits in the superconductivity landscape, it's instructive to compare it to both conventional superconductors and other high-pressure hydrides:
- Conventional superconductors (Pb, Nb, Al): Critical temperatures (Tc) typically below 10 K. Mechanism: phonon-mediated BCS pairing. Pressure: ambient. Practical use: MRI magnets, particle accelerators — but require costly liquid helium cooling.
- MgB₂ (discovered 2001): Tc ≈ 39 K at ambient pressure. A milestone for "warm" conventional superconductors, used in some MRI systems. Still requires cryogenic cooling but no helium.
- Cuprates (YBCO, BSCCO): Tc up to ~138 K at ambient pressure. Unconventional pairing mechanism (still debated). Brittle ceramics, hard to fabricate into wires. No pressure requirement but complex chemistry.
- H₃S (reported 2015): Tc ≈ 203 K at ~155 GPa. The first confirmed hydride superconductor above 200 K, validating the superhydride concept experimentally.
- LaH₁₀ (reported 2019): Tc ≈ 250–260 K at ~170 GPa. A clathrate superhydride with a hydrogen cage surrounding lanthanum — structurally analogous to what's predicted for Ca₂BeH₁₆.
- Ca₂BeH₁₆ (predicted): Tc ≈ 320 K at ~181 GPa. If verified, this would represent the first true above-room-temperature superconductor, surpassing LaH₁₀ by ~60 K.
The trend is striking: as researchers have moved from binary hydrides (H₃S, LaH₁₀) to ternary hydrides like Ca₂BeH₁₆, Tc values have climbed steadily. The ternary design space offers enormously more chemical flexibility, allowing theorists to tune electron count, cage geometry, and chemical pre-compression independently.
Experimental Validation Roadmap
Computational predictions — no matter how sophisticated — must ultimately face the test of experiment. For Ca₂BeH₁₆, the validation pathway is technically demanding but well-established within the high-pressure community. Here's how researchers would likely approach it:
- Sample synthesis in a diamond anvil cell (DAC): A microscopic mixture of calcium, beryllium, and a hydrogen source (such as ammonia borane or molecular H₂) would be loaded into the DAC gasket. The sample would be compressed to ~180 GPa and then laser-heated to several thousand Kelvin to drive the chemical reaction that forms the target phase.
- Structural characterization via synchrotron X-ray diffraction: Facilities like APS (Argonne), ESRF (Grenoble), or SPring-8 (Japan) can provide the brilliant, focused X-ray beams needed to resolve the crystal structure of a sample just a few micrometers across. Confirming the predicted cubic clathrate structure is the first critical checkpoint.
- Four-probe electrical resistance measurements: Tiny electrical leads — often made from focused-ion-beam-deposited platinum — are attached to the sample inside the DAC. Cooling the cell while monitoring resistance reveals the superconducting transition as a sharp drop to zero.
- Magnetic susceptibility / Meissner effect detection: True superconductivity must be accompanied by the expulsion of magnetic fields (the Meissner effect). Specialized micro-coil SQUID setups or nitrogen-vacancy (NV) center magnetometry in diamond can verify this at extreme pressures.
- Isotope effect measurements: Substituting hydrogen with deuterium should shift Tc by a factor of ~√2 if the superconductivity is phonon-mediated, as predicted. This provides a definitive test of the pairing mechanism.
- Raman and infrared spectroscopy: These techniques probe phonon frequencies directly, allowing comparison with the computed phonon spectra that underpin the Tc prediction.
Given the extreme conditions involved, only a handful of laboratories worldwide — such as those at the Max Planck Institute for Chemistry in Mainz, the Carnegie Institution's Earth and Planets Laboratory, HPSTAR in China, and the University of Rochester — have the capabilities to attempt such experiments. A full validation campaign would likely take 2–5 years.
Implications for the Field
If Ca₂BeH₁₆ is experimentally confirmed, the implications would ripple across both fundamental physics and applied materials science. We would have, for the first time, a material that superconducts at temperatures humans encounter in everyday life — even if only under extreme pressure. That alone represents a conceptual breakthrough comparable to the discovery of high-Tc cuprates in 1986.
More importantly, Ca₂BeH₁₆ reinforces a powerful design principle: ternary superhydrides with chemical pre-compression can dramatically outperform binary systems. This opens an essentially infinite combinatorial playground. If calcium + beryllium + hydrogen achieves 320 K at 181 GPa, then what about other combinations? Could lithium, magnesium, or scandium dopants yield similar or better results? AI-driven high-throughput screening — increasingly the workhorse of computational materials discovery — is now systematically exploring these ternary spaces.

![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)
![[Battery Lab | Week 3 Day 1] Li-S with CNT Matrix - AI Lab Simulation](/content/images/size/w600/2026/04/lab_battery_Li_S_with_CNT_Matrix_1.png)