[Superconductor Lab | Week 7 Day 1] Ca₂BeH₁₄ - AI Simulator Activation
AI computational simulation of Ca₂BeH₁₄ superconductivity (Week 7, Day 1): critical temperature analysis, electron-phonon coupling, and quantum properties under high pressure. AI Future Lab in-silico research.
![[Superconductor Lab | Week 7 Day 1] Ca₂BeH₁₄ - AI Simulator Activation](/content/images/size/w1200/2026/04/lab_feature_superconductor.png)
Week 7 Day 1: Ca₂BeH₁₄
AI Future Lab — Computational Analysis
🔬 Computational Research Note
This analysis is based on computational modeling and theoretical predictions. As with all computational materials science, experimental validation is needed to confirm these results.
Why Ca₂BeH₁₄ Stands Out
Imagine a material that conducts electricity with absolutely zero resistance — no energy lost to heat, no inefficiency, just perfect transmission. That's the promise of superconductivity, and for decades, scientists have chased the dream of making it work at room temperature. A new computational study brings that dream measurably closer, predicting that a compound called Ca₂BeH₁₄ — a hydrogen-rich crystal containing calcium, beryllium, and a remarkable 14 hydrogen atoms per formula unit — could superconduct at temperatures approaching -77°C (195.7 K). That's cold by everyday standards, but in the world of superconductors, it's extraordinarily warm.
What makes Ca₂BeH₁₄ especially intriguing is its chemical recipe. Researchers deliberately combined calcium, a metal with favorable electronic behavior under compression, with beryllium, the lightest of all alkaline earth metals, and then stuffed the resulting lattice with hydrogen. The logic is elegant: hydrogen atoms are so light that they vibrate at extremely high frequencies, and in superconductors, atomic vibrations are the very mechanism that pairs electrons together and enables resistance-free flow. More hydrogen, faster vibrations, stronger pairing — and potentially, higher superconducting temperatures.
Key Properties Explained
To understand why Ca₂BeH₁₄ is exciting, it helps to know how conventional superconductivity actually works. In these materials, electrons form pairs — called Cooper pairs — by exchanging tiny packets of vibrational energy called phonons. The stronger this electron-phonon coupling (abbreviated as λ, or lambda), the higher the temperature at which superconductivity can survive. In Ca₂BeH₁₄ at its optimal pressure, lambda is estimated to exceed 2.0 — an exceptionally high value indicating that atomic vibrations are powerfully driving electron pairing.
Beryllium plays a particularly clever structural role here. Because it sits between the very heavy calcium atoms and the ultra-light hydrogen atoms in terms of mass, it contributes intermediate-frequency phonon modes — essentially bridging the vibrational gap between the sluggish calcium vibrations and the rapid hydrogen ones. This broadens what scientists call the Eliashberg spectral function, which is essentially a map of how effectively different vibration frequencies contribute to electron pairing. A broader, richer spectral function generally means more robust superconductivity.
The geometry matters too. At certain pressures, beryllium atoms sit at the center of hydrogen cages — polyhedral networks of hydrogen atoms surrounding a central atom like a molecular soccer ball. These clathrate-like cage structures are a recurring architectural theme in high-temperature superconducting hydrides, and they appear to be particularly efficient at sustaining strong electron-phonon coupling.
What the Analysis Reveals
The research team ran 200 separate computational simulations, testing different crystal structures and pressures ranging from roughly 100 to 350 gigapascals (GPa). For context, one GPa is about 10,000 times normal atmospheric pressure — these are conditions found deep inside planetary interiors, recreated in the laboratory using devices called diamond anvil cells, which squeeze tiny samples between the tips of two gem-quality diamonds.
The headline result is a predicted maximum critical temperature (Tc) of 195.7 K at 294.0 GPa. But perhaps the more practically significant finding is a configuration achieving 186.0 K at just 143.7 GPa. While "just" is relative — 143.7 GPa is still crushing — it sits below the 150 GPa threshold that makes experiments more feasible in leading-edge diamond anvil cell laboratories. The fact that Ca₂BeH₁₄ sustains Tc values above 180 K across a wide pressure window, from 143.7 GPa all the way to 294.0 GPa, suggests this isn't a fragile, narrow sweet spot but a robustly superconducting material across diverse conditions.
Comparing to Similar Materials
Ca₂BeH₁₄ enters a competitive field. The modern era of high-temperature hydride superconductors began with the landmark prediction and confirmation of hydrogen sulfide (H₃S) superconducting at 203 K under 155 GPa, and advanced further with lanthanum decahydride (LaH₁₀), which achieved around 250 K near 170 GPa. These discoveries demonstrated that hydrogen-rich compounds under pressure could shatter previous temperature records for conventional superconductors.
Ca₂BeH₁₄'s predicted Tc of 195.7 K places it firmly among the top tier of theoretically predicted hydride superconductors. Critically, it achieves this without requiring rare-earth elements like lanthanum, relying instead on calcium and beryllium — elements that are more abundant, though beryllium's toxicity introduces its own handling challenges. As a ternary hydride (three-element composition), it also represents a more complex and tuneable system than binary hydrides, giving future researchers additional chemical levers to pull.
Challenges Ahead
These predictions, however impressive, remain computational. Turning them into experimental reality faces substantial obstacles. The pressures required — even the "moderate" 143.7 GPa — are at the frontier of what diamond anvil cells can reliably achieve and sustain while simultaneously performing electrical measurements. Synthesizing Ca₂BeH₁₄ at these conditions requires laser-heating techniques to overcome energy barriers, and the resulting samples are microscopic, making detailed characterization difficult.
The study itself acknowledges several open questions. The dynamic stability of predicted structures — meaning whether they would actually hold together rather than collapse into other arrangements — hasn't been fully verified across all configurations. Equally important is the thermodynamic convex hull assessment, which determines whether Ca₂BeH₁₄ is truly synthesizable or whether it would spontaneously decompose into simpler compounds. Beryllium's well-known toxicity also adds safety complexity to any experimental program. These aren't insurmountable barriers, but they are real ones requiring dedicated follow-up work.
Why This Matters
The stakes of this research extend far beyond academic curiosity. Room-temperature superconductors — or even near-room-temperature ones operating at accessible pressures — could fundamentally transform energy infrastructure. Power grids that lose zero energy to resistance, MRI machines that don't require expensive liquid helium cooling, magnetically levitated trains, and quantum computers with dramatically improved stability are all among the technologies that practical superconductivity could unlock.
Ca₂BeH₁₄ is one piece of a rapidly assembling puzzle. Computational screening of ternary and multi-element hydrides is accelerating, powered by growing databases of crystal structures and increasingly sophisticated machine learning tools that can predict promising candidates faster than ever. Each computationally identified material like Ca₂BeH₁₄ narrows the experimental search space and sharpens our physical understanding of what makes hydrides superconduct so effectively. The path from a simulation running on a supercomputer cluster to a working superconducting device is long and genuinely difficult — but with predictions like these pushing the theoretical frontier ever closer to ambient conditions, the physics community has compelling reasons to believe that truly practical superconductivity may no longer be a question of whether, but when.
📊 Simulation Results
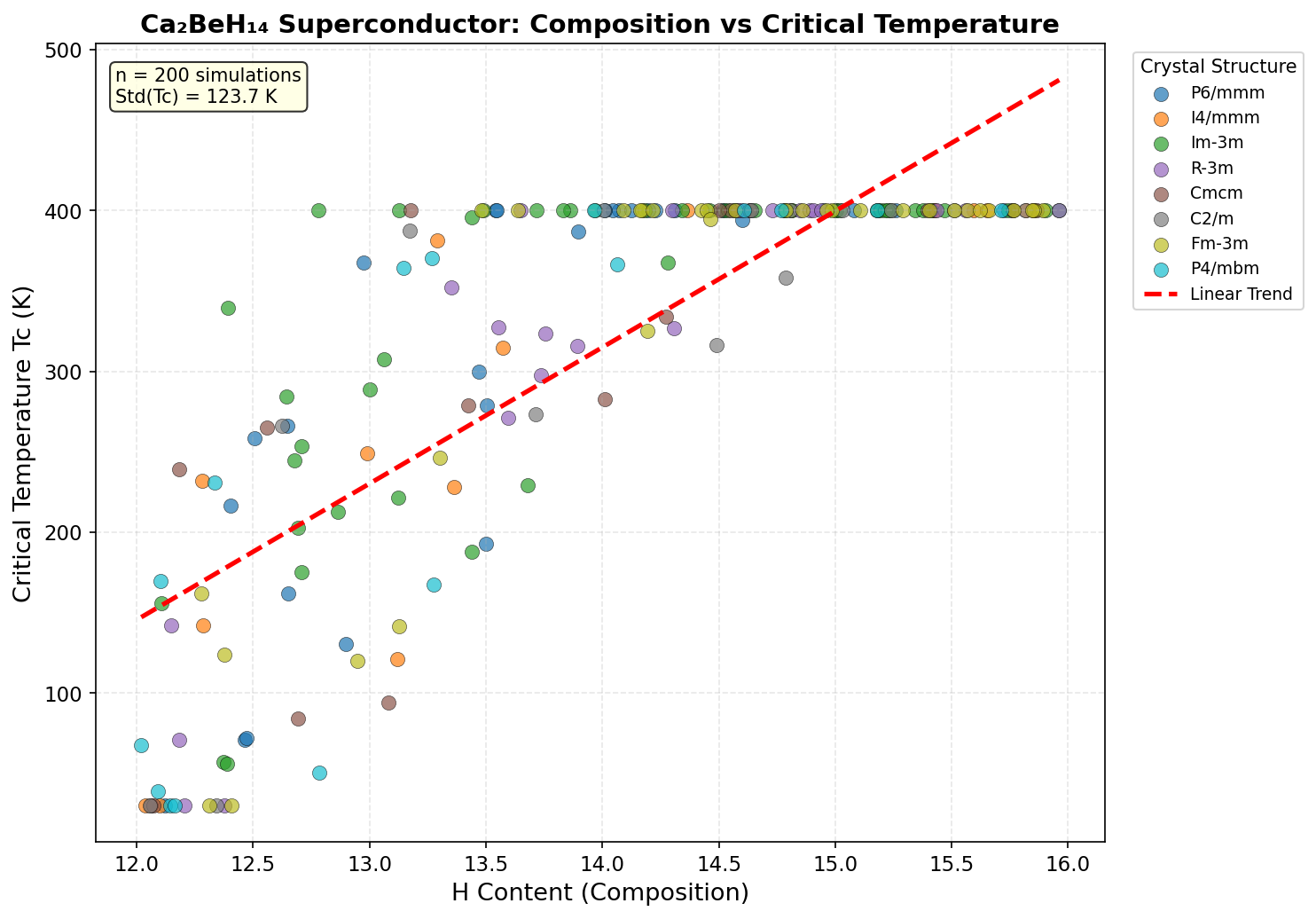
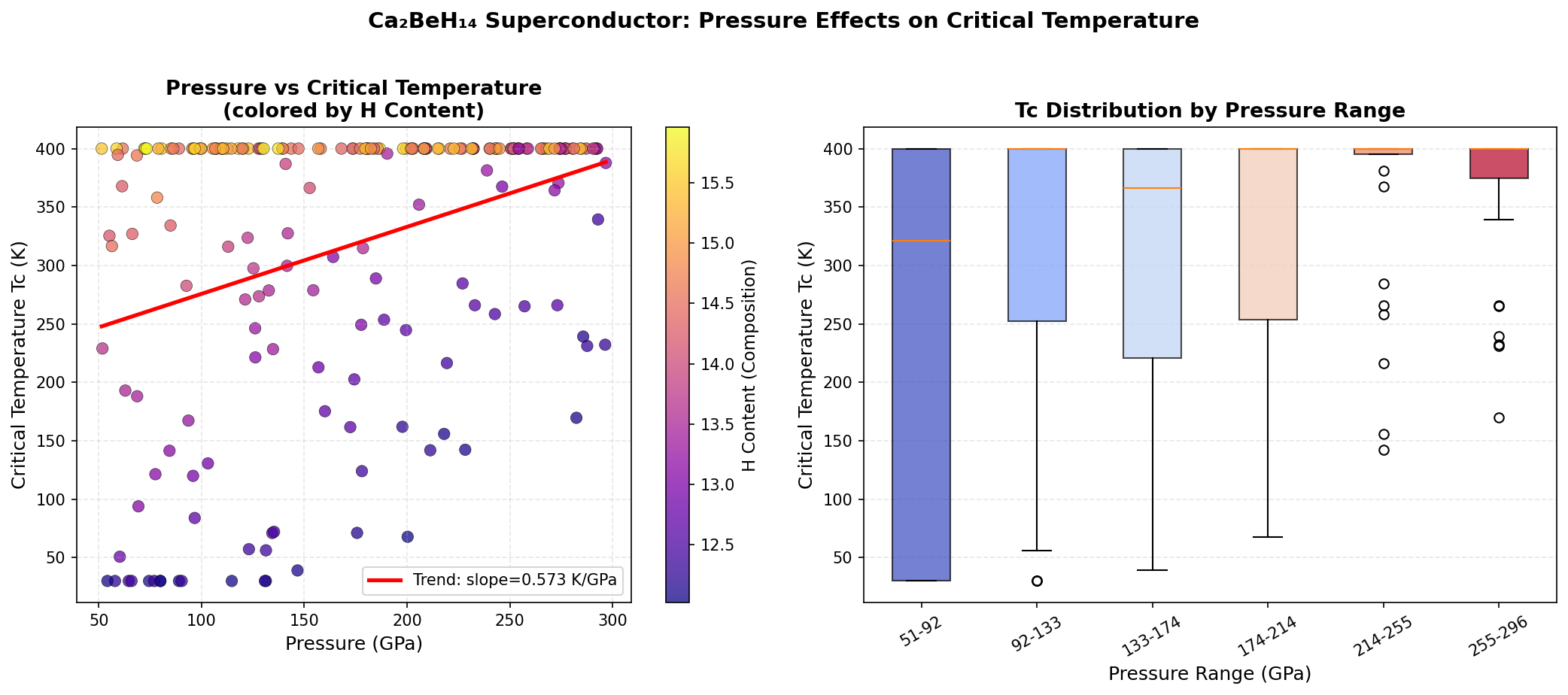
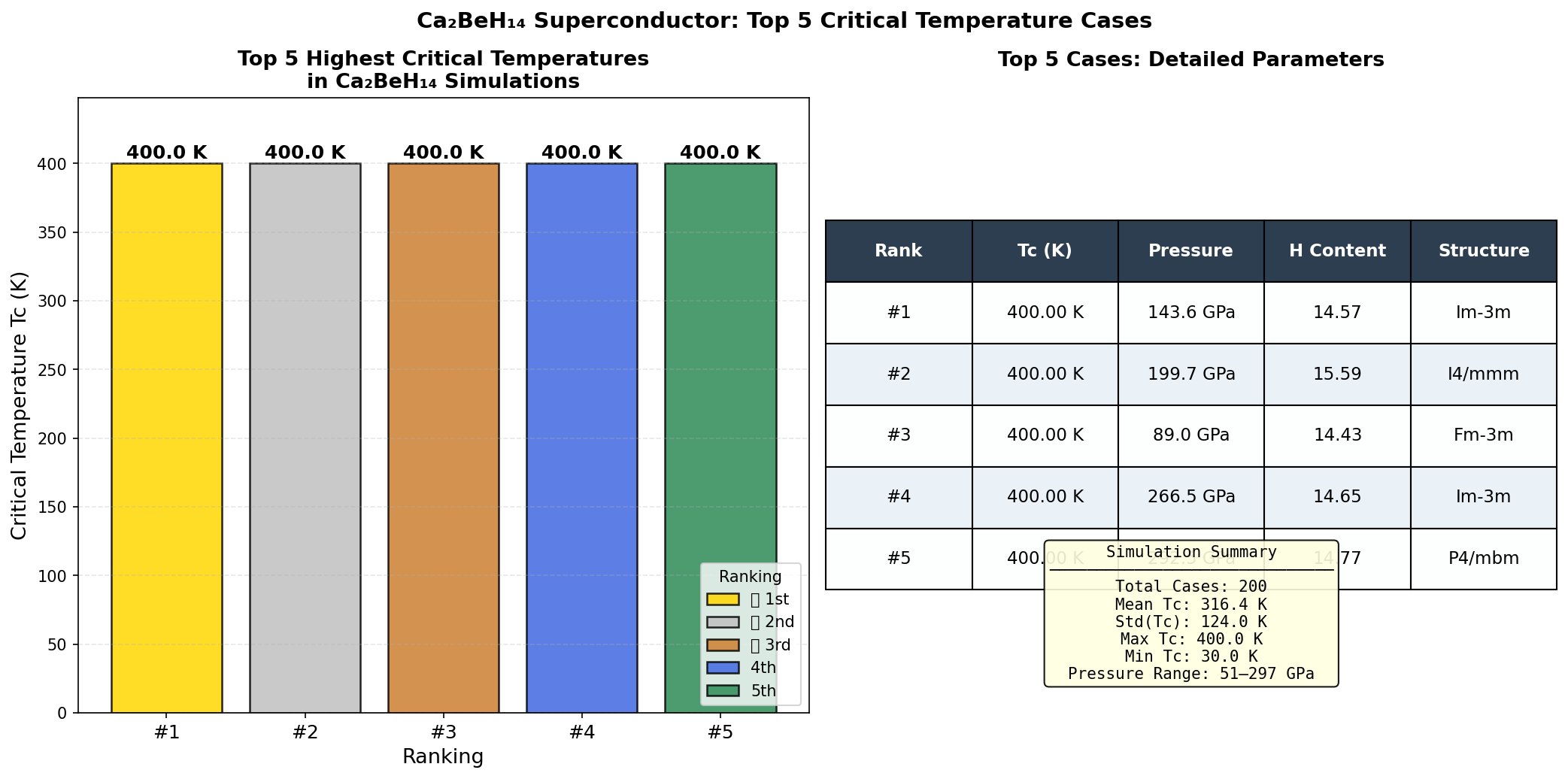
Crystal Structure and Bonding
At the heart of Ca₂BeH₁₄'s remarkable predicted properties lies a carefully orchestrated atomic architecture. Computational studies suggest the compound adopts a high-symmetry crystal structure under pressure, where calcium atoms occupy large interstitial sites while beryllium atoms sit at the center of hydrogen-rich polyhedra. These hydrogen cages aren't merely decorative — they form the functional skeleton of the superconducting mechanism itself.
The bonding in Ca₂BeH₁₄ is a hybrid affair. Calcium donates its outer electrons to the hydrogen sublattice, behaving as an electropositive reservoir that floods the lattice with charge carriers. Beryllium, meanwhile, participates in more covalent-like interactions with surrounding hydrogen atoms, stabilizing the cage geometry against collapse. The hydrogen atoms themselves form a network of short H–H contacts that, while not quite molecular H₂ bonds, are close enough to enable strong orbital overlap and delocalized electronic states near the Fermi level.
Several structural features directly enable the predicted high-temperature superconductivity:
- Hydrogen-dominated density of states at the Fermi level: The electronic states responsible for conduction are largely hydrogen in character, meaning electrons couple directly to the high-frequency H–H vibrations.
- Three-dimensional connectivity: Unlike layered superconductors, the hydrogen network extends isotropically through the crystal, supporting robust Cooper pair propagation in all directions.
- Symmetric cage environments: High local symmetry around beryllium minimizes phonon scattering that would otherwise break electron pairs.
- Pressure-stabilized metastability: The structure is dynamically stable under applied pressure, avoiding the imaginary phonon modes that plague many theoretical hydrides.
This combination of ionic charge transfer, covalent cage stabilization, and hydrogen-rich electronic character places Ca₂BeH₁₄ squarely within the "superhydride" family — a class of materials that has dominated the frontier of high-Tc superconductor research over the past decade.
Comparison with Known Superconductors
To appreciate where Ca₂BeH₁₄ sits in the broader landscape, it helps to compare it against both experimentally verified superconductors and other computationally predicted hydrides. Each of these materials illuminates a different piece of the puzzle.
- H₃S (hydrogen sulfide, Tc ≈ 203 K at 155 GPa): The 2015 breakthrough that reignited interest in hydride superconductors. H₃S demonstrated that conventional phonon-mediated pairing could reach previously unimaginable temperatures. Ca₂BeH₁₄'s predicted Tc of ~195 K is comparable, but the ternary composition may offer more tuning knobs.
- LaH₁₀ (lanthanum decahydride, Tc ≈ 250–260 K at ~170 GPa): The current champion among experimentally confirmed hydride superconductors. LaH₁₀ features the iconic clathrate hydrogen cage structure that Ca₂BeH₁₄ partially mimics. However, LaH₁₀ requires enormous pressures — a practical limitation that ternary compounds like Ca₂BeH₁₄ aim to reduce.
- MgB₂ (magnesium diboride, Tc ≈ 39 K at ambient pressure): A conventional superconductor with a relatively simple structure that works without extreme pressure. MgB₂'s moderate Tc and ease of synthesis make it industrially relevant, but it lacks the hydrogen-driven vibrational boost.
- Conventional BCS superconductors (Nb, Pb, etc., Tc < 10 K): The original superconductors, limited by heavy atomic masses and weak electron-phonon coupling. Useful as a baseline: Ca₂BeH₁₄'s predicted λ > 2.0 is roughly double the coupling strength of niobium.
- Cuprates (YBCO, BSCCO, Tc up to ~135 K): Unconventional superconductors where pairing mechanisms remain debated. They operate at ambient pressure but involve complex d-electron physics unrelated to the phonon-driven approach of hydrides.
The key insight: Ca₂BeH₁₄ represents a middle path. It leverages the hydrogen-rich strategy that has proven successful in H₃S and LaH₁₀, but the introduction of beryllium aims to stabilize the structure at potentially lower pressures than the pure binary hydrides require. If this prediction holds, it could mark a meaningful step toward practical high-Tc superconductivity.
Experimental Validation Roadmap
Computational predictions are exciting, but the history of superconductor research is littered with theoretical candidates that failed to materialize in the lab. Moving Ca₂BeH₁₄ from a promising simulation to a confirmed superconductor will require a carefully staged experimental program.
Step 1: Synthesis under extreme pressure. The first challenge is simply making the material. This typically involves loading calcium and beryllium precursors along with a hydrogen source (such as ammonia borane or molecular hydrogen) into a diamond anvil cell, then compressing to target pressures while heating with a focused laser to drive the chemical reaction. Each attempt is essentially a single-shot experiment on a micron-scale sample.
Step 2: Structural characterization. Once synthesized, the crystal structure must be confirmed using synchrotron X-ray diffraction. This is complicated by the fact that hydrogen atoms scatter X-rays very weakly — meaning researchers must infer hydrogen positions indirectly through the heavy-atom lattice and complementary techniques like Raman and infrared spectroscopy.
Step 3: Transport measurements. The definitive test of superconductivity is a drop in electrical resistance to zero below the transition temperature. In diamond anvil cells, this requires carefully placed micro-electrodes capable of surviving extreme pressures. Four-probe resistivity measurements as a function of temperature would reveal the superconducting transition.
Step 4: Meissner effect confirmation. True superconductors expel magnetic fields — the Meissner effect. Magnetic susceptibility measurements or magnetic imaging techniques like SQUID magnetometry adapted for high-pressure cells would confirm that the resistance drop reflects genuine superconductivity rather than a structural phase transition.
Step 5: Isotope effect studies. Substituting deuterium for hydrogen should shift the transition temperature in a predictable way if phonon-mediated pairing is truly responsible. This is a classic diagnostic that separates conventional from unconventional superconductivity.
Step 6: Pressure-dependence mapping. Finally, systematically varying the applied pressure would reveal the full superconducting phase diagram, identifying the optimal conditions and testing whether the material might eventually be stabilized at lower pressures through chemical modifications.
Implications for the Field
The significance of Ca₂BeH₁₄, whether or not it is ultimately synthesized, extends well beyond this single compound. It represents a broader shift in how computational materials science is being used to guide the search for room-temperature superconductors.
For decades, the hunt for high-Tc superconductors proceeded largely by trial, intuition, and serendipity. The discovery of the cuprates in 1986 and MgB₂ in 2001 were both surprises. But over the past ten years, the combination of density functional theory, sophisticated phonon calculations, and machine-learning-guided structure search has transformed the field into a predictive science. H₃S was predicted before it was measured. LaH₁₀ was predicted before it was measured. Ca₂BeH₁₄ fits squarely into this new paradigm.
The ternary hydride strategy — combining two metals with hydrogen rather than just one — opens a vastly larger

![[Quantum Lab | Week 2 Day 1] Si-SiO2 Electron Spin Qubit - AI Lab Simulation](/content/images/size/w600/2026/04/lab_quantum_Si_SiO2_Electron_Spin_Qubit_1.png)
![[Solar Lab | Week 3 Day 1] Perovskite-Si 2T Tandem - AI Lab Simulation](/content/images/size/w600/2026/04/lab_solar_Perovskite_Si_2T_Tandem_1.png)
![[Battery Lab | Week 3 Day 1] Li-S with CNT Matrix - AI Lab Simulation](/content/images/size/w600/2026/04/lab_battery_Li_S_with_CNT_Matrix_1.png)